Abstract
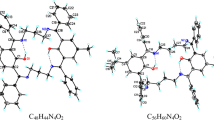
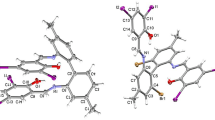
Two Schiff base derivatives, 4-(2-amino-3-pyridyliminomethyl)phenol (I) and 3-(2-amino-3-pyridyliminomethyl)nitrobenzene (II), were synthesised and characterised by spectroscopy. The structure of I was determined by single crystal X-ray diffraction studies. The asymmetric Schiff base derived from 2,3-diaminopyridine selectively recognise transition and heavy metal cations, and some anion. Ligands I and II form stable complexes with Cu2+, Zn2+, Pb2+, Al3+ whereas ligand I also binds F~ ions. The stoichiometry for the host: cation is 1: 1 and 2: 1. The addition of F~ ion in CH3CN to ligand I causes a colour change of the solution from colourless to yellow. The binding behaviour of ligand I towards several ions was investigated using density functional theory calculations.
Similar content being viewed by others
References
Abdel-Rahman, L. H., El-Khatib, R. M., Nassr, L. A. E., Abu-Dief, A. M., & El-Din Lashin, F. (2013). Design, characterization, teratogenicity testing, antibacterial, antifungal and DNA interaction of few high spin Fe(II) Schiff base. Analytica Chimica Acta, 746, 98–106. DOI: 10.1016/j.aca.2012.08.024.
Amin, R. M., Abdel-Kader, N. S., & El-Ansary, A. L. (2012). Microplate assay for screening the antibacterial activity of Schiff bases derived from substituted benzopyran-4-one. Spectrochimica Acta Part A, 95, 517–525. DOI: 10.1016/j.saa.2012.04.042.
Azadbakht, R., Almasi, T., Keypour, H., & Rezaeivala, H. (2013). A new asymmetric Schiff base system as fluorescent chemosensor for Al3+ ion. Inorganic Chemistry Communications, 33, 63–67. DOI: 10.1016/j.inoche.2013.03.014.
Aziz, A. A. A. (2013). A novel highly sensitive and selective optical sensor based on a symmetric tetradentate Schiff-base embedded in PVC polymeric film for determination of Zn2+ ion in real samples. Journal of Luminescence, 143, 663–669. DOI: 10.1016/j.jlumin.2013.06.020.
Carreno, A., Gacitua, M., Schott, E., Zarate, X., Manriquez, J. M., Preite, M., Ladeira S., Castel, A., Pizarro, N., Vega, A., Chavez, I., & Arratia-Perez, R. (2015). Experimental and theoretical studies of the ancillary ligand (E)-2-((3-amino-pyridin-4-ylimino)-methyl)-4,6-di-tert-butylphenol in the rhenium(I) core. New Journal of Chemistry, 39, 57255734. DOI: 10.1039/c5nj00772k.
Cimerman, Z., Galesic, N., & Bosner, B. (1992). Structure and spectroscopic characteristics of Schiff bases of salicylaldehyde with 2,3-diaminopyridine. Journal of Molecular Structure, 274, 131–144. DOI: 10.1016/0022-2860(92)80152-8.
Cimerman, Z., Galic, N., & Bosner, B. (1997). The Schiff bases of salicylaldehyde and aminopyridines as highly sensitive analytical reagents. Analytica Chimica Acta, 343, 145–153. DOI: 10.1016/s0003-2670(96)00587-9.
Dai, C. H., & Mao, F. L. (2013). Structure of a new Schiff base cobalt(III) complex with antibacterial activity. Journal of Structural Chemistry, 54, 624–629. DOI: 10.1134/s0022476613030244.
Devaraj, S., Tsui, Y. K., Chiang, C. Y., & Yen, Y. P. (2012). A new dual functional sensor: Highly selective colorimetric chemosensor for Fe3+ and fluorescent sensor for Mg2+. Spectrochimica Acta Part A, 96, 594–599. DOI: 10.1016/j.saa.2012.07.032.
Dubey, P. K., & Ratnam, C. V. (1977). Formation of heterocyclic rings containing nitrogen: Part XXVI–Condensation of pyridine 2,3-diamine with aromatic aldehydes. Proceedings of the Indian Academy of Sciences–Section A, 85, 204–209. DOI: 10.1007/bf03049482.
