Abstract
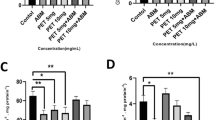
The aim of this research was to examine the applicability of some chronic toxicological tests in the determination of exposure to xenobiotics present in concentrations below No Observed Adverse Effect Level (NOAEL) and below the detection limit of analytical instruments. In the present experiment tested chemicals (Malathion, Aroclor-1254 and 3-methylcholanthrene (3-MC)) were mixed with wheat grains and given to male mice as feed over a period of 12 months. 7-ethoxyresorufin-O-deethylase (EROD) activity with the 3-MC and Aroclor-1254 treatments reached the peak at 9th month of exposure (26.7 and 42.4 pmol−1 mgprot−1, respectively), while malathion did not have significant influence. Glutathione (GSH) level depletion was highest after three months of exposure. Unexpectedly, acetylcholinesterase (AChE) activity increased after treatment with malathion, an organophosphorous insecticide. In conclusion, low-level concentrations chronically administered exert certain effects on the levels of selected enzymes, e.g. biomarkers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmed, R. S., Seth, V., Pasha, S. T., Banerjee, B. D. (2000) Influence of dietary ginger (Zingiber officinales Rosc) on oxidative stress induced by malathion in rats. Food Chem. Toxicol. 38, 443–450.
Akhgari, M., Abdollahi, M., Kebryaeezadeh, A., Hosseini, R., Sazevari, O. (2003) Biochemical evidence for free radical-induced lipid peroxidation as a mechanism for subchronic toxicity of malathion in blood and liver of rats. Hum. Exp. Toxicol. 22, 205–211.
Alvares, A. P., Kappas, A. (1977) The inducing properties of polychlorinated biphenyls on hepatic monooxygenases. Clin. Pharmacol. Ther. 22, 809–816.
Amer, S. M., Fahmy, M. A., Aly, F. A. E., Farghaly, A. A. (2002) Cytogenetic studies on the effect of feeding mice with stored wheat grains treated with malathion. Mutat. Res. 513, 1–10.
Binelli, A., Ricciardi, F., Riva, C., Provini, A. (2006) New evidences for old biomarkers: Effects of several xenobiotics on EROD and AChE activities in Zebra mussel (Dreissena polymorpha). Chemosphere 62, 510–519.
Bocquené, C., Bellanger, C., Cadiou, Y., Galgani, F. (1995) Joint action of combinations of pollutants on the acetylcholinesterase activity of several marine species. Ecotoxicology 4, 266–279.
Bradford, M. M. (1976) Arapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254.
Cohen, S. D. (1980) Mechanisms of toxicological interactions involving organophosphate insecticides. Fundam. Appl. Toxicol. 4, 315–324.
Day, K. E., Scott, I. M. (1990) Use of acetylcholinesterase activity to detect sublethal toxicity in stream invertebrates exposed to low concentrations of organophosphate insecticides. Aquat. Toxicol 18, 101–113.
De Sousa, G., Fontaine, F., Pralavorio, M., Botta-Fridlund, D., Letreut, Y., Rahmani, R. (1997) Insecticides cytotoxicity and CYP1A1/2 induction in primary human and rat hepatocytes cultures. Toxicol. In Vitro 11, 451–457.
De Vos, R. H., Van Dokkum, W., Schouten, A., De Jong-Berkhout, P. (1990) Polycyclic aromatic hydrocarbons in dutch total diet samples (1984-1986). Food Chem. Toxicol. 28, 263–268.
Delescluse, C., Ledirac, N., de Sousa, G., Pralavorio, M., Botta-Fridlund, D., Letreut, Y., Rahmani, R. (1997) Comparative study of CYP1A1 induction by 3-methylcholanthrene in various human hepatic and epidermal cell types. Toxicol. In Vitro 11, 443–450.
Deshpande, S. S. (2002) Handbook of Food Toxicology. Marcel Dekker Inc., New York, pp. 829.
Ellman, G. L. (1959) Tissue sulphydryl group. Arch. Biochem. Biophys. 82, 70–77.
Ellman, G. L., Courtney, K. D., Andres, Jr., V., Featherstone, R. M. (1961) A new and rapid colori-metric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95.
Elton, R. C., Garle, M. J., Fry, J. R. (2000) Glutathione depletion in a liver microsomal assay as an in vitro biomarker for reactive metabolite formation. Biomarkers 5, 285–294.
EPA (1995) Great Lakes Water Quality Initiative Technical Support Document for Wildlife Criteria, EPA-820-B-95-009. U.S. Environmental Protection Agency, Office of Water, Washington, D.C.
Fisher, T., Crane, M., Callaghan, A. (2003) Induction of cytochrome P450 activity in individual Chirnomus riparius larvae exposed to xenobiotics. Ecotoxicol. Environ. Saf. 54, 1–6.
Forsyth, C. S., Chambers, J. E. (1989) Activation and degradation of the phosphorothionate insecticides parathion and EPN by rat brain. Biochem. Pharmacol. 38, 1597–1603.
