Abstract
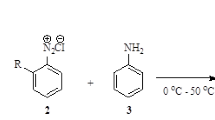
The Sandmeyer reaction of anilines to generate aryl azides, followed by the Ru(porphyrin)CO-catalyzed addition to styrenes affording N-aryl aziridines was successfully performed for the first time in mesoreactors, under continuousflow conditions. Mesofluidic technology allowed for a rapid screening of different parameters and a quick identification of the optimized reaction conditions for the two separate steps. The two optimized reactions were then combined in a single continuous process that allowed a safe and efficient synthesis of N-arylaziridines from convenient commercially available starting materials.
Article PDF
Similar content being viewed by others
References
For reviews, see: (a) Degennaro, L.; Trinchera, P.; Luisi, R. Chem. Rev. 2014, 114, 7881–7929
Callebaut, G.; Meiresonne, T.; De Kimpe, N.; Mangelinckx, S. Chem. Rev. 2014, 114, 7954–8015
Cardoso, A. L.; Pinho e Melo, T. M. V. D. Eur. J. Org. Chem. 2012, 6479–6501
Stankovic, S.; D’hooghe, M.; Catak, S.; Eum, H.; Waroquier, M. I.; Van Speybroeck, V.; De Kimpe, N. J. Chem. Soc. Rev. 2012, 41, 643–665
Ohno, H. Chem. Rev. 2014, 114, 7784–7814.
See ref. 1 and (a) Pellissier, H. Adv. Synth. Catal. 2014, 356, 1899–1935
Chawla, R.; Singh, A. K.; Yadav, L. D. S. RSC Adv. 2013, 3, 11385–11403
Sweeney, J. B.; Yudin, A. Aziridines and Epoxides in Organic Synthesis; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, 2006.
Fingerhut, A.; Serdyuka, O. V.; Tsogoeva, S. B. Green Chem. 2015, 17, 2042–2058
Fantauzzi, S.; Caselli, A.; Gallo, E. Dalton Trans. 2009, 5434–5443.
Recent reviews: (a) Shin, K.; Kim, H.; Chang, S. Acc. Chem. Res. 2015, 48, 1040–1052
Intrieri, D.; Zardi, P.; Caselli, A.; Gallo, E. Chem. Commun. 2014, 50, 11440–11453.
(a) Jin, L.-M.; Xu, X.; Lu, H.; Cui, X.; Wojtas, L.; Zhang, X. P. Angew. Chem., Int. Ed. 2013, 52, 5309–5313
Fukunaga, Y.; Uchida, T.; Ito, Y.; Matsumoto, K.; Katsuki, T. Org. Lett. 2012, 14, 4658–4661
Kim, C.; Uchida, T.; Katsuki, T. Chem. Commun. 2012, 7188–7190
Uchida, T.; Katsuki, T. Chem. Rec. 2014, 14, 117–129
Jung, N.; Bräse, S. Angew. Chem., Int. Ed. 2012, 51, 5538–5540
Cramer, S. A.; Jenkins, D. M. J. Am. Chem. Soc. 2011, 133, 19342–19345.
Smith, C. J.; Smith, C. D.; Nikbin, N.; Ley, S. V.; Baxendale, I. R. Org. Biomol. Chem. 2011, 9, 1927–1937
Delville, M. M. E.; Nieuwland, P. J.; Janssen, P.; Koch, K.; van Hesta, J. C. M.; Rutjes, F. P. J. T Chem. Eng. J. 2011, 167, 556–559.
For recent applications of organic azides to the total synthesis of valuable compounds under continuous flow conditions, see: (a) Pitts, A. K.; O’Hara, F.; Snell, R. H.; Gaunt, M. J. Angew. Chem., Int. Ed. 2015, 54, 5451–5455
Ott, D.; Borukhova, S.; Hessel, V. Green Chem. 2016, 18, 1096–1116.
Selected recent books and reviews: (a) Darvas, F.; Hessel, V.; Dorman, G. Flow Chemistry; De Gruyter: Berlin, 2014
Jensen, K. F.; Reizmana, B. J.; Newman, S. G. Lab Chip 2014, 14, 3206
Wirth, T. Microreactors in Organic Synthesis and Catalysis, 2nd ed.; Wiley-VCH: Weinheim, 2013
Wiles, C.; Watts, P. Green Chem. 2012, 14, 38–54
Wegner, J.; Ceylan, S.; Kirschning, A. Chem. Commun. 2011, 47, 4583–4592.
For recent reviews on steoreoselective catalytic reactions in flow see: (a) Atodiresei, I.; Vila, C.; Rueping, M. ACS Catal. 2015, 5, 1972–1985
Puglisi, A.; Benaglia, M.; Porta, R.; Coccia, F. Curr. Organocatal. 2015, 2, 79–101
Munirathinam, R.; Huskens, J.; Verboom, W. Adv. Synth. Catal. 2015, 357, 1093–1123
Rodríguez-Escrich, C.; Pericàs, M. A. Eur. J. Org. Chem. 2015, 1173–1188
Puglisi, A.; Benaglia, M.; Chiroli, V. Green Chem. 2013, 15, 1790–1813
Tsubogo, T.; Ishiwata, T.; Kobayashi, S. Angew. Chem., Int. Ed. 2013, 52, 6590–6604
Zhao, D.; Ding, K. ACS Catal. 2013, 3, 928–944. For some very recent examples about continuous-flow synthetic methodologies, see
Poh, J.-S.; Tran, D. N.; Battilocchio, C.; Hawkins, J. M.; Ley, S. V. Angew. Chem., Int. Ed. 2015, 54, 7920–7923
Fabry, D. C.; Ronge, M. A.; Rueping, M. Chem. Eur. J. 2015, 21, 5350–5354
Tran, D. N.; Battilocchio, C.; Lou, S. B.; Hawkins, J. M.; Ley, S. V. Chem. Sci. 2015, 6, 1120–1125.
