Abstract
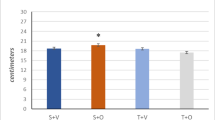
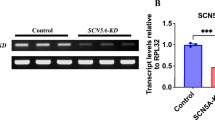
OBJECTIVE: In skeletal muscle, glucocorticoids induce catabolism and proteolysis which is accomplished via the ubiquitin (Ub) proteolytic pathway. Cardiac muscle is a striated muscle which, compared to skeletal muscle, more abundantly expresses components of the Ub pathway, thus suggesting an important role for this pathway in heart physiology. The aim of our study was to explore the role of the Ub pathway in heart muscle physiology. DESIGN: We treated rats for three days with a pharmacologic dose of dexamethasone (DEXA) 0.5mg/100g body weight (BW). An attempt was also made to counteract the DEXA effect by co-treatment with insulinlike growth factor-1 (IGF-1; 0.35mg/100g BW). RESULTS: DEXA treatment caused a 7.8% decrease in heart weight compared to control (p<0.05) and also increased heart tissue levels of the ubiquitin-conjugating enzyme E2 and the 20S proteasome protein. Myofibrillar proteins degraded by the ubiquitin pathway (α-actin, myoglobin, and troponin 1) were all decreased by DEXA, while ubiquitinated forms of α-actin were increased. Co-treatment with IGF-1 completely prevented DEXA-induced decrease in heart weight, an effect which was accompanied by decreased heart tissue levels of several ubiquitinated proteins including α-actin, the 20S proteasome protein, E2-14kDa mRNA, and C-3 proteasome subunit mRNA, while the levels of non-ubiquinated α-actin, myoglobin, and troponin 1 were all partially restored. CONCLUSION: These results demonstrate that DEXA activates the ubiquitin proteolytic pathway in the heart and that IGF-1 efficiently counteracts this effect. Our findings reveal a possible mechanism for the anti-proteolytic actions of IGF-1 and its cardioprotective role involving the Ub pathway.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Goldberg AL, 1969 Protein turnover in skeletal muscle. II. Effects of denervation and cortisone on protein catabolism in skeletal muscle. J Biol Chem 244: 3223–3229.
Dardevet D, Sornet C, Taillandier D, Savary I, Attaix D, Grizard J, 1995 Sensitivity and protein turnover response to glucocorticoids are different in skeletal muscle from adult and old rats. Lack of regulation of the ubiquitin-proteasome proteolytic pathway in aging. J Clin Invest 96: 2113–2119.
Auclair D, Garrel DR, Chaouki Zerouala A, Ferland LH, 1997 Activation of the ubiquitin pathway in rat skeletal muscle by catabolic doses of glucocorticoids. Am J Physiol 272: C1007–1016.
Menconi M, Fareed M, O’Neal P, Poylin V, Wei W, Hasselgren PO, 2007 Role of glucocorticoids in the molecular regulation of muscle wasting. Crit Care Med 35:.
Tisdale MJ, 2007 Is there a common mechanism linking muscle wasting in various disease types? Curr Opin Support Palliat Care 1: 287–292.
Eldridge AG, O’Brien T, Therapeutic strategies within the ubiquitin proteasome system. Cell Death Differ 17: 4–13.
Mitch WE, Goldberg AL 1996 Mechanisms of muscle wasting. The role of the ubiquitin-proteasome pathway. N Engl J Med 335: 1897–1905
Liu Z, Miers WR, Wei L, Barrett EJ, 2000 The ubiquitin-proteasome proteolytic pathway in heart vs. skeletal muscle: effects of acute diabetes. Biochem Biophys Res Commun 276: 1255–1260.
Bulteau AL, Szweda LI, Friguet B, 2002 Age-dependent declines in proteasome activity in the heart. Arch Biochem Biophys 397: 298–304.
Fielitz J, van Rooij E, Spencer JA, et al, 2007 Loss of muscle-specific RING-finger 3 predisposes the heart to cardiac rupture after myocardial infarction. Proc Natl Acad Sci U S A 104: 4377–4382.
Willis MS, Schisler JC, Li L, et al, 2009 Cardiac muscle ring finger-1 increases susceptibility to heart failure in vivo. Circ Res 105: 80–88.
Fang CX, Dong F, Thomas DP, Ma H, He L, Ren J, 2008 Hypertrophic cardiomyopathy in high-fat diet-induced obesity: role of suppression of forkhead transcription factor and atrophy gene transcription. Am J Physiol Heart Circ Physiol 295: H1206–H1215.
Weekes J, Morrison K, Mullen A, Wait R, Barton P, Dunn MJ, 2003 Hyperubiquitination of proteins in dilated cardiomyopathy. Proteomics 3: 208–216.
Okada K, Wangpoengtrakul C, Osawa T, Toyokuni S, Tanaka K, Uchida K, 1999 4-Hydroxy-2-nonenalmediated impairment of intracellular proteolysis during oxidative stress. Identification of proteasomes as target molecules. J Biol Chem 274: 23787–23793.
