Abstract
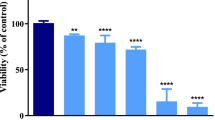
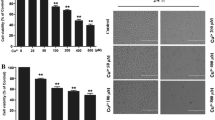
High concentrations of the trace metal zinc (Zn) have previously been shown to provide transient protection of cells from apoptotic death. The molecular mechanisms responsible for this protection are not known. Thus, this work explored the ability of Zn to protect human neurons in culture (NT2-N) from Cu-mediated death and tested the hypotheses that the tumor-suppressor protein p53 plays a role in Cu-induced neuronal death and is part of the mechanism of Zn protection. Copper toxicity (100 µM) resulted in significant apoptotic neuronal death by 12 h. Addition of 100 µM Zn to Cu-treated cells increased neuronal death. However, the addition of 700 µM Zn to Cu-treated cells resulted in neuronal viability that was not different from untreated controls through 24 h. p53 mRNA abundance, while increased by the addition of Cu and 100 µM Zn, was decreased to 50% of control with the addition of 500 µM Zn in Cu-treated cells, and to 10% of control with 700 µM Zn. Consistent with its role as a transcription factor, both Western analysis and immunocytochemistry showed significant increases in nuclear p53 protein levels in Cu toxicity. The role of p53 in Cu-mediated apoptosis was further confirmed by elimination of apoptosis in Cu-treated cells that had been transfected with a dominant-negative p53 construct to prevent p53 expression. Furthermore, the addition of 500–700 µM Zn prevented the movement of p53 into the nucleus suggesting that Zn not only protects neurons from Cu toxicity by regulating p53 mRNA abundance but also by preventing the translocation of p53 to the nucleus.
Similar content being viewed by others
References
Beham A., Marin M. C., Fernandez A., Herrmann J., Brisbay S., Tari A. M., et al. (1997) Bcl-2 inhibits p53 nuclear import following DNA damage. Oncogene 15, 2767–2772.
Berg D., Weishaupt A., Francis M. J., Miura N., Yang X. L., Goodyer I. D., et al. (2000) Changes of copper-transporting proteins and ceruloplasmin in the lentiform nuclei in primary adult-onset dystonia. Ann. Neurol. 47, 827–830.
Canzoniero L. M. T., Turetsky D. M., and Choi D. W. (1999) Measurement of intracellular free zinc concentrations accompanying zinc-induced neuronal death. J. Neurosc. 19(1–6), RC31.
Choi D. W. and Koh J. Y. (1998) Zinc and brain injury. Annu. Rev. Neurosci. 21, 347–375.
Curtin J. C., Dragnev K. H., Sekula D., Christie A. J., Dmitrovsky E., and Spinella M. J. (2001) Retinoic acid activates p53 in human embryonal carcinoma through retinoid receptor-dependent stimulation of p53 transactivation function. Oncogene 20, 2559–2569.
Cuthbert J. A. (1995) Wilson’s disease: a new gene and an animal model for an old disease J. Investig. Med. 43, 323–336.
Fiskum G. (2000) Mitochondrial participation in ischemic and traumatic neural cell death. J. Neurotrauma 17, 843–855.
Fraker P. J. and Telford W. G. (1997) A reappraisal of the role of zinc in life and death decisions of cells. Proc. Soc. Exp. Biol. Med. 215, 229–236.
Frederickson C. J., Suh S. W., Silva D., Frederickson C. J., and Thompson R. B. (2000) Importance of zinc in the central nervous system: The zinc-containing neuron. J. Nutr. 130, 1471S-1483S.
Frederickson C. J., Klitenick M. A., Manton W. I., and Kirkpatrick J. B. (1983) Cytoarchitectonic distribution of zinc in the hippocampus of man and the rat. Brain Res. 273, 335–339.
Hatayama T., Asai Y., Wakatsuki T., Kitamura T., and Imahara H. (1993) Regulation of hsp70 synthesis induced by cupric sulfate and zinc sulfate in thermotolerant HeLa cells. J. Biochem. 114, 592–597.
Ishido M., Suzuki T., Adachi T., and Kunimoto M. (1999) Zinc stimulates DNA synthesis during its antiapoptotic action independently with increments of an antiapoptotic protein, Bcl-2, in porcine kidney LLC-PK (1) cells. J. Pharmacol. Exp. Ther. 290, 923–928.
Kerr J. F., Gobe G. C., Winterford C. M., and Harmon B. V. (1995) Anatomical methods in cell death. Methods Cell Biol. 46, 1–27.
Kim E. Y., Koh J. Y., Kim Y. H, Sohn S., Joe E., and Gwag B. J. (1998) Zn+2 entry produces oxidative neuronal necrosis in cortical cell cultures. Eur. J. Neurosci. 11, 327–334.
Kim H. J., Yoon H. R., Washington S., Chang I. I., Oh Y. J., and Suh Y. J. (1997) DNA strand scission and PC12 cell death induced by salsolinol and copper. Neurosci. Lett. 238, 95–98.
Kubota M., Iida Y., Magata Y., Kitamura Y., Kawashima H., Saji H. (2000) Mechanisms of [2,3-butanedione bis (N4-dimethylthiosemicarbazone)] zinc neurons against N-methyl-D-aspartate receptor-mediated glutamate cytotoxicity. Jpn. J. Pharmacol. 84, 334–338.
Lee J., Prohaska, J. R., and Thiele, D. J. (2001) Essential role for mammalian copper transporter ctr1 in copper homeostasis and embryonic development. PNAS 98, 6842–6847.
Liang S. H., and Clarke M. F. (1999) A bipartite nuclear localization signal is required for p53 nuclear import regulated by a carboxy-terminal domain. J. Biol. Chem. 274, 32699–32703.
