Abstract
Introduction
We investigated the main factors affecting the efficacy of protoplast isolation, including leaf-obtaining period, cutting shapes of leaf material, enzyme concentration, enzymolysis time, and centrifugal speed.
Methods
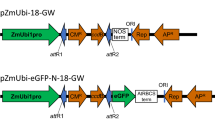
Protoplast isolation was optimal on the condition of 20 days of leaf materials, 2-mm filament of leaves, 1.6% RS and 0.8% R-10, 80 min of enzymolysis, and 700 rpm of centrifugation, resulting in the best yield (1.19 X 106 protoplasts/g FW) and vitality (80.34%) of mesophyll protoplasts. The transient expression vector pGFPl with green fluorescent protein was transfected into the obtained protoplasts from castor by polyethylene glycol-mediated method with a transformation efficiency of 12.37%.
Results
Moreover, the applicability of the system for studying the subcellular localization of Re FATA (an acyl-ACP thioesterase) was validated via the protoplast isolation and transient expression protocol in this study.
Discussion
Collectively, the efficient mesophyll protoplast isolation and protoplast transient expression system facilitate to analyze the function of specific gene in castor (Ricinus communis L).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anthony, P., Davey, M. R., Power, J. B. (1995) An improved protocol for the culture of cassava leaf protoplasts. Plant Cell Tiss. Organ. Cult. 42, 299–302.
Arroyo-Caro, J. M., Chileh, T., Kazachkov, M. (2013) The multigene family of lysophosphatidate acyltransferase (LPAT)-related enzymes in Ricinus communis: cloning and molecular characterization of two LP AT genes that are expressed in castor seeds. Plant Sci. 200, 29–40.
Auld, D. L., Rolfe, R. D., McKeon, T. A. (2001) Development of castor with reduced toxicity. J. New Seeds 3, 61–69.
Burris, K. P., Dlugosz, E. M., Collins, A. G. (2016) Development of a rapid, low-cost protoplast transfection system for switchgrass (Panicum virgatum L.). Plant Cell Rep. 35, 693–704.
Chen, S., Tao, L., Zeng, L. (2010) A highly efficient transient protoplast system for analyzing defence gene expression and protein-protein interactions in rice. Mol. Plant Pathol. 7, 417–427.
Davey, M. R., Anthony, P., Power, J. B. (2005) Plant protoplasts: status and biotechnological perspectives. Biotechnol. Adv. 23, 131–171.
De Caroli, M., Lenucci, M. S., Manualdi, F. (2015) Molecular dissection of Phaseolus vulgaris polygalacturonase-inhibiting protein 2 reveals the presence of hold/release domains affecting protein trafficking toward the cell wall. Front. Plant Sci. 6, 1–15.
Eastmond, P. J. (2004) Cloning and characterization of the acid lipase from castor beans. J. Biol. Chem. 279, 45540–5545.
Faraco, M., Di, S. G., Spelt, K. (2011) One protoplast is not the other! Plant Physiol. 156, 474–78.
Gressel, J. (2008) Transgenics are imperative for biofuel crops. Plant Sci. 174, 246–263.
Guo, J., Morrell-Falvey, J. L., Labbe, J. L. (2012) Highly efficient isolation of Populus mesophyll protoplasts and its application in transient expression assays. PLoS One 7, e44908.
He, X., Turner, C., Chen, G. Q. (2004) Cloning and characterization of a cDNA encoding diacylglycerol acyltransferase from castor bean. Lipids 39, 311–318.
Kim, H. U., Lee, K. R., Go, Y. S. (2011) Endoplasmic reticulum-located PDAT1-2 from castor bean enhances hydroxy fatty acid accumulation in transgenic plants. Plant Cell Physiol. 52, 983–993.
Kim, M. J., Baek, K., Park, C. M. (2009) Optimization of conditions for transient Agrobacterium-mediated gene expression assays in Arabidopsis. Plant Cell Rep. 28, 1159–1167.
Li, G. R., Huang, F. L., Wang, W. Y. (2012) Optimization of callus induct conditions of Ricinus communis anthers. J. Inner Mongolia Univ. Nat. 27, 670–673.
Liu, Y., Yang, Y. L., Yin, X. G. (2017) Expression of JcFATA gene in Jatropha curcas and its promoter cloning and analysis. J. Agric. Biotechnol. 25, 214–221.
Malathi, B., Ramesh, S., Rao, K. V. (2006) Agrobacterium-mediated genetic transformation and production of semilooper resistant transgenic castor (Ricinus communis L.). Euphytica 147, 441–449.
