Abstract
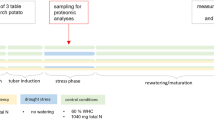
A high number of protease inhibitors (PI) have been identified in diverse plant species but information about their role in plant stress responses is still fragmentary. Transcript profiling of six published serine and cysteine protease inhibitor sequences in water-deprived plants from four winter wheat (Triticum aestivum) varieties with varying tolerance was performed in order to outline PIs predominantly accumulating under drought. Expression was analyzed by real time RT-qPCR. Considerable transcript accumulation of Bowman–Birk type PI WALI3 (BBPI) was detected in drought stressed leaves suggesting an important regulatory role of BBPI in adjustment of protein metabolism in leaves under dehydration. Serpin transcripts were less represented in water-deprived plants. Transient accumulation of cystatin transcripts revealed organ-specificity. Under drought cystatin and serpin expression in the leaves of the most drought tolerant variety “Katya” tended to preserve relatively stable levels close to the controls. This preliminary data will serve for future detailed study of regulation of proteolysis in winter wheat subjected to unfavorable environmental factors for development of molecular-based strategies for selection of tolerant varieties.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barrs, C., Weatherley, P.E. 1968. A re-examination of the relative turgidity technique forestimating water deficit in leaves. Aust. J. Biol. Sci. 15:413–428.
Christova, P.K., Christov, N.K., Imai, R. 2006. A cold inducible multidomain cystatin from winter wheat inhibits growth of the snow mold fungus, Microdochium nivale. Planta 223:1207–1218.
de Almeida, B.B., da Silva, W.G., Moreira, M.A., de Barros, E.G. 2012. In silico characterization and expression analysis of the multigene family encoding the Bowman–Birk protease inhibitor in soybean. Mol. Biol. Rep. 39:327–334.
Diop, N.N., Kidrič, M., Repellina, A., Gareila, M., d’Arcy-Lameta, A., Pham Thi, A.T., Zuily-Fodil, Y. 2004. A multicystatin is induced by drought-stress in cowpea (Vigna unguiculata (L.) Walp.) leaves. FEBS Letters 577:545–550.
Dramé, K.N., Passaquet, C., Repellin, A., Zuily-Fodil, Y. 2013. Cloning, characterization and differential expression of a Bowman-Birk inhibitor during progressive water deficit and subsequent recovery in peanut (Arachis hypogaea) leaves. J. Plant Physiol. 170:225–229.
Feldman, M.L., Andreu, A.B., Korgan, S., Lobato, M.C., Huarte, M., Walling, L.L., Daleo, G.R. 2014. PLPKI: A novel serine protease inhibitor as a potential biochemical marker involved in horizontal resistance to Phytophthora infestans. Plant Breeding 133:275–280.
Fluhr, R., Lampl, N., Roberts, T.H. 2012. Serpin protease inhibitors in plant biology. Physiologia Plantarum 145:95–102.
Jangpromma, N., Saito, A., Araki, T., Jaisil, P., Songsri, P., Daduang, S., Kawaguchi, Y., Dhiravisit, A., Thammasirirak, S. 2014. Molecular cloning and characterization in eukaryotic expression systems of a sugarcane cysteine protease inhibitor gene involved in drought tolerance. Turkish J. of Bot. 38:724–736.
Kidrič, M., Kos, J., Sabotič, J. 2014. Proteases and their endogenous inhibitors in the plant response to abiotic stress. Botanica Serbica 38:139–158.
Lampl, N., Alkan, N., Davydov, O., Fluhr, R. 2013. Set-point control of RD21 protease activity by AtSerpin1 controls cell death in Arabidopsis. The Plant J. 74:498–510.
Livak, K.J., Schmittgen, T.D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2–ΔΔCq method. Methods 25:402–408.
López-Otín, C., Bond, J.S. 2008. Proteases: multifunctional enzymes in life and disease. The J. of Biol. Chem. 283:30433–30437.
Ma, S., Gong, Q., Bohnert, H.J. 2006. Dissecting salt stress pathways. J. Exp. Bot. 57:1097–1107.
