Abstract
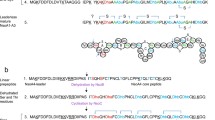
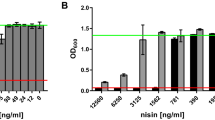
The lantibiotic nisin is a ribosomally synthesised and post-translationally modified antimicrobial peptide produced by strains of Lactococcus lactis, and used as safe and natural preservative in food industry. The nisA structural gene encodes ribosomally synthesised and biologically inactive a 57 amino acid precursor peptide (NisA) which undergoes several post-translational modifications. In this study, we report the expression of precursor nisin as a His6-tagged peptide in Escherichia coli and its purification using a nickel affinity column. The technique of spliced-overlap extension PCR was used to amplify the nisA gene and the T7 promoter region of pET-15b vector. This approach was used to introduce six histidine residues at the C-terminus of prenisin. The identity of the expressed peptide was confirmed by N-terminal sequencing. The expressed His-tagged prenisin was purified under denaturing conditions, and named as prenisin-His6. The purified prenisin-His6 was analyzed by SDS-PAGE, Western blotting and mass spectroscopy. These results showed that the nisin precursor peptide can be successfully produced using an E. coli expression system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Buchman, G. W., Banerjee, S., Hansen, J. N. (1988) Structure, expression and evolution of a gene encoding the precursor of nisin, a small protein antibiotic. J. Biol. Chem. 263, 16260–16266.
Bycroft, B. W., Chan, W. C., Roberts, G. C. K. (1991) Synthesis and characterization of pro- and prepeptides related to nisin and subtilin. In: Jung, G., Sahl, H. G. (eds) Nisin and Novel Lantibiotics. ESCOM, Leiden, pp. 204–217.
Casadaban, M. J., Cohen, S. N. (1980) Analysis of gene control signals by DNA fusion and cloning in Escherichia coli. J. Mol. Biol. 138, 179–207.
Cotter, P. D., Hill, C., Ross, R. P. (2005) Bacteriocins: Developing innate immunity for food. Nat. Rev. Microbiol. 3, 777–788.
Delves-Broughton, J., Blackburn, P., Evans, R. J., Hugenholtz, J. (1996) Applications of the bacteriocin, nisin. Antonie van Leeuwenhoek 69, 193–202.
Dodd, H. M., Horn, N., Gasson, M. J. (1990) Analysis of the genetic determinant for the peptide antibiotic nisin. J. Gen. Microbiol. 136, 555–566.
Field, D., Hill, C., Cotter, P. D., Ross, R. P. (2010) The dawning of a ‘Golden era’ in lantibiotic bioengineering. Mol. Microbiol. 78, 1077–1087.
Hirel, P. H., Schmitter, M. J., Dessen, P., Fayat, G., Blanquet, S. (1989) Extent of N-terminal methionine excision from Escherichia coli proteins is governed by the side-chain length of the penultimate amino acid. Proc. Natl. Acad. Sci. USA 86, 8247–8251.
Horn, N., Martinez, M. I., Martinez, J. M., Hernandez, P. E., Gasson, M. J., Rodriguez, J. M., Dodd, H. M. (1998) Production of pediocin PA-1 by Lactococcus lactis using the lactococcin A secretory apparatus. Appl. Environ. Microbiol. 64, 818–823.
Kaletta, C., Entian, K. D. (1989) Nisin, a peptide antibiotic: cloning and sequencing of the nisA gene and post-translational processing of its peptide product. J. Bacteriol. 171, 1597–1601.
Karakas Sen, A., Narbad, A., Horn, N., Dodd, H. M., Parr, A. J., Colquhoun, I., Gasson, M. J. (1999) Post-translational modification of nisin. The involvement of NisB in the dehydration process. Eur. J. Biochem. 261, 524–532.
Kluskens, L. D., Kuipers, A., Rink, R., de Boef, E., Fekken, S., Driessen, A. J. M., Kuipers, O. P., Moll, G. N. (2005) Post-translational modification of therapeutic peptides by NisB, the dehydratase of the lantibiotic nisin. Biochemistry-US 44, 12827–12834.
Koponen, O., Tolonen, M., Qiao, M., Wahlström, G., Helin, J., Saris, P. E. J. (2002) NisB is required for the dehydration and NisC for the lanthionine formation in the post-translational modification of nisin. Microbiology 148, 3561–3568.
Kuipers, A., de Boef, E., Rink, R., Fekken, S., Kluskens, L. D., Driessen, A. J. M., Leenhouts, K., Kuipers, O. P., Moll, G. N. (2004) NisT, the transporter of the lantibiotic nisin, can transport fully modified, dehydrated, and unmodified prenisin and fusions of the leader peptide with non-lantibiotic peptides. J. Biol. Chem. 279, 22176–22182.
Kupke, T., Stevanovic, S., Ottenwalder, B., Metzger, J. W., Jung, G., Gotz, F. (1993) Purification and characterization of EpiA, the peptide substrate for post-translational modifications involved in epidermin biosynthesis. FEMS Microbiol. Lett. 112, 43–48.
Langridge, J., Langridge, P., Bergquist, P. L. (1980) Extraction of nucleic acids from agarose gels. Anal. Biochem. 103, 264–271.
Majer, F., Schmid, D. G., Altena, K., Bierbaum, G., Kupke, T. (2002) The flavoprotein MrsD catalyzes the oxidative decarboxylation reaction involved in formation of the peptidoglycan biosynthesis inhibitor mersacidin J. Bacteriol. 184, 1234–1243.
Nilsson, B. L., Soellner, M. B., Raines, R. T. (2005) Chemical synthesis of proteins. Annu. Rev. Biophys. Biomol. Struct. 34, 91–118.
Van der Meer, J. R., Polman, J., Beerthuyzen, M. M., Siezen, R. J., Kuipers, O. P., de Vos, W. M. (1993) Characterization of the Lactococcus lactis nisin A operon genes nisP, encoding a subtilisinlike serine protease involved in precursor processing, and nisR, encoding a regulatory protein involved in nisin biosynthesis. J. Bacteriol. 175, 2578–2588.
Xie, L., Miller, L. M., Chatterjee, C., Averin, O., Kelleher, N. L., van der Donk, W. A. (2004) Lacticia 481: In vitro reconstruction of lantiobiotic segwwwWbase activity. Science 303, 679–681.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Karakas-Sen, A., Narbad, A. Heterologous Expression and Purification of NisA, the Precursor Peptide of Lantibiotic Nisin from Lactococcus Lactis. BIOLOGIA FUTURA 63, 301–310 (2012). https://doi.org/10.1556/ABiol.63.2012.2.11
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.63.2012.2.11