Abstract
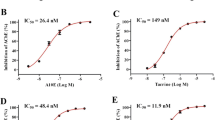
Eight inhibitors of acetylcholinesterase (AChE), tacrine, bis-tacrine, donepezil, rivastigmine, galantamine, heptyl-physostigmine, TAK-147 and metrifonate, were compared with regard to their effects on AChE and butyrylcholinesterase (BuChE) in normal human brain cortex. Additionally, the IC50 values of different molecular forms of AChE (monomeric, G1, and tetrameric, G4) were determined in the cerebral cortex in both normal and Alzheimer’s human brains. The most selective AChE inhibitors, in decreasing sequence, were in order: TAK-147, donepezil and galantamine. For BuChE, the most specific was rivastigmine. However, none of these inhibitors was absolutely specific for AChE or BuChE. Among these inhibitors, tacrine, bis-tacrine, TAK-147, metrifonate and galantamine inhibited both the G1 and G4 AChE forms equally well. Interestingly, the AChE molecular forms in Alzheimer samples were more sensitive to some of the inhibitors as compared with the normal samples. Only one inhibitor, rivastigmine, displayed preferential inhibition for the G1 form of AChE. We conclude that a molecular form-specific inhibitor may have therapeutic applications in inhibiting the G1 form, which is relatively unchanged in Alzheimer’s brain.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Atack, J. R., Perry, E. K., Bonham, J. R., Perry, R. H., Tomlinson, B. E., Blessed, G., Fairbairn, A. (1983) Molecular forms of acetylcholinesterase in senile dementia of Alzheimer’s type: selective loss of the intermediate (10S) form. Neurosci. Lett. 40, 199–204.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254.
Ellman, G. L., Courtney, D. K., Andres, V., Featherstone, R. M. (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95.
Enz, A., Chappuis, A., Probst, A. (1992) Different influence of inhibitors on acetylcholinesterase molecular forms G1 and G4 isolated from Alzheimer’s disease and control brains. In: Shafferman, A., Velan, B. (eds) Multidisciplinary Approaches to Cholinesterase Functions. Plenum Press, New York, pp. 243–249.
Fishman, E. B., Siek, G. C., MacCallum, R. D., Bird, E. D., Volicer, L., Marquis, J. K. (1986) Distribution of the molecular forms of acetylcholinesterase in human brain: alterations in dementia of Alzheimer type. Ann. Neurol. 19, 246–252.
Giacobini, E. (1997) From molecular structure to Alzheimer therapy. Jpn. J. Pharmacol. 74, 225–241.
Giacobini, E. (2001) Do cholinesterase inhibitors have disease-modifying effects in Alzheimer’s disease? CNS Drugs 15, 85–91.
Johnson C. D., Russel, R. L. (1975) A rapid, simple radiometric assay for cholinesterase, suitable for multiple determinations. Anal. Biochem. 64, 229–238.
Kasa, P., Rakonczay, Z., Gulya, K. (1997) The cholinergic system in Alzheimer’s disease. Progr. Neurobiol. 52, 511–535.
Nakamura, S., Kawashima, S., Nakano, S., Tsuji, T., Araki, W. (1990) Subcellular distribution of acetylcholinesterase in Alzheimer’s disease: abnormal localization and solubilization. J. Neural Transm. Suppl. 30, 13–23.
Ogane, N., Giacobini, E., Struble, R. (1992) Preferential inhibition of acetylcholinesterase molecular forms in rat brain. Neurochem. Res. 17, 489–495.
Ogane, N., Giacobini, E., Struble, R. (1992) Differential inhibition of acetylcholinesterase molecular forms in normal and Alzheimer disease brain. Brain Res. 589, 307–312.
Pang, Y. P., Quiram, P., Jelacic, T., Hong, F., Brimijoin, S. (1996) Highly potent, selective, and low cost bis-tetrahydroaminacrine inhibitors of acetylcholinesterase. Steps toward novel drugs for treating Alzheimer’s disease. J. Biol. Chem. 271, 23646–23649.
Rakonczay, Z. (1988) Cholinesterase and its molecular forms in pathological states. Progr. Neurobiol. 31, 311–330.
Rakonczay, Z., Brimijoin, S. (1988) Monoclonal antibodies to human brain acetylcholinesterase: Properties and applications. Cell. Molec. Neurobiol. 8, 85–93.
Rakonczay, Z., Mallol, I., Schenk, H., Vincendon, G., Zanetta, J.-P. (1981) Purification and properties of the membrane-bound acetylcholinesterase from adult rat brain. Biochim. Biophys. Acta 657, 243–256.
Rakonczay, Z., Kovács, I. (1998) Cholinesterases in Alzheimer’s disease and cholinesterase inhibitors in Alzheimer’s therapy. Acta. Biol. Hung. 49, 55–70.
Rakonczay, Z., Vincendon, G., Zanetta, J.-P. (1981) Heterogeneity of rat brain acetylcholinesterase: A study by gel filtration and gradient centrifugation. J. Neurochem. 37, 662–669.
Siek, G. C., Katz, L. S., Fishman, E. B., Korosi, T. S., Marquis, J. K. (1990) Molecular forms of acetylcholinesterase in subcortical areas of normal and Alzheimer’s disease brain. Biol. Psychiatry 27, 573–580.
Skau, K. A. (1981) Ethopropazine inhibition of AChE molecular forms. Pharmacologist 24, 224.
Vigny, M., Bon, S., Massoulie, J., Leterrier, F. (1978) Active-site catalytic efficiency of acetyl-cholinesterase molecular forms in Electrophorus, Torpedo, rat and chicken. Eur. J. Biochem. 85, 317–323.
Younkin, S. G., Goodridge, B., Katz, J., Lockett, G., Nafziger, D., Usiak, M. F., Younkin, L. H. (1986) Molecular forms of acetylcholinesterase in Alzheimer’s disease. Fed. Proc. 45, 2982–2988.
Zhao, Q., Tang, X. C. (2002) Effects of huperzine A on acetylcholinesterase isoforms in vitro: comparison with tacrine, donepezil, rivastigmine and physostigmine. Eur. J. Pharmacol. 455, 101–107.
Acknowledgements
The expert technical assistance of Mrs Mária Sós is gratefully acknowledged. Supported by grants from OTKA (T038346) and ETT (020/2000).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Maria Wollemann on the occasion of her 80th birthday.
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Rakonczay, Z. Potencies and Selectivities of Inhibitors of Acetylcholinesterase and Its Molecular Forms in Normal and Alzheimer’s Disease Brain. BIOLOGIA FUTURA 54, 183–189 (2003). https://doi.org/10.1556/ABiol.54.2003.2.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.54.2003.2.7