Erdemir, S., Kocyigit, O., Alici, O., & Malkondu, S. (2013). ‘Naked-eye’ detection of F− ions by two novel colorimetric receptors. Tetrahedron Letters, 54, 613–617. DOI: 10.1016/j.tetlet.2012.11.138.
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Montgomery, J. A., Jr., Vrenen, T., Kudin, K. N., Burant, J. C., Illa, J. M., Iyengar, S. S., Tomasi, J., Barone, V., Mennuci, B., Cossi, M., Scalmani, G. Rega, N., Petersson, G. A., Nakatsuji, H., Hada, M., Ehara, M., Toyota, K., Fukuda, K. R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Klene, M., Li, X., Knox, J. E., Hratchian, H. P., Cross, J. B., Bakken, V., Adamo, V. C., Jaramillo, J., Gomperts, R., Stratmann, R. E., Yazyev, O., Ausin, A. J., Cammi, R., Pomelli, C., Ochterski, J., Ayala, P. Y., Morokuma, K., Voth, G. A., Salavador, P., Dannenberg, J. J., Zakrzewski, V. G., Dopprich, S., Daniels, A.D., Strain. M. C., Farkas, O., Malick, D. K., Rabuck, A. D., Raghavashari, K., Foresman, J. B., Orlitz, J. V., Cui, Q., Baboul, A., Cliffors, S., Cioslowski, J., Stefanov, B. B., Liu, G., Liashenko, A., Piskorz, P., Komaromo, I., Martin, R. L., Fox, D. J., Keith, T., Al-Laham, M. A., Peng, C. Y., Nanyakkara, A., Challacombe, M., Gill, P. M. W., Johnson, B., Chen, W., Wong, J. L. Gonzalez, C., & Pople, J. (2004). Gaussian 03, Revision 03 [computer software]. Wallingford, CT, USA: Gaussian Inc.
Grivani, G., & Akherati, A. (2013). Polymer-supported bis (2-hydroxyanyl) acetylacetonato molybdenyl Schiff base catalyst as effective, selective and highly reusable catalyst in epoxidation of alkenes. Inorganic Chemistry Communications, 28, 90–93. DOI: 10.1016/j.inoche.2012.11.015.
Gupta, V. K., Singh, A. K., Ganjali, M. R., Norouzi, P., Faridbod, F., & Mergu, N. (2013). Comparative study of colorimetric sensors based on newly synthesized Schiff bases. Sensors and Actuators B, 182, 642–651. DOI: 10.1016/j.snb.2013.03. 062.
Heo, Y., Kang, Y. Y., Palani, T., Lee, J., & Lee, S. (2012). Synthesis, characterization of palladium hydroxysalen complex and its application in the coupling reaction of arylboronic acids: Mizoroki-Heck type reaction and decarboxylative couplings. Inorganic Chemistry Communications, 23, 1–5. DOI: 10.1016/j.inoche.2012.05.013.
Huang, C. Y., Wan, C. F., Chir, J. L., & Wu, A. T. (2013). A Schiff-based colorimetric fluorescent sensor with potential for detection of fluoride ions. Journal of Fluorescence, 23, 1107–1111. DOI: 10.1007/s10895–013–1257-z.
Jarvo, E. R., Lawrence, B. M., & Jacobsen, E. N. (2005). Highly enantio- and regioselective quinone Diels-Alder reactions cat-alyzed by a tridentate [(Schiff base)CrIII] complex. Angewandte Chemie International Edition, 44, 6043–6046. DOI: 10.1002/anie.200502176.
Jeong, T., Lee, H. K., Jeong, D. C., & Jeon, S. (2005). A lead(II)-selective PVC membrane based on a Schiff base complex of N,N′-bis(salicylidene)-2,6-pyridinediamine. Talanta, 65, 543–548. DOI: 10.1016/j.talanta.2004.07.016.
Jeewoth, T., Bhowon, M. G., & Wah, H. L. K. (1999). Synthesis, characterization and antibacterial properties of Schiff bases and Schiff base metal complexes derived from 2,3-diamino-pyridine. Transition Metal Chemistry, 24, 445–448. DOI: 10.1023/a: 1006917704209.
