Abstract
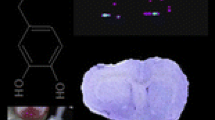
Imaging mass spectrometry (IMS) is a powerful technique that combines the chemical and spatial analysis of surface materials. It allows spatial localization of peptides, proteins or lipids that are recorded in parallel without the need of a label. It is currently one of the most rapidly developing techniques in the proteomics toolbox. In the present study, accurate mass matrix-assisted laser desorption/ionization imaging mass spectrometry (MALD IMS) was used for direct molecular mapping of nervous tissue at micrometer spatial resolution. Cryosections of the whole brain of the terrestrial snail, Helix pomatia, were placed on indium-tin-oxide (ITO)-coated conductive glass slides and covered with a thin layer of α-cyano-4-hydroxycinnamic acid (CHCA) matrix by electro spray deposition. High-resolution molecular ion maps of well-known neuropeptides, such as FMRFamide were constructed. FMRFamide is known to exert powerful modulatory effect on synaptic transmission in molluscs. FMRFamide was predominantly localized in the cluster of neurons in the pro-, meso- and postcerebral regions of cerebral ganglia, pedal ganglia and right parietal ganglia of the central nervous system. Our present study, using MALDI IMS confirmed the distribution of FMRFamide containing cells in the Helix central nervous system previously detected by antibody dependent immunohistochemistry.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aebersold, R., Mann, M. (2003) Mass spectrometry-based proteomics. Nature 422, 198–207.
Altelaar, A. F., Luxembourg, S. L., McDonnell, L. A., Piersma, S. R., Heeren, R. M. (2007) Imaging mass spectrometry at cellular length scales. Nature Prot. 2, 1185–1196.
Bastiaens, P. I. H., Pepperkok, R. (2000) Observing proteins in their natural habitat: the living cell. Trends Biochem. Sci. 25, 631–637.
Caprioli, R., Farmer, T., Gile, G. (1997) Molecular imaging of biological samples: localization of peptides and proteins using MALDI-TOF MS. Anal. Chem. 69, 4751–4760.
Chaurand, P., Cornett, D. S., Caprioli, R. M. (2006) Molecular imaging of thin mammalian tissue sections by mass spectrometry. Curr. Opin. Biotechnol. 17, 431–436.
Chaurand, P., Schriver, K. E., Caprioli, R. M. (2007) Instrument design and characterization for high resolution MALDI-MS imaging of tissue sections. J. Mass. Spectrom. 42, 476–489.
Cooke, I. R., Gelperin, A. (1988) Distribution of FMRFamide-like immunoreactivity in the nervous system of the slug Limax maximus. Cell Tissue Res. 253, 69–76.
Croll, R. P., van Minnen, J. (1992) Distribution of the peptide Ala-Pro-Gly-Trp-NH2 (APGWamide) in the nervous system and periphery of the snail Lymnaea stagnalis as revealed by immunocytochemistry and in situ hybridization. J. Comp. Neurol. 324, 567–574.
De Lange, R. P. J., van Minnen, J. (1997) Diversity in cell specific co-expression of four neuropeptide genes involved in control of male copulation behaviour in Lymnaea stagnalis. J. Neurosci. 78, 289–299.
Elekes, K., Nässel, D. R. (1990) Distribution of FMRFamide-like immunoreactive neurons in the central nervous system of the snail Helix pomatia. Cell Tissue Res. 262, 177–190.
Elekes, K., Ude, J. (1993) An immunogold electron microscopic analysis of FMRFamide-like immunoreactive neurons in the CNS of Helix pomatia: ultrastructure and synaptic connections. J. Neurocytol. 22, 1–13.
Hanash, S. (2003) Disease proteomics. Nature 422, 226–232.
Horobin, R. W. (2002) Biological staining: mechanisms and theory. Histochem. 77, 3–13.
Ikeda, T., Kiss, T., Hiripi, L., Fujisawa, Y., Kubota, I., Muneoka, Y. (1991) MIP (Mytilus inhibitory peptide) analogues isolated from the ganglia of the pulmonate mollusc Helix pomatia. In: Shimonishi, Y. (ed.) Peptide Chemistry 1990. Protein Research Foundation, pp. 357–362.
