Abstract
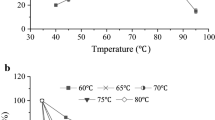
To elucidate how temperature effects subsite mapping of a thermostable α-amylase from Bacillus licheniformis (BLA), a comparative study was performed by using 2-chloro-4-nitrophenyl (CNP) β-maltooligosides with degree of polymerisation (DP) 4–10 as model substrates. Action patterns, cleavage frequencies and subsite binding energies were determined at 50 °C, 80 °C and 100 °C. Subsite map at 80 °C indicates more favourable bindings compared to the hydrolysis at 50 °C. Hydrolysis at 100 °C resulted in a clear shift in the product pattern and suggests significant differences in the active site architecture. Two preferred cleavage modes were seen for all substrates in which subsite (+2) and (+3) were dominant, but CNP-G1 was never formed. In the preferred binding mode of shorter oligomers, CNP-G2 serves as the leaving group (79%, 50%, 59% and 62% from CNP-G4, CNP-G5, CNP-G6 and CNP-G7, respectively), while CNP-G3 is the dominant hydrolysis product from CNP-G8, CNP-G9, and CNP-G10 (62%, 68% and 64%, respectively). The high binding energy value (−17.5 kJ/mol) found at subsite (+2) is consistent with the significant formation of CNP-G2. Subsite mapping at 80 °C and 100 °C confirms that there are no further binding sites despite the presence of longer products.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allen, J. D., Thoma, J. A., (1976) Subsite mapping of enzymes. Depolymerase computer modelling. Biochem. J. 159, 105–120.
Allen, J. D., Thoma, J. A., (1976) Subsite mapping of enzymes. Application of the depolymerase computer model to two alpha-amylases. Biochem. J. 159, 121–132.
Brosnan, M. P., Kelly, C. T., Fogarty, W. M., (1992) Investigation of the mechanisms of irreversible thermoinactivation of Bacillus stearothermophilus alpha-amylase. Eur. J. Biochem. 203, 225–231.
Davies, G. J., Wilson, K. S., Henrissat, B. (1997) Nomenclature for sugar-binding subsites in glycosyl hydrolases. Biochem. J. 321, 557–559.
Declerck, N., Machius, M., Chambert, R., Wiegand, G., Huber, R., Gaillardin, C. (1997) Hyperthermostable mutants of Bacillus licheniformis alpha-amylase: thermodynamic studies and structural interpretation. Protein Eng. 10, 541–549.
Farkas, E., Jánossy, L., Harangi, J., Kandra, L., Lipták, A. (1997) Synthesis of chromogenic substrates of alpha-amylases on a cyclodextrin basis. Carbohydr. Res. 303, 407–415.
Guzman-Maldonado, H., Paradez-Lopez, D. (1995) Amylolytic enzymes and products derived from starch: a review. Crit. Rev. Food Sci. Nutr. 35, 373–403.
Gyémánt, G., Hovánszki, G., Kandra, L. (2002) Subsite mapping of the binding region of alpha-amylases with a computer program. Eur. J. Biochem. 269, 5157–5162.
Kandra, L., Gyémánt, G., Pál, M., Petró, M., Remenyik, J., Lipták, A. (2001) Chemoenzymatic synthesis of 2-chloro-4-nitrophenyl beta-maltoheptaoside acceptor-products using glycogen phosphorylase b. Carbohydr. Res. 333, 129–136.
Kandra, L., Gyémánt, G., Remenyik, J., Hovánszki, G., Lipták, A. (2002) Action pattern and subsite mapping of Bacillus licheniformis alpha-amylase (BLA) with modified maltooligosaccharide substrates. FEBS Letters 518, 79–82.
Machius, M., Declerck, N., Huber, R., Wiegand, G. (1998) Activation of Bacillus licheniformis alpha-amylase through a disorder → order transition of the substrate-binding site mediated by a calcium-sodium-calcium metal triad. Structure 6, 281–292.
Marchal, L. M., van de Laar, A. M. J., Goetheer, E., Schimmelpennink, E. B., Bergsma, J., Beeftink, H. H., Tramper, J. (1999) Effect of temperature on the saccharide composition obtained after alpha-amylolysis of starch. J. Biotechnol. Bioeng 63, 344–355.
Morgan, F. J., Priest, F. G., (1981) Characterization of a thermostable alpha-amylase from Bacillus licheniformis. J. Appl. Bacteriol. 50, 107–114.
Nakakuki, T., Azuma, K., Kainuma, K. (1985) Action patterns of various exo-amylases and the anomeric configurations of their products. Carbohydr. Res. 128, 297–310.
Saito, N. (1973) A thermophilic extracellular α-amylase from Bacillus licheniformis. Arch. Biochem. Biophys. 155, 290–298.
Tomazik, S. J., Kibanov, A. M., (1988) Mechanisms of irreversible thermal inactivation of Bacillus alpha-amylases. J. Biol. Chem., 263, 3092–3096.
Vieille, C., Burdette D. S., Zeikus, J. G., (1996) Thermozymes. Biotechnol. Annu. Rev. 2, 1–83.
Zale, S. E., Klibanov, A. M., (1986) Why does ribonuclease irreversibly inactivate at high temperatures? Biochemistry 25, 5432–5444.
Acknowledgements
This work was supported by the Hungarian Scientific Research Fund (OTKA T043499, T047075), and a scholarship from the Hungarian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kandra, L., Remenyik, J., Gyémánt, G. et al. Effect of Temperature on Subsite Map of Bacillus licheniformis α-Amylase. BIOLOGIA FUTURA 57, 367–375 (2006). https://doi.org/10.1556/ABiol.57.2006.3.10
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.57.2006.3.10