Ji, C., Day, S. E., & Silvers, W. C. (2008). Catalytic reduction of 1- and 2-bromooctanes by a dinickel(I) Schiff base complex containing two salen units electrogenerated at carbon cathodes in dimethylformamide. Journal of Electroanalytical Chemistry, 622, 15–21. DOI: 10.1016/j.jelechem.2008.04.023.
Jimenez-Sanchez, A., Farfan, N., & Santillan, R. (2013). A reversible fluorescent-colorimetric Schiff base sensor for Hg2+ ion. Tetrahedron Letters, 54, 5279–5283. DOI: 10.1016/j.tetlet.2013.07.072.
Kleij, A. W., Tooke, D. M., Spek, A. L., & Reek, J. N. H. (2005). A convenient synthetic route for the preparation of nonsymmetric metallo-salphen complexes. European Journal of Inorganic Chemistry, 22, 4626–4632. DOI: 10.1002/ejic.200500628.
Kumar, K. S., Ganguly, S., Veerasamy, R., & De Clercq, E. (2010). Synthesis, antiviral activity and cytotoxicity evaluation of Schiff bases of some 2-phenyl quinazoline-4(3)#-ones. European Journal of Medicinal Chemistry, 45, 5474–5479. DOI: 10.1016/j.ejmech.2010.07.058.
Kumar, M. S., Kumar, S. L. A., & Sreekanth, A. (2013). An efficient triazole-based fluorescent “turn-on” receptor for nakedeye recognition of F− and AcO−: UV-visible, fluorescence and 1H NMR studies. Materials Science and Engineering: C, 33, 3346–3352. DOI: 10.1016/j.msec.2013.04.018.
Lin, C. Y., Huang, K. F., & Yen, Y. P. (2013). A new selective colorimetric and fluorescent chemodosimeter for HSO based on hydrolysis of Schiff base. Spectrochimica Acta Part A, 115, 552–558. DOI: 10.1016/j.saa.2013.06.083.
Liu, G., & Shao, J. (2013). Ratiometric fluorescence and colorimetric sensing of anion utilizing simple Schiff base derivatives. Journal of Inclusion Phenomena and Macrocyclic Chemistry, 76, 99–105. DOI: 10.1007/s10847-012-0177-x.
Ourari, A., Khelafi, M., Aggoun, D., Jutand, A., & Amatore, C. (2012). Electrocatalytic oxidation of organic substrates with molecular oxygen using tetradentate ruthenium(III)-Schiff base complexes as catalysts. Electrochimica Acta, 75, 366–370. DOI: 10.1016/j.electacta.2012.05.021.
Qiao, X., Ma, Z. Y., Xie, C. Z., Xue, F., Zhang, Y. W., Xu, J. Y., Qiang, Z. Y., Lou, J. S., Chen, G. J., & Yan, S. P. (2011). Study on potential antitumor mechanism of a novel Schiff base copper(II) complex: Synthesis, crystal structure, DNA binding, cytotoxicity and apoptosis induction activity. Journal of Inorganic Biochemistry, 105, 728–737. DOI: 10.1016/j.jinorgbio.2011.01.004.
Reena, V., Suganya, S., & Velmathi, S. (2013). Synthesis and anion binding studies of azo-Schiff bases: Selective colorimetric fluoride and acetate ion sensors. Journal of Fluorine Chemistry, 153, 89–95. DOI: 10.1016/j.jfluchem.2013.05.010.
Sahin, Z. M., Doganci, E., Yildiz, S. Z., Tuna, M., Yilmaz, F., Yerli, Y., & Gürür, M. (2010). Synthesis and characterization of two-armed poly(e-caprolactone) polymers initiated by Schiff’s base complexes of copper(II) and nickel(II). Synthetic Metals, 160, 1973–1980. DOI: 10.1016/j.synthmet.2010.07.018.