Martínez, M., Cambra, I., González-Melendi, P., Santamaría, M.E., Díaz, I. 2012. C1A cysteine-proteases and their inhibitors in plants. Physiologia Plantarum Spec. Iss. Plant Proteases 145:85–94.
Massonneau, A., Condamine, P., Wisniewski, J.P., Zivy, M., Rogowsky, P.M. 2005. Maize cystatins respond to developmental cues, cold stress and drought. Biochimica et Biophysica Acta 1729:186–199.
Mosolov, V.V., Valueva, T.A. 2005. Proteinase inhibitors and their function in plants: a review. Applied Biochem. and Microbiol. 41:261–282.
Pernas, M., Sanchez-Monge, R., Salcedo, G. 2000. Biotic and abiotic stress can induce cystatin expression in chestnut. FEBS Letters 467:206–210.
Quain, M.D., Makgopa, M.E., Marquez-Garcia, B., Comadira, G., Fernandez-Garcia, N., Olmos, E., Schnaubelt, D., Kunert, K.J., Foyer, C.H. 2014: Ectopic phytocystatin expression leads to enhanced drought stress tolerance in soybean (Glycine max) and Arabidopsis thaliana through effects on strigolactone pathways and can also result in improved seed traits. Plant Biotechnol. J. 12:903–913.
Rawlings, N.D., Waller, M., Barrett, A.J., Bateman, A. 2014. MEROPS: the database of proteolytic enzymes, their substrates and inhibitors. Nucleic Acids Res. 42:D503–D509.
Shan, L., Li, C., Chen, F., Zhao, S., Xia, G. 2008. A Bowman-Birk type protease inhibitor is involved in the tolerance to salt stress in wheat. Plant Cell Environ. 31:1128–1137.
Simova-Stoilova, L., Vaseva, I., Grigorova, B., Demirevska, K., Feller, U. 2010. Proteolytic activity and cysteine protease expression in wheat leaves under severe soil drought and recovery. Plant Physiol. and Biochem. 48:200–206.
Simova-Stoilova, L., Vassileva, V., Petrova, T., Tsenov, N., Demirevska, K., Feller, U. 2006. Proteolytic activity in wheat leaves during drought stress and recovery. General and Appl. Plant Physiol. Special Issue:91–100.
Snowden, K.C., Gardner, R.C. 1993. Five genes induced by aluminum in wheat (Triticum aestivum L.) roots. Plant Physiol. 103:855–861.
Wang, M., Qin, L., Xie, C., Li, W., Yuan, J., Kong, L., Yu, W., Xia, G., Liu, S. 2014. Induced and constitutive DNA methylation in a salinity-tolerant wheat introgression line. Plant Cell Physiol. 55:1354–1365.
Yarullina, L.G., Veselova, S.V., Ibragimov, R.I., Shpirnaya, I.A., Kasimova, R.I., Akhatova, A.R., Tsvetkov, V.O., Maksimov, I.V. 2014. Search for molecular markers of wheat resistance to fungal pathogens. Agric. Sci. 5:722–729.
Zhang, X., Liu, S., Takano, T. 2008. Two cysteine proteinase inhibitors from Arabidopsis thaliana, AtCYSa and AtCYSb, increasing the salt, drought, oxidation and cold tolerance. Plant Mol. Biol. 68:131–143.
Acknowledgements
This study was supported by a joint project “Regulation of proteolysis under water stress in di- and monocotyledonous plants” (REPRODIMO) - DNTC 01-12/13.12.2011. The excellent technical assistance of R. Nenkova, B. Juperlieva-Mateeva and A. Kostadinova is highly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Vaseva, I.I., Zehirov, G., Kirova, E. et al. Transcript Profiling of Serine- and Cysteine Protease Inhibitors in Triticum aestivum Varieties with Different Drought Tolerance. CEREAL RESEARCH COMMUNICATIONS 44, 79–88 (2016). https://doi.org/10.1556/0806.43.2015.032
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/0806.43.2015.032