Hackenberger, B. K., Jaric-Perkusic, D., Stepic, S. (2008) Effect of temephos on Cholinesterase activity in the earthworm Eisenia fetida (Oligochaeta, Lumbricidae). Ecotoxicol. Environ. Saf. 71, 583–589.
Hamilton, D. J., Holland, P. T., Ohlin, B., Murray, W. J., Amrus, A., Baptista, G. C., Kovacicova, J. (1997) Optimum use of available residue data in the estimation of dietary intake of pesticides. Pure Appl. Client. 69, 1373–1410.
Hazarika, A., Sarkar, S. N, Hajare, S., Kataria, M., Malik, J. K. (2003) Influence of malathion pre-treatment on the toxicity of anilofos in male rats: a biochemical interaction study. Toxicology 185, 1–8.
Hyne, R. V., Maher, W. A. (2003) Invertebrate biomarkers: links to toxicosis that predict population decline. Ecotoxicol. Environ. Saf. 54, 366–374.
Jimenez, B. D., Stegeman, J. J. (1990) Detoxication enzymes as indicators of environmental stress on fish. In: Adams, S. M. (ed.) Biological Indicators of Stress in Fish. Am. Fish Soc. Symp. 8, pp. 67–79.
John, S., Kale, M., Rathore, N., Bhatnagar, D. (2001) Protective effect of vitamin E in dimethoate and malathion induced oxidative stress in rat erythrocytes. J. Nutr. Biochem. 12, 500–504.
Jung, D. K., Klauss, T., Fent, K. (2001) Cytochrome P450 induction by nitrated hydrocarbons, aza-renes and binary mixtures in fish hepatoma cell line PLHC-1. Environ Toxicol. Chem. 20, 149–159.
Larsson, B. K. (1986) Formation of polycyclic aromatic hydrocarbons during the smoking and grilling of food. Prag. Clin. Biol. Res. 206, 169–180.
Lin, L.-F. H., Mismer, D., Lile, J. D, Armes, L. G., Butler, III, E. T., Collins, F. (1989) Purification, cloning and expression of ciliary neurotrophic factor (CNTF). Science 246, 1023–1025.
Lindström-Seppä, P., Korytko, P. J., Hahn, M. E., Stegeman, J. J. (1994) Uptake of waterborne 3,3’,4,4-’tetrachlorobiphenyl and organ and cee-specific induction of cytochrome P4501 Ain adult and larval fathead minnow Pimephales promelas. Aquat. Toxicol. 28, 147–167.
Matthews, H. B. (1982) In: Jakoby, W. B, Bend, J. R., Caldwell, J. (eds) Aryl Halides. Metabolic Basis of Detoxification. Metabolism of Functional Groups. Academic Press, New York, pp. 51–68.
Meister, A. (1985) The fall and rise of cellular glutathione levels: enzyme-based approaches. Curr. Top Cell. Regul. 26, 383–394.
Mondon, J. A., Nowak, B. F., Sodergren, A. (2001) Persistent organic pollutants in oysters Crassostrea gigas and sand flathead Platycephalus bassensis from tasmanian estuarine and coastal waters. Mar. Pollut.Bull. 42, 157–164.
Moore, A., Scott, A. P., Lower, N., Katsiadaki, I., Greenwood, L. (2003) The effects of 4-nonylphenol and atrazine on Atlantic salmon (Salmo salar L.) smolts. Aquaculture 222, 253–263.
Muthuvel, R., Venkataraman, P., Krishnamoorthy, G., Gunadharini, D. N., Kanagaraj, P., Stanley, A. J. (2006) Antioxidant effect of ascorbic acid on PCB (Aroclor 1254) induced oxidative stress in hypothalamus of albino rats. Clin. Chim. Acta 365, 297–303.
Nachtomi, E., Lipstein, B., Kary, S. (1989) Toxicological aspects of activated sludge feeding. Bull. Environ. Contam. Toxicol. 43, 879–885.
Newman, S. L., Guzelian, P. S. (1983) Identification of the cyanopregnenolone inducible form of hepatic cytochrome P-450 as a catalyst of Aldrin epoxidation. Biochem. Pharmacol. 32, 1529–1531.
Omura, T., Sato, R. (1964) The carbon monooxide-binding pigment of liver microsomes. Evidence for its hemoprotein nature. J. Biol. Chem. 239, 2370–2378.
Pedrajas, J. R., Peinado, J., Lopez-Barea, J. (1995) Oxidative stress in fish exposed to model xenobiotics. Oxidatively modified forms of Cu, Zn-superoxide dismutase as potential biomarkers. Chem. Biol. Interact. 98, 267–282.
Preston, B. D., Miller, J. A., Miller, E. C. (1984) Reactions of 2,2’,5,5’-tetrachlorobiphenyl-3,4-oxide with methionine, cysteine and glutathione in relation to the formation of methylthio-metabolites of 2,2’,5,5’-tetrachlorobiphenyl in the rat and mouse. Chem. Biol. Interact. 50, 289–294.