Miao, Y., Jiang, L. (2007) Transient expression of fluorescent fusion proteins in protoplasts of suspension cultured cells. Nature Protoc. 2, 2348–2353.
Murashige, T., Skoog, F. (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 15, 473–497.
Nanjareddy, K., Arthikala, M. K., Blanco, L. (2016) Protoplast isolation, transient transformation of leaf mesophyll protoplasts and improved Agrobacterium-mediated leaf disc infiltration of Phaseolus vulgaris: tools for rapid gene expression analysis. BMC Biotechnol. 16, 1–14.
Patat-Ochatt, E. M., Ochatt, S. J., Power, J. B. (1988) Plant regeneration from protoplasts of apple rootstocks and scion varieties (Malus X domestica Borkh.). J. Plant Physiol. 133, 460–465.
Raikar, S. V., Braun, R. H., Bryant, C. (2008) Efficient isolation, culture and regeneration of Lotus corniculatus protoplasts. Plant Biotechnol. Rep. 2, Ill-Ill.
Sailaja, M., Tarakeswari, M., Sujatha, M. (2008) Stable genetic transformation of castor (Ricinus communis L.) via particle gun-mediated gene transfer using embryo axes from mature seeds. Plant Cell Rep. 27, 1509–1519.
Salas, J. J., Ohlrogge, J. B. (2002) Characterization of substrate specificity of plant FatA and FatB acyl-ACP thioesterases. Arch. Biochem. Biophys. 403, 25–34.
Shao, Z. M., Chen, Y. S., Huang, F. L. (2012) Effects of low temperature pretreatment and light conditions on callus induction of castor anthers. J. Inner Mongolia Univ. Nat. 27, 189–193.
Sheen, J. (2001) Signal transduction in maize and Arabidopsis mesophyll protoplasts. Plant Physiol. 127, 1466–1475.
Siritunga, D., Ariasgarzon, D., White, W. (2007) Over-expression of hydroxynitrile lyase in transgenic cassava roots accelerates cyanogenesis and food detoxification. Plant Biotechnol. J. 2, 37–3.
Sujatha, M., Lakshminarayana, M., Tarakeswari, M. (2009) Expression of the crylEC gene in castor (Ricinus communis L.) confers field resistance to tobacco caterpillar (Spodoptera litura Fabr) and castor semilooper (Achoea Janata L.). Plant Cell Rep. 28, 935–946.
Sujatha, M., Sailaja, M. (2005) Stable genetic transformation of castor (Ricinus communis L.) via Agrobacterium tumefaciens-mediated gene transfer using embryo axes from mature seeds. Plant Cell Rep. 23, 803–810.
Tang, R., Peng, X. Q., Xie, X. M. (2015) Protoplast isolation from elephant grass (Pennisetum purpureum ’Huanan’) for transient gene expression. Acta. Agrectir. Sinica. 23, 571–579.
Wang, H., Wang, W., Zhan, J. (2015) An efficient PEG-mediated transient gene expression system in grape protoplasts and its application in subcellular localization studies of flavonoids biosynthesis enzymes. Sci. Hortic. 191, 82–89.
Wang, Q., Huang, W. D., Jiang, Q. (2013) Lower levels of expression of FATA 2 gene promote longer siliques with modified seed oil content in Arabidopsis thaliana. Plant Mol. Biol. Rep. 31, 1368–1375.
Wu, J. Z., Liu, Q., Geng, X. S. (2017) Highly efficient mesophyll protoplast isolation and PEG-mediated transient gene expression for rapid and large-scale gene characterization in cassava (Manihot esculenta Crantz). BMC Biotechnol. 17, 1–8.
Yao, L., Liao, X., Gan, Z. (2016) Protoplast isolation and development of a transient expression system for sweet cherry (Prunus avium L.). Sci. Hortic. 209, 14–21.
Yoo, S. D., Cho, Y. H., Sheen, J. (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc. 2, 1565–1572.
Zhang, Y., Su, J., Duan, S. (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7, 1–14.
Zhu, L., Wang, B., Zhou, J. (2005) Protoplast isolation of callus in Echinacea augustifolia. Colloid. Surface. B 44, 1–5.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Y., Xue, Y., Tang, J. et al. Efficient mesophyll protoplast isolation and development of a transient expression system for castor-oil plant (Ricinus communis L.). BIOLOGIA FUTURA 70, 8–15 (2019). https://doi.org/10.1556/019.70.2019.02
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/019.70.2019.02