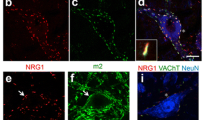
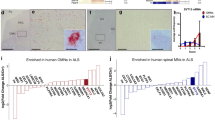
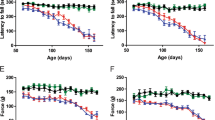
Abstract
Selective degeneration and death of motor neurons in SOD1 mutant-mediated amyotrophic lateral sclerosis (ALS) is accompanied by axonal disorganization and reduced slow axonal transport in the three most frequently used mouse models of mutant SOD1-mediated ALS. To test whether suppression of axonal degeneration (frequently known as Wallerian degeneration) could slow disease development, we took advantage of a spontaneous mouse mutant Wld s (Wallerian degeneration slow) in which the programmed axonal degenerative process that is normally activated after axonal injury is significantly delayed. Despite its effectiveness in delaying axonal loss in other neurodegenerative models, the presence of Wlds did not slow disease onset, ameliorate mutant motor neuron death, axonal degeneration, or preserve synaptic attachments in mice that develop disease from ALS-linked SOD1 mutants SOD1G37R or SOD1G85R. However, presynaptic endings in both the presence and absence of Wlds showed high accumulations of mitochondria and synaptic vesicles, implicating errors of retrograde transport as a consequence of SOD1-mutant damage to axons.
Similar content being viewed by others
References
Araki T., Sasaki Y., and Milbrandt J. (2004) Increased nuclear NAD biosynthesis and SIRT1 activation prevent axonal degeneration. Science 305, 1010–1013.
Bruijn L. I., Becher M. W., Lee M. K., et al. (1997) ALS-linked SOD1 mutant G85R mediates damage to astrocytes and promotes rapidly progressive disease with SOD1-containing inclusions. Neuron 18, 327–338.
Clement A. M., Nguyen M. D., Roberts E. A., et al. (2003) Wild-type nonneuronal cells extend survival of SOD1 mutant motor neurons in ALS mice. Science 302, 113–117.
Cleveland D. W. and Rothstein J. D. (2001) From Charcot to Lou Gehrig: deciphering selective motor neuron death in ALS. Nat. Rev. Neurosci. 2, 806–819.
Coleman M. P. and Perry V. H. (2002) Axon pathology in neurological disease: a neglected therapeutic target. Trends Neurosci. 25, 532–537.
Conforti L., Tarlton A., Mack T. G., et al. (2000) A Ufd2/D4Cole1e chimeric protein and overexpression of Rbp7 in the slow Wallerian degeneration (Wlds) mouse. Proc. Natl. Acad. Sci. USA 97, 11,377–11,382.
Crawford T. O., Hsieh S. T., Schryer B. L., and Glass J. D. (1995) Prolonged axonal survival in transected nerves of C57BL/Ola mice is independent of age. J. Neurocytol. 24, 333–340.
Ferri A., Sanes J. R., Coleman M. P., Cunningham J. M., and Kato A. C. (2003) Inhibiting axon degeneration and synapse loss attenuates apoptosis and disease progression in a mouse model of motoneuron disease. Curr. Biol. 13, 669–673.
Fischer L. R., Culver D. G., Tennant P., et al. (2004) Amyotrophic lateral sclerosis is a distal axonopathy: evidence in mice and man. Exp. Neurol. 185, 232–240.
Gillingwater T. H., Ingham C. A., Coleman M. P., and Ribchester R. R. (2003) Ultrastructural correlates of synapse withdrawal at axotomized neuromuscular junctions in mutant and transgenic mice expressing the Wld gene. J. Anat. 203, 265–276.
Gillingwater T. H. and Ribchester R. R. (2001) Compartmental neurodegeneration and synaptic plasticity in the Wld(s) mutant mouse. J. Physiol 534, 627–639.
Gillingwater T. H., Thomson D., Mack T. G., et al. (2002) Age-dependent synapse withdrawal at axotomised neuromuscular junctions in Wld(s) mutant and Ube4b/Nmnat transgenic mice. J. Physiol 543, 739–755.
Glass J. D., Brushart T. M., George E. B., and Griffin J. W. (1993) Prolonged survival of transected nerve fibres in C57BL/Ola mice is an intrinsic characteristic of the axon. J. Neurocytol. 22, 311–321.
Hafezparast M., Klocke R., Ruhrberg C., et al. (2003) Mutations in dynein link motor neuron degeneration to defects in retrograde transport. Science 300, 808–812.
Hirano A., Donnenfeld H., Sasaki S., and Nakano I. (1984) Fine structural observations of neurofilamentous changes in amyotrophic lateral sclerosis. J. Neuropathol. Exp. Neurol. 43, 461–470.
Kawabuchi M., He J. W., Ting L. W., Zhou C. J., Wang S., and Hirata K. (2000) Morphological features of nerve terminal degeneration as part of the remodeling process in the motor endplate in adult muscles. Ultrastruct. Pathol. 24, 279–289.
Kawamura Y., Dyck P. J., Shimono M., Okazaki H., Tateishi J., and Doi H. (1981) Morphometric comparison of the vulnerability of peripheral motor and sensory neurons in amyotrophic lateral sclerosis. J. Neuropathol. Exp. Neurol. 40, 667–675.
Kokubo Y., Kuzuhara S., Narita Y., et al. (1999) Accumulation of neurofilaments and SOD1-immunoreactive products in a patient with familial amyotrophic lateral sclerosis with I113T SOD1 mutation. Arch. Neurol. 56, 1506–1508.
Krajewski S., Tanaka S., Takayama S., Schibler M. J., Fenton W., and Reed J. C. (1993) Investigation of the subcellular distribution of the bcl-2 oncoprotein: residence in the nuclear envelope, endoplasmic reticulum, and outer mitochondrial membranes. Canc. Res. 53, 4701–4714.