Rattner, B. A., Melancon, M. J., Custer, T. W., Hothem, R. L., King, K. A., LeCaptain, L. J., Spann, J. W., Woodin, B. R., Stegeman, J. J. (1993) Biomonitoring environmental contamination with pipping black-crowned night-heron embryos: induction of cytochrome P450. Environ. Toxicol. Chem. 12, 1719–1732.
Reidy, G. F., Rose, H.A., Stacey, N. H. (1987) Effect of length of exposure to malathion on xenobiotic biotransformation in male rat liver. Toxicol. Lett. 38, 193–199.
Robbins, C. (1991) Poisoned Harvest: A Consumers Guide to Pesticide Use and Abuse. Victor Gollancz Ltd., pp. 300–313.
Ruus, A., Ugland, K. I., Skaare, J. U. (2002) Influence of trophic position on organochlorine concentrations and compositional patterns in a marine food web. Environ. Toxicol. Chem. 21, 2356–2364.
Schelnk, D., DiGuilio, R. T. (2002) In: Adams, S. M. (ed.) Biochemical Responses as Indicators of Aquatic Ecosystem Health. Biological Indicators of Aquatic Ecosystem Stress. AFS, Bethesda, pp. 14–17.
Sridevi, N., Venkataraman, R, Senthilkumar, K., Krishnamoorthy, G., Arunakaran, J. (2007) Oxidative stress modulates membrane bound ATPases in brain regions of PCB (Aroclor 1254) exposed rats: protective role of atocopherol. Biomed. Pharmacother 61, 435–440.
Srikanth, N. S., Seth, P. K. (1990) Alterations in xenobiotic metabolizing enzymes in brain and liver of rats coexposed to endosulfan and malathion. J. Appl. Toxicol. 10, 157–160.
Tsakiris, S., Angelogianni, P., Schulpis, K. H., Stavridis, J. C. (2000) Protective effect of L-phenyl-alanine on rat brain acetylcholinesterase inhibition induced by free radicals. Clin. Biochem. 3, 103–106.
Uygun, U., Koksel, H., Atli, A. (2005) Residue levels of malathion and ist metabolites and fenitro-thion in post-harvest treated wheat during storage, milling and baking. Food Chem. 92, 643–647.
Venkataraman, P., Krishnamoorthy, G., Vengatesh, G., Srinivasan, N., Aruldhas, M. M., Arunakaran, J. (2008) Protective role of melatonin on PCB (Aroclor 1254) induced oxidative stress and changes in acetylcholine esterase and membrane bound ATPases in cerebellum, cerebral cortex and hippocampus of adult rat brain. Int. J. Dev. Neurosci. 26, 585–591.
Venkataraman, P., Muthuvel, R., Krishnamoorthy, G., Arunkumar, A., Sridhar, M., Srinivasan, N., Balasubramanian, K, Aruldhas, M. M., Arunakaran, J. (2007) PCB (Aroclor 1254) enhances oxidative damage in rat brain regions: protective role of ascorbic acid. Neurotoxicology 28, 490–498.
Vincent, D. R., Bradshaw, W. S., Booth, G. H., Seegmiller, R. E., Allen, S. D. (1992) Effect of PCB and DES on rat monoamine oxidase, acetylcholinesterase, testosterone, and estradiol ontogeny. Bull. Environ. Contam. Toxicol. 48, 884–893.
White, R. D, Hahn, M. E., Lockhart, W. L., Stegeman, J. J. (1994) Catalytic and immunochemical characterization of hepatic microsomal cytochromes P450 in beluga whale (Delphinapterus leucas). Toxicol. Appl. Pharmacol. 126, 45–57.
Wilkinson, C. F., Murray, M., Marcus, C. B. (1984) Interactions of methylenedioxyphenyl compounds with cytochrome P-450 and effects of microsomal oxidation. Rev. Biochem. Toxicol. 6, 27–63.
Yawetz, A., Benedek-Segal, M., Woodin, B. (1997) Cytochrome P-4501A immunoassay in freshwater turtles and exposure to PCBs and environmental pollutants. Environ. Toxicol. Chem. 16, 1802–1806.
Yu, Z., Xu, M., Santana-Rios, G., Shen, R., Izquierdo-Pulido, M., Williams, D. E., Dashwood, R. H. (2001) A comparison of whole wheat, refined wheat and wheat bran as inhibitors of heterocyclic amines in the Salmonella mutagenicity assay and in the rat colonic aberrant crypt focus assay. Food Chem. Toxicol. 39, 655–665.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hackenberger, B.K., Jarić, D., Hackenberger, D. et al. Effects of Chronic Dietary Exposure to a Low-Dose of Malathion, Aroclor-1254 and 3-Methylcholanthrene on Three Biomarkers in Male Mice. BIOLOGIA FUTURA 61, 423–433 (2010). https://doi.org/10.1556/ABiol.61.2010.4.6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.61.2010.4.6