Abstract
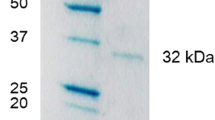
An alginate lyase with high specifc enzyme activity was purifed from Pseudomonas stutzeri MSEA04, isolated from marine brown algae. The alginate lyase was purifed by precipitation with ammonium sulphate, acetone and ethanol individually. 70% ethanol fraction showed maximum specifc activity (133.3 U/mg). This fraction was re-purifed by anion exchange chromatography DEAE- Cellulose A-52. The loaded protein was separated into 3 peaks. The second protein peak was the major one which contained 48.2% of the total protein recovered and 79.4% of the total recovered activity. The collected fractions of this peak were subjected to further purifcation by re-chromatography on Sephadex G-100. Alginate lyase activity was fractionated in the Sephadex column into one major peak, and the specifc activity of this fraction reached 116 U/mg. The optimal substrate concentration, pH and temperature for alginate lyase activity were 8 mg/ml, pH 7.5 and 37 °C, respectively. While, Km and Vmax values were 1.07 mg alginate/ ml and 128.2 U/mg protein, respectively. The enzyme was partially stable below 50 °C, and the activity of the enzyme was strongly enhanced by K+, and strongly inhibited by Ba+2, Cd+2, Fe+2 and Zn+2. The purifed enzyme yielded a single band on SDS-PAGE with molecular weight (40.0 kDa).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Badur, A. H., Jagtap, S. S., Yalamanchili, G., Lee, J. K., Zhao, H., Rao, C. V. (2015) Alginate Lyases from Alginate-Degrading Vibrio splendidus 12B01 are endolytic. Appl. Environ. Microbiol. 81, 1865–1873.
Beltagy, E. A., Youssef, A. S., El-Shenawy, M. A., El-Assar, S. A. (2012) Purifcation of kappa (k)-carrageenase from locally isolated Cellulosimicrobium cellulans. Afri. J. Biotechnol. 11, 11438–11446.
Cao, L., Xie, L., Xue, X., Tan, H., Liu, Y., Zhou, S. (2007) Purifcation and characterization of alginate lyase from Streptomyces species strain A5 isolated from banana rhizosphere. J. Agric. Food Chem. 55, 5113–5117.
El-Katatny, M. H., Hetta, A. M., Shaban, G. M., El-Komy, H. M. (2003) Improvement of cell wall degrading enzymes production by alginate encapsulated Trichoderma spp. Food Technol. Biotechnol. 41, 219–225.
Fu, X. T., Lin, H., Kim, S. M. (2007) Purifcation and characterization of a Na+/K+ dependent alginate lyase from turban shell gut Vibrio sp. YKW-34. Enz. Microbiol. Technol. 41, 828–834.
Gimmestad, M., Sletta, H., Ertesvåg, H., Bakkevig, K., Jain, S., Suh, S.-J., Skjåk-Bræk, G., Ellingsen, T. E., Ohman, D. E., Valla, S. (2003) The Pseudomonas fuorescens AlgG protein, but not its mannuronan C-5-epimerase activity, is needed for alginate polymer formation. J. Bacteriol. 185, 3515–3523.
Kim, H. S., Lee, C. G., Lee, E. Y. (2011) Alginate Lyase: Structure, Property, and Application. Biotechnol. Bioprocess Eng. 16, 843–851.
Laemmli, U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685.
Li, J. W., Dong, S., Song, J., Li, C. B., Chen, X. L., Xie, B. B., Zhang, Y. Z. (2011) Purifcation and Characterization of a Bifunctional Alginate Lyase from Pseudoalteromonas sp SM0524. Mar. Drugs 9, 109–123.
Lipman, D. J. (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402.
Lineweaver, H., Burke, D. (1934) The determination of enzyme dissociation constants. J. Am. Chem. Soc. 56, 658–666.
Lowry, O. H., Rosbrough, N. J., Farr, A. L., Randall, R. J. (1951) Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265–275.
Michaud, P., Da Costa, A., Courtois, B., Courtois, J. (2003) Polysaccharide lyases: Recent developments as biotechnological tools. Crit. Rev. Biotechnol. 23, 233–266.