For a comprehensive review on the synthesis of natural products in flow, see: Pastre, J. C.; Browne, D. L.; Ley, S. V. Chem. Soc. Rev. 2013, 42, 8849–8869. For a recent example, see
Newton, S.; Carter, C. F.; Pearson, C. M.; de C. Alves, L.; Lange, H.; Thansandote, P.; Ley, S.V. Angew. Chem., Int. Ed. 2014, 53, 4915–4920.
For recent reviews on the synthesis of API under continuous flow conditions, see: (a) Gutmann, B.; Cantillo, D.; Kappe, C. O. Angew. Chem., Int. Ed. 2015, 54, 6688–6728
Porta, R.; Benaglia, M.; Puglisi, A. Org. Process Res. Dev. 2016, 20, 2–25. For a recent example of a process involving hazardous reagents for the preparation of API in flow, see
De Angelis, S.; De Renzo, M.; Carlucci, C.; Degennaro, L.; Luisi, R. Org. Biomol. Chem. 2016, 14, 4304–4311.
Fortt, R.; Wootton, R. C. R.; de Mello, A. J. Org. Process Res. Dev. 2003, 7, 762–768.
Weber, M.; Yilmaz, G.; Wille, G. Chim. Oggi/Chem. Today 2011, 29, 8–10.
Tsuritani, T.; Mizuno, H.; Nonoyama, N.; Kii, S.; Akao, A.; Sato, K.; Yasuda, N.; Mase, T. Org. Process Res. Dev. 2009, 13, 1407–1412.
Tseberlidis, G.; Zardi, P.; Caselli, A.; Cancogni, D.; Fusari, M.; Lay, L.; Gallo, E.; Organometallics 2015, 34, 3774–3781
Zardi, P.; Savoldelli, A.; Carminati, D.; Caselli, A.; Ragaini, F.; Gallo, E. ACS Catal. 2014, 4, 3820–3823
Zardi, P.; Caselli, A.; Macchi, P.; Ferretti, F.; Gallo, E. Organometallics 2014, 33, 2210–2218
Intrieri, D.; Mariani, M.; Caselli, A.; Ragaini, F.; Gallo, E. Chem. Eur. J. 2012, 18, 10487–10490.
Manca, G.; Gallo, E.; Intrieri, D.; Mealli, C. ACS Catal. 2014, 823–832
Manca, G.; Mealli, C.; Carminati, D. M.; Intrieri, D.; Gallo, E. Eur. J. Inorg. Chem. 2015, 4885–4893.
For recent reports of our research group on catalyzed reactions in microand mesoreactors, see: (a) Porta, R.; Benaglia, M.; Coccia, F.; Rossi, S.; Puglisi, A. Symmetry 2015, 7, 1395–1409
Rossi, S.; Benaglia, M.; Puglisi, A.; De Filippo, C. C.; Maggini, M. J. Flow Chem. 2015, 5, 17–21.
Fantauzzi, S.; Gallo, E.; Caselli, A.; Piangiolino, C.; Ragaini, F.; Cenini, S. Eur. J. Org. Chem. 2007, 6053–6059
Zardi, P.; Pozzoli, A.; Ferretti, F.; Manca, G.; Mealli, C.; Gallo, E. Dalton Trans. 2015, 44, 10479–10489.
Rossi, S.; Puglisi, A.; Benaglia, M.; Carminati, D. M.; Intrieri, D.; Gallo, E. Catal. Sci. Technol. 2016, DOI: 10.1039/c6cy00207b.
For some recent examples of flow aziridine synthesis, see: (a) Hsueh, N.; Clarkson, G. J.; Shipman, M. Org. Lett. 2015, 17, 3632–3635
Scholz, S. O.; Farney, E. P.; Kim, S.; Bates, D. M.; Yoon, T. P. Angew. Chem., Int. Ed. 2016, 55, 2239–2242
Baumann, M.; Baxendale, I. R. Synlett 2016, 27, 159–163
Blackham, E. E.; Knowles, J. P.; Burgess, J.; Booker-Milburn, K. I. Chem. Sci. 2016, 7, 2302–2307.
The four solutions were analyzed by GC and, as expected, contained the desired product only, the azide, in increasing yield during time; the unreacted starting aniline remained in the aqueous phase and it was not detected.
It is known that phenylazide explodes when heated at ordinary pressure, and occasionally at lower pressures. See, for example, Org. Synth. 1942, 22, 96.
Fantauzzi, S.; Gallo, E.; Caselli, A.; Ragaini, F.; Macchi, P.; Casati, N.; Cenini, S. Organometallics 2005, 24, 4710–4713.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Rossi, S., Puglisi, A., Intrieri, D. et al. From Anilines to Aziridines: A Two-Step Synthesis under Continuous-Flow Conditions. J Flow Chem 6, 234–239 (2016). https://doi.org/10.1556/1846.2016.00027
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/1846.2016.00027