Bulteau AL, Lundberg KC, Humphries KM, et al, 2001 Oxidative modification and inactivation of the protea-some during coronary occlusion/reperfusion. J Biol Chem 276: 30057–30063.
Sicard RE, Werner JC, 1992 Dexamethasone induces a transient relative cardiomegaly in neonatal rats. Pediatr Res 31: 359–363.
Griffin EE, Wildenthal K, 1978 Regulation of cardiac protein balance by hydrocortisone: interaction with insulin. Am J Physiolg 234: E306–313.
Rannels SR, Rannels DE, Pegg AE, Jefferson LS, 1978 Glucocorticoid effects on peptide-chain initiation in skeletal muscle and heart. Am J Physiol 235: E134–139
Kelly FJ, Goldspink DF, 1982 The differing responses of four muscle types to dexamethasone treatment in the rat. Biochem J 208: 147–151.
Clark AF, DeMartino GN, Wildenthal K, 1986 Effects of glucocorticoid treatment on cardiac protein synthesis and degradation. Am J Physiol 250: C821–827.
Frost RA, Nystrom GJ, Jefferson LS, Lang CH, 2007 Hormone, cytokine, and nutritional regulation of sepsis-induced increases in atrogin-1 and MuRF1 in skeletal muscle. Am J Physiol Endocrinol Metab 292: E501–512.
Lang CH, Huber D, Frost RA 2007 Burn-induced increase in atrogin-1 and MuRF-1 in skeletal muscle is glucocorticoid independent but downregulated by IGF-I. Am J Physiol Regul Integr Comp Physiol 292: R328–336.
Clemmons DR, 2009 Role of IGF-I in skeletal muscle mass maintenance. Trends Endocrinol Metab 20: 349–356.
Schakman O, Gilson H, Thissen JP 2008 Mechanisms of glucocorticoid-induced myopathy. J Endocrinol 197: 1–10.
Fang CH, Li BG, Wang JJ, Fischer JE, Hasselgren PO, 1997 Insulin-like growth factor 1 stimulates protein synthesis and inhibits protein breakdown in muscle from burned rats. JPEN J Parenter Enteral Nutr 21: 245–251.
Chrysis D, Underwood LE, 1999 Regulation of components of the ubiquitin system by insulin-like growth factor I and growth hormone in skeletal muscle of rats made catabolic with dexamethasone. Endocrinology 140: 5635–5641.
Chrysis D, Zhang J, Underwood LE 2002 Divergent regulation of proteasomes by insulin-like growth factor I and growth hormone in skeletal muscle of rats made catabolic with dexamethasone. Growth Horm IGF Res 12: 434–441.
Schakman O, Kalista S, Bertrand L, et al, 2008 Role of Akt/GSK-3beta/beta-catenin transduction pathway in the muscle anti-atrophy action of insulin-like growth factor-I in glucocorticoid-treated rats. Endocrinology 149: 3900–3908.
Cittadini A, Ishiguro Y, Stromer H, et al, 1998 Insulinlike growth factor-1 but not growth hormone augments mammalian myocardial contractility by sensitizing the myofilament to Ca2+ through a wortmannin-sensitive pathway: studies in rat and ferret isolated muscles. Circ Res 83: 50–59.
Reiss K, Cheng W, Ferber A, et al, 1996 Overexpression of insulin-like growth factor-1 in the heart is coupled with myocyte proliferation in transgenic mice. Proc Natl Acad Sci U S A 93: 8630–8635.
Cittadini A, Stromer H, Katz SE, et al, 1996 Differential cardiac effects of growth hormone and insulin-like growth factor-1 in the rat. A combined in vivo and in vitro evaluation. Circulation 93: 800–809.
Young LH, Renfu Y, Hu X, et al, 1999 Insulin-like growth factor I stimulates cardiac myosin heavy chain and actin synthesis in the awake rat. Am J Physiol 276: E143–150.
Komamura K, Shirotani-Ikejima H, Tatsumi R, et al, 2003 Differential gene expression in the rat skeletal and heart muscle in glucocorticoid-induced myopathy: analysis by microarray. Cardiovasc Drugs Ther 17: 303–310.
Kim HT, Kim KP, Lledias F, et al, 2007 Certain pairs of ubiquitin-conjugating enzymes (E2s) and ubiquitin-protein ligases (E3s) synthesize nondegradable forked ubiquitin chains containing all possible isopeptide linkages. J Biol Chem 282: 17375–17386.
Kedar V, McDonough H, Arya R, Li HH, Rockman HA, Patterson C, 2004 Muscle-specific RING finger 1 is a bona fide ubiquitin ligase that degrades cardiac troponin I. Proc Natl Acad Sci U S A 101: 18135–18140.
Solomon V, Goldberg AL 1996 Importance of the ATP-ubiquitin-proteasome pathway in the degradation of soluble and myofibrillar proteins in rabbit muscle extracts. J Biol Chem 271: 26690–26697.
Loo JA, Berhane B, Kaddis CS, et al, 2005 Electrospray ionization mass spectrometry and ion mobility analysis of the 20S proteasome complex. J Am Soc Mass Spectrom 16: 998–1008.