Miyake, O., Hashimoto, W., Murata, K. (2003) An exotype alginate lyase in Sphingomonas sp. A1: overexpression in Escherichia coli, purifcation, and characterization of alginate lyase IV (A1–IV). Protein Expr. Purif. 29, 33–41.
Nakagawa, A., Ozaki, T., Chubachi, K., Hosoyama, T., Okubo, T., Iyobe, S., Suzuki, T. (1998) An effective method for isolating alginate lyase-producing Bacillus sp. ATB-1015 strain and purifcation and characterization of the lyase. J. Appl. Microbiol. 84, 328–335.
Osawa, T., Matsubara, Y., Muramatsu, T., Kimura, M., Kakuta, Y. (2005) Crystal structure of the alginate (poly alpha-L-guluronate) lyase from Corynebacterium sp. at 1.2 A resolution. J. Mol. Biol. 345, 1111–1118.
Park, D., Jagtap, S., Nair, S. (2014) Structure of a PL17 Family Alginate Lyase Demonstrates Functional Similarities among Exotype Depolymerases. J. Biol. Chem. 289, 8645–8655.
Park, H. H., Kam, N., Lee, E. Y., Kim, H. S. (2012) Cloning and characterization of a novel oligoalginate lyase from a newly isolated bacterium Sphingomonas sp. MJ-3. Mar. Biotechnol. 14, 189–202.
Peterson, E. A., Sober, H. A. (1962) Column chromatography of protein: Substituted cellulases. In: S. Colowich, N. Kapllan (eds) Methods in Enzymology. Vol. 5 New York. 3–27.
Seiderer, L. J., Newell, R. C., Cook, P. A. (1982) Quantitative signifcance of style enzymes from two marine mussels (Choromytilus meridionalis Krauss and Perna perna Linnaeus) in relation to diet. Mar. Biol. Lett. 3, 257–271.
Singh, R. P., Gupta, V., Kumari, P., Kumar, M., Reddy, C. R. K., Prasad, K., Jha, B. (2011) Purifcation and partial characterization of an extracellular alginate lyase from Aspergillus oryzae isolated from brown seaweed. J. Appl. Phycol. 23, 755–762.
Wang, Y., Guo, E. W., Yu, W. G., Han, F. (2013) Purifcation and characterization of a new alginate lyase from a marine bacterium Vibrio sp. Biotechnol. Lett. 35, 703–708.
Wang, Y. H., Yu, G. L., Wang, X. M., Lv, Z. H., Zhao, X., Wu, Z. H., Ji, W. S. (2006) Purifcation and Characterization of Alginate Lyase from Marine Vibrio sp. YWA. Acta Biochim. Biophys. Sinica 38, 633–638.
Wenjun, H., Jingyan, G., Yuanyuan, C., Huihui, L., Yuezhong, L., Fuchuan, L. (2015) A Novel Alginate Lyase (Aly5) from a Polysaccharide-Degrading Marine Bacterium Flammeovirga sp. MY04: Effects of Module Truncation to the Biochemical Characteristics, Alginate-Degradation Patterns, and Oligosaccharide-Yielding Properties. Appl. Environ. Microb.; doi:10.1128/AEM.03022-15.
Yamasaki, M., Moriwaki, S., Miyake, O., Hashimoto, W., Murata, K., Mikami, B. (2004) Structure and Function of a Hypothetical Pseudomonas aeruginosa Protein PA1167 Classifed into Family PL-7 A NOVEL ALGINATE LYASE WITH A β-SANDWICH FOLD. J. Biol. Chem. 279, 31863–31872.
Zhu, Y., Wu, L., Chen, Y., Ni, H., Xiao, A., Cai, H. (2015) Characterization of an extracellular biofunctional alginate lyase from marine Microbulbifer sp. ALW1 and antioxidant activity of enzymatic hydrolysates. Microbiol. Res. 182, 49–58.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Beltagy, E.A., El-Borai, A., Lewiz, M. et al. Purification and Characterization of Alginate Lyase from Locally Isolated Marine Pseudomonas Stutzeri MSEA04. BIOLOGIA FUTURA 67, 305–317 (2016). https://doi.org/10.1556/018.67.2016.3.8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.67.2016.3.8