Schiff, H. (1866). Eine neue Reihe organischer Diamine. Annalen der Chemie und Pharmacie, 140, 92–137. DOI: 10.1002/jlac.18661400106. (in German)
Schilf, W., Kamienski, B., Rozwadowski, Z., Ambroziak, K., Bieg, B., & Dziembowska, T. (2004). Solid state 15N and 13C NMR study of dioxomolybdenum(VI) complexes of Schiff bases derived from trans-1,2-cyclohexanediamine. Journal of Molecular Structure, 700, 61–65. DOI: 10.1016/j.molstruc.2003.11.055.
Sen, S., Mukherjee, M., Chakrabarty, K., Hauli, I., Mukhopadhyay, S. K., & Chattopadhyay, P. (2013). Cell permeable fluorescent receptor for detection of H2PO4− in aqueous solvent. Organic & Biomolecular Chemistry, 11, 1537–1544. DOI: 10.1039/c2ob27201f.
Sheldrick, G. M. (2008). A short history of SHELX. Acta Crystallographica Section A, 64, 112–122. DOI: 10.1107/s0108767307043930.
Udhayakumari, D., Saravanamoorthy, S., & Velmathi, S. (2012). Colorimetric and fluorescent sensing of transition metal ions in aqueous medium by salicylaldimine based chemosensor. Materials Science and Engineering: C, 32, 1878–1882. DOI: 10.1016/j.msec.2012.05.005.
Waldeck, D. H. (1991). Photoisomerization dynamics of stilbenes. Chemical Reviews, 91, 415–436. DOI: 10.1021/cr00003a007.
Yang, Y. X., Xue, H. M., Chen, L. C., Sheng, R. L., Li, X. Q., & Li, K. (2013). Colorimetric and highly selective fluorescence “turn-on” detection of Cr3+ by using a simple Schiff base sensor. Chinese Journal of Chemistry, 31, 377–380. DOI: 10.1002/cjoc.201200852.
Yao, L. H., Wang, L., Zhang, J. F., Tang, N., & Wu, J. C.(2012). Ring opening polymerization of L-lactide by an electron-rich Schiff base zinc complex: An activity and kinetic study. Journal of Molecular Catalysis A, 352, 57–62. DOI: 10.1016/j.molcata.2011.10.012.
Yildiz, M., Unver, H., Erdener, D., Kiraz, A., & Iskeleli, N. O. (2009). Synthesis, spectroscopic studies and crystal structure of (E)-2-(2,4-dihydroxybenzylidene)thiosemicarbazone and (E)-2-[(1#-indol-3-yl)methylene]thiosemicarbazone. Journal of Molecular Structure, 919, 227–234. DOI: 10.1016/j.molstruc.2008.09.008.
Yuan, X. J., Wang, R. Y., Mao, C. B., Wu, L., Chu, C. Q., Yao, R., Gao, Z. Y., Wu, B. L., & Zhang, H. Y. (2012). New Pb(II)-selective membrane electrode based on a new Schiff base complex. Inorganic Chemistry Communications, 15, 29–32. DOI: 10.1016/j.inoche.2011.09.031.
Zhang, L., Ni, X. F., Sun, W. L., & Shen, Z. Q. (2008). Polymerization of isoprene catalyzed by neodymium heterocyclic Schiff base complex. Chinese Chemical Letters, 19, 734–738. DOI: 10.1016/j.cclet.2008.03.007.
Zhou, G. P., Hui, Y. H., Wan, N. N., Liu, Q. J., Xie, Z. F., & Wang, J. D. (2012a). Mn(OAc)2/Schiff base as a new efficient catalyst system for the Henry reaction of nitroalkanes with aldehydes. Chinese Chemical Letters, 23, 690–694. DOI: 10.1016/j.cclet.2012.04.018.
Zhou, Y. M., Zhou, H., Zhang, J. L., Zhang, L., & Niu, J. Y. (2012b). Fe3+-selective fluorescent probe based on aminoantipyrine in aqueous solution. Spectrochimica Acta Part A, 98, 14–17. DOI: 10.1016/j.saa.2012.08.025.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pazik, A., Kamińska, B., Skwierawska, A. et al. Synthesis, structural and spectroscopic properties of asymmetric Schiff bases derived from 2,3-diaminopyridine. Chem. Pap. 70, 1204–1217 (2016). https://doi.org/10.1515/chempap-2016-0058
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/chempap-2016-0058