Pearl JM, Nelson DP, Schwartz SM, et al, 2002 Glucocorticoids reduce ischemia-reperfusion-induced myocardial apoptosis in immature hearts. Ann Thorac Surg 74: 830–837.
Chen QM, Alexander D, Sun H, et al, 2005 Corticosteroids inhibit cell death induced by doxorubicin in cardiomyocytes: induction of antiapoptosis, antioxidant, and detoxification genes. Mol Pharmacol 67: 1861–1873.
Reeve JL, Szegezdi E, Logue SE, et al, 2007 Distinct mechanisms of cardiomyocyte apoptosis induced by doxorubicin and hypoxia converge on mitochondria and are inhibited by Bcl-xL. J Cell Mol Med 11: 509–520.
Oehri M, Ninnis R, Girard J, Frey FJ, Keller U, 1996 Effects of growth hormone and IGF-I on glucocorticoid-induced protein catabolism in humans. Am J Physiol 270: E552–558.
Jacob R, Barrett E, Plewe G, Fagin KD, Sherwin RS, 1989 Acute effects of insulin-like growth factor I on glucose and amino acid metabolism in the awake fasted rat. Comparison with insulin. J Clin Invest 83: 1717–1723.
Tomas FM, Knowles SE, Owens PC, et al, 1992 Insulinlike growth factor-I (IGF-I) and especially IGF-I variants are anabolic in dexamethasone-treated rats. Biochem J 282(Pt 1): 91–97.
Jagoe RT, Lecker SH, Gomes M, Goldberg AL, 2002 Patterns of gene expression in atrophying skeletal muscles: response to food deprivation. Faseb J 16: 1697–1712.
Fang CH, Li BG, Tiao G, Wang JJ, Fischer JE, Hasselgren PO, 1998 The molecular regulation of protein breakdown following burn injury is different in fast- and slow-twitch skeletal muscle. Int J Mol Med 1: 163–169.
Sacheck JM, Ohtsuka A, McLary SC, Goldberg AL, 2004 IGF-I stimulates muscle growth by suppressing protein breakdown and expression of atrophy-related ubiquitin ligases, atrogin-1 and MuRF1. Am J Physiol Endocrinol Metab 287: E591–601.
Sandri M, Sandri C, Gilbert A, et al, 2004 Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 117: 399–412.
Stitt TN, Drujan D, Clarke BA, et al, 2004 The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol Cell 14: 395–403.
Otani H, Yamamura T, Nakao Y, et al, 2000 Insulin-like growth factor-I improves recovery of cardiac performance during reperfusion in isolated rat heart by a wortmannin-sensitive mechanism. J Cardiovasc Pharmacol 35: 275–281.
Friehs I, Stamm C, Cao-Danh H, McGowan FX, del Nido PJ, 2001 Insulin-like growth factor-1 improves postischemic recovery in hypertrophied hearts. Ann Thorac Surg 72: 1650–1656.
Tivesten A, Caidahl K, Kujacic V, et al, 2001 Similar cardiovascular effects of growth hormone and insulinlike growth factor-I in rats after experimental myocardial infarction. Growth Horm IGF Res 11: 187–195.
Yamashita K, Kajstura J, Discher DJ, et al, 2001 Reperfusion-activated Akt kinase prevents apoptosis in transgenic mouse hearts overexpressing insulin-like growth factor-1. Circ Res 88: 609–614.
Matthews KG, Devlin GP, Stuart SP, et al, 2005 Intrapericardial IGF-I improves cardiac function in an ovine model of chronic heart failure. Heart Lung Circ 14: 98–103.
Saetrum Opgaard O, Wang PH 2005 IGF-I is a matter of heart. Growth Horm IGF Res 15: 89–94.
Le Corvoisier P, Hittinger L, Chanson P, Montagne O, Macquin-Mavier I, Maison P, 2007 Cardiac effects of growth hormone treatment in chronic heart failure: A meta-analysis. J Clin Endocrinol Metab 92: 180–185.
Gomes AV, Zong C, Ping P, 2006 Protein degradation by the 26S proteasome system in the normal and stressed myocardium. Antioxid Redox Signal 8: 1677–1691.
Willis MS, Patterson C, 2006 Into the heart: the emerging role of the ubiquitin-proteasome system. J Mol Cell Cardiol 41: 567–579.
Majetschak M, Patel MB, Sorell LT, Liotta C, Li S, Pham SM, 2008 Cardiac proteasome dysfunction during cold ischemic storage and reperfusion in a murine heart transplantation model. Biochem Biophys Res Commun 365: 882–888.
Mearini G, Schlossarek S, Willis MS, Carrier L, 2008 The ubiquitin-proteasome system in cardiac dysfunction. Biochim Biophys Acta 1782: 749–763.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chrysis, D., Chagin, A.S. & Sävendahl, L. Insulin-like growth factor-1 restores dexamethasone-induced heart growth arrest in rats: the role of the ubiquitin pathway. Hormones 10, 46–56 (2011). https://doi.org/10.14310/horm.2002.1292
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.14310/horm.2002.1292