Ikeda, T., Kuroki, Y., Kubota, I., Minakata, H., Nomoto, K., Miki, W., Kiss, T., Hiripi, L., Muneoka, Y. (1992) SSFVRIamide peptides–A new family of neuropeptides distributed interphyletically. In: Suzuki, A. (ed.) Peptide Chemistry 1991. Protein Research Foundation, pp. 65–70.
Ikeda, T., Minakata, H., Fujita, T., Muneoka, Y., Kiss, T., Hiripi, L., Nomoto, K. (1993) Neuropeptides isolated from Helix pomatia Part 1. Peptides related to MIP, buccalin, myomodulin-CARP and SCP. In: Yanaihara, N. (ed.) Peptide Chemistry 1992 (Proceedings of the 2nd Japan Symposium). ESCOMScience Publishers B.V., Leiden, The Netherlands, pp. 576–578.
Kaletas, B. K., van der Wiel, I. M. (2009) Sample preparation issues for tissue imaging by imaging MS. Proteomics 9, 2622–2633.
Kiss, T., Pirger, Z. (2006) Neuropeptides as modulators and hormones in terrestrial snails: Their occurrence, distribution and physiological significance. In Satake, H. (ed.) Invertebrate neuropeptides and hormones: basic knowledge and recent advances. Transworld Research Network, Kerala, India, pp. 75–110.
Kobayashi, M., Fujimoto, K., Kubota, I. (1991) Structure and action of novel neuropeptides isolated from a pulmonate, the African giant snail. In: Kits, K. S., Boer, H. H., Joose, J. (eds) Molluscan Neurobiology. North-Holland Publishing Company, Amsterdam, pp. 260–264.
Kobayashi, M., Muneoka, Y. (1990) Structure and action of molluscan neuropeptides. Zool. Sci. 7, 801–814.
McDonnell, L. A., Piersma, S. R., Maarten Altelaar, A. F., Mize, T. H., Luxembourg, S. L., Verhaert, P. D., van Minnen, J., Heeren, R. M. (2005) Subcellular imaging mass spectrometry of brain tissue. J. Mass. Spectrom. 40, 160–168.
Meyer, T., Teruel, M. N. (2003) Fluorescence imaging of signaling networks. Trends Cell Biol. 13, 101–106.
Minakata, H., Ikeda, T., Fujita, T., Kiss, T., Hiripi, L., Muneoka, Y., Nomoto, K. (1993) Neuropeptides isolated from Helix pomatia Part 2. FMRFamide-related peptides, S-Iamide peptides FR peptides and others. In: Yanaihara, N. (ed.) Peptide Chemistry 1992 (Proceedings of the 2nd Japan Symposium). ESCOMScience Publishers B.V., Leiden, pp. 579–582.
Pirger, Z., Lubics, A., Reglodi, D., Laszlo, Z., Mark, L., Kiss, T. (2010) Mass spectrometric analysis of activity-dependent changes of neuropeptide profile in the snail, Helix pomatia. Neuropeptides 44, 475–483.
Suzuki, H., Kimura, T., Sekiguchi, T., Mizukami, A. (1997) FMRFamide-like-immunoreactive primary sensory neurons in the olfactory system of the terrestrial mollusc, Limax marginatus. Cell Tissue Res. 289, 339–345.
Wouters, F. S., Verveer, P. J., Bastiaens, P. I. H. (2001) Imaging biochemistry inside cells. Trends Cell Biol. 11, 203–211.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented during the 12th ISIN Symposium on Invertebrate Neurobiology, August 31–September 4, 2011, Tihany, Hungary.
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Márk, L., Maasz, G. & Pirger, Z. High Resolution Spatial Distribution of Neuropeptides by Maldi Imaging Mass Spectrometry in the Terrestrial Snail, Helix pomatia. BIOLOGIA FUTURA 63 (Suppl 2), 113–122 (2012). https://doi.org/10.1556/ABiol.63.2012.Suppl.2.15
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.63.2012.Suppl.2.15