LaMonte B. H., Wallace K. E., Holloway B. A., et al. (2002) Disruption of dynein/dynactin inhibits axonal transport in motor neurons causing late-onset progressive degeneration. Neuron 34, 715–727.
Liu J., Lillo C., Jonsson P. A., et al. (2004) Toxicity of familial ALS-linked SOD1 mutants from selective recruitment to spinal mitochondria. Neuron 43, 5–17.
Lunn E. R., Perry V. H., Brown M. C., Rosen H., and Gordon S. (1989) Absence of Wallerian degeneration does not hinder regeneration in peripheral nerve. Eur. J. Neurosci. 1, 27–33.
Mack T. G., Reiner M., Beirowski B., et al. (2001) Wallerian degeneration of injured axons and synapses is delayed by a Ube4b/Nmnat chimeric gene. Nat. Neurosci. 4, 1199–1206.
Mi W., Conforti L., and Coleman M. P. (2002) Genotyping methods to detect a unique neuroprotective factor (Wld(s)) for axons. J. Neurosci. Meth. 113, 215–218.
Morris R. L. and Hollenbeck P. J. (1995) Axonal transport of mitochondria along microtubules and F-actin in living vertebrate neurons. J. Cell Biol. 131, 1315–1326.
Nangaku M., Sato-Yoshitake R., Okada Y., et al. (1994) KIF1B, a novel microtubule plus end-directed monomeric motor protein for transport of mitochondria. Cell 79, 1209–1220.
Pardo C. A., Xu Z. S., Borchelt D. R., Price D. L., Sisodia S. S., and Cleveland D. W. (1995) Superoxide-dismutase is an abundant component in cell-bodies, dendrites, and axons of motor-neurons and in a subset of other neurons. Proc. Natl. Acad. Sci. USA 92, 954–958.
Pasinelli P., Belford M. E., Lennon N., et al. (2004) Amyotrophic lateral sclerosis-associated SOD1 mutant proteins bind and aggregate with Bcl-2 in spinal cord mitochondria. Neuron 43, 19–30.
Perry V. H., Brown M. C., Lunn E. R., Tree P., and Gordon S. (1990) Evidence that very slow wallerian degeneration in C57BL/Ola mice is an intrinsic property of the peripheral nerve. Eur. J. Neurosci. 2, 802–808.
Rao M. V., Garcia M. L., Miyazaki Y., et al. (2002) Gene replacement in mice reveals that the heavily phosphorylated tail of neurofilament heavy subunit does not affect axonal caliber or the transit of cargoes in slow axonal transport. J. Cell Biol. 158, 681–693.
Rosen D. R., Siddique T., Patterson D., et al. (1993) Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362, 59–62.
Rouleau G. A., Clark A. W., Rooke K., et al. (1996) SOD1 mutation is associated with accumulation of neurofilaments in amyotrophic lateral sclerosis. Ann. Neurol. 39, 128–131.
Samsam M., Mi W., Wessig C., et al. (2003) The Wlds mutation delays robust loss of motor and sensory axons in a genetic model for myelin-related axonopathy. J. Neurosci. 23, 2833–2839.
Shibata N., Hirano A., Kobayashi M., et al. (1996) Intense superoxide dismutase-1 immunoreactivity in intracytoplasmic hyaline inclusions of familial amyotrophic lateral sclerosis with posterior column involvement. J. Neuropathol. Exp. Neurol. 55, 481–490.
Waller A. V. (1850) Experiments on the section of glosso-pharyngeal and hypoglossal nerves of the frog, and observations on the alterations produced thereby in the structure of their primitive fibres. Phil. Trans. R. Soc. Lond. B. Biol. Sci. 140, 423–429.
Wang M. S., Davis A. A., Culver D. G., and Glass J. D. (2002) Wlds mice are resistant to paclitaxel (taxol) neuropathy. Ann. Neurol. 52, 442–447.
Wang M. S., Fang G., Culver D. G., Davis A. A., Rich M. M., and Glass J. D. (2001b) The Wlds protein protects against axonal degeneration: a model of gene therapy for peripheral neuropathy. Ann. Neurol. 50, 773–779.
Wang M., Wu Y., Culver D. G., and Glass J. D. (2001a) The gene for slow Wallerian degeneration (Wld(s)) is also protective against vincristine neuropathy. Neurobiol. Dis. 8, 155–161.
Williamson T. L., Bruijn L. I., Zhu Q. Z., et al. (1998) Absence of neurofilaments reduces the selective vulnerability of motor neurons and slows disease caused by a familial amyotrophic lateral sclerosis-linked superoxide dismutase 1 mutant. Proc. Natl. Acad. Sci. USA 95, 9631–9636.
Williamson T. L. and Cleveland D. W. (1999) Slowing of axonal transport is a very early event in the toxicity of ALS-linked SOD1 mutants to motor neurons. Nat. Neurosci. 2, 50–56.
Wong P. C., Pardo C. A., Borchelt D. R., et al. (1995) An adverse property of a familial ALS-linked SOD1 mutation causes motor-neuron disease characterized by vacuolar degeneration of mitochondria. Neuron 14, 1105–1116.
Yin X., Kidd G. J., Pioro E. P., et al. (2004) Dysmyelinated lower motor neurons retract and regenerate dysfunctional synaptic terminals. J. Neurosci. 24, 3890–3898.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vande Velde, C., Garcia, M.L., Yin, X. et al. The neuroprotective factor Wlds does not attenuate mutant SOD1-mediated motor neuron disease. Neuromol Med 5, 193–203 (2004). https://doi.org/10.1385/NMM:5:3:193
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/NMM:5:3:193