Libonati J. P., Fitch C. A., Rutkowski N. J., and Levenson C. W. (2000) Zinc regulation of cobalt-induced apoptosis in cultured human neurons. Nutr. Neurosci. 3, 425–433.
Manev H., Kharlamov E., Uz T., Mason R. P., and Cagnoli C. M. (1997) Characterization of zinc-induced neuronal death in primary cultures of rat cerebellar granule cells. Exp. Neurol. 146, 171–178.
Martin L. J., Al-Abdulla N. A., Brambrink A. M., Kirsch J. R., Sieber F. E., and Portera-Cailliau C. (1998) Neurodegeneration in excitotoxicity, global cerebral ischemia, and target deprivation: A perspective on the contributions of apoptosis and necrosis. Brain Res. Bull. 46, 281–309.
Meplan C., Mann K., and Hainaut P. (1999) Cadmium induces conformational modifications of wild-type p53 and suppresses p53 response to DNA damage in cultured cells. J. Biol. Chem. 274, 31663–31670.
Narayanan V. S., Fitch C. A., and Levenson C. W. (2001) Tumor suppressor protein p53 mRNA and subcellular localization are altered by changes in cellular copper in human Hep G2 cells. J. Nutr. 131, 1427–1432.
Oda K., Arakawa H., Tanaka T., Matsuda K., Tanikawa C., Mori T., et al. (2000) p53 AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by ser-46-phosphorylated p53. Cell 102, 849–862.
Perry D. K., Smyth M. J., Stennicke H. R., Salvesen G. S., Duriez P., Poirier G. G., and Hannun Y. A. (1997) Zinc is a potent inhibitor of the apoptotic protease, caspase-3. A novel target for zinc in the inhibition of apoptosis. J. Biol. Chem. 272, 18530–18533.
Powell S. R. (2000) The antioxidant properties of zinc. J. Nutr. 130, 1447S-1454S.
Reaves, S. K., Fanzo, J. C., Arima, K., Wu, J. Y. J., Wang, Y. R., and Lei, K. Y. (2000) Expression of the p53 tumor suppressor gene is upregulated by depletion of intracellular zinc in HepG2 cells. J. Nutr. 130, 1688–1694.
Sagripanti J. L., Goering P. L., and Lamanna A. (1991) Interaction of copper with DNA and antagonism by other metals. Toxicol. Appl. Pharmacol. 110, 477–485.
Saito T., Okabe M., Hosokawa T., Kurasaki M., Hata A., Endo F., et al. (1999) Immunohistochemical determination of the Wilson copper-transporting P-type ATPase in the brain tissues of the rat. Neurosci. Lett. 266, 13–16.
Sarkar B. (2000) Copper transport and its defect in Wilson disease: characterization of the copper-binding domain of Wilson disease ATPase. J. Inorg. Biochem. 79, 187–191.
Scheinberg H. and Sternlieb I. (1984) Wilson’s Disease, Vol. XXIII, W. B. Saunders Company, Philadelphia, PA.
Scheinberg I. H. and Sternlieb I. (1979) The role of radiocopper in the diagnosis of Wilson’s disease. Gastroenterology 77, 138–142.
Schmidt-Kastner R., Truettner J., Zhao W., Belayev L., Krieger C., Busto R., and Ginsberg M. D. (2000) Differential changes of bax, caspase-3 and p21 mRNA expression after transient focal brain ischemia in the rat. Brain Res. Mol. Brain Res. 79, 88–101.
Sensi S. L., Yin H. Z., Carriedo S. G., Rao S. S., and Weiss J. H. (1999) Preferential Zn+2 influx through Ca+2-permeable AMPA/kainate channels triggers prolonged mitochondrial superoxide production. Proc. Natl. Acad. Sci. USA 96, 2414–2419.
Sheline C. T., Behrens M. M., and Choi D. W. (2000) Zinc-induced cortical neuronal death: contribution of energy failure attributable to loss of NAD+ and inhibition of glycolysis. J. Neurosci. 20, 3139–3146.
Strand S., Hofmann W. J., Grambihler A., Hug H., Volkmann M., Otto G., et al. (1998) Hepatic failure and liver cell damage in acute Wilson’s disease involve CD95 (APO-1/Fas) mediated apoptosis. Nat. Med. 4, 588–593.
Tkeshelashvili L. K., McBride T., Spence K., and Loeb L. A. (1991) Mutation spectrum of copper-induced DNA damage. J. Biol. Chem. 266, 6401–6406.
Truong-Tran A. Q., Ho L. H., Chai F., and Zalewski P. D. (2000) Cellular zinc fluxes and the regulation of apoptosis/gene-directed cell death. J. Nutr. 130, 1459S-1466S.
Waggoner D. J., Bartnikas T. B., and Gitlin J. D. (1999) The role of copper in neurodegenerative disease. Neurobiol. Dis. 6, 221–230.
Wood R. J. (2000) Assessment of marginal zinc status in humans. J. Nutr. 130, 1350S-1354S.
Xiang H., Hochman D. W., Saya H., Fujiwara T., Schwartzkroin P. A., and Morrison R. S. (1996) Evidence for p53-mediated modulation of neuronal viability. J. Neurosci. 16, 6753–6765.
Zago M. P. and Oteiza P. I. (2001) The antioxidant properties of zinc: Interactions with iron and antioxidants. Free Rad. Biol. Med. 31, 266–274.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
VanLandingham, J.W., Fitch, C.A. & Levenson, C.W. Zinc inhibits the nuclear translocation of the tumor suppressor protein p53 and protects cultured human neurons from copper-induced neurotoxicity. Neuromol Med 1, 171–182 (2002). https://doi.org/10.1385/NMM:1:3:171
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/NMM:1:3:171