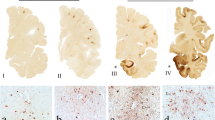
Abstract
Traumatic brain injury is a cause of high mortality and morbidity and is an area of intense research. Apoptosis plays a crucial role in the pathogenesis of head injury, and the inhibition of apoptosis can potentially reverse the deleterious effects and lead to better functional outcome. Elucidation of the apoptotic pathway and its role in traumatic brain injury will provide potential targets for therapeutic intervention. This article aims to review the current wealth of literature on apoptosis and traumatic head injury and explores the current status of therapeutic strategies available.
Similar content being viewed by others
References
Rosner MJ, Rosner SD, Johnson AH. Cerebral perfusion pressure: management protocol and clinical results. J Neurosurg. 1995;83:949–962.
Rosner MJ. Introduction to cerebral perfusion pressure management. Neurosurg Clin N Am 1995;6:761–773.
Cruz J. The first decade of continuous monitoring of jugular bulb oxyhemoglobin saturation: management strategies and clinical outcome. Crit Care Med 1998;26:344–351.
Czosnyka M, Smielewski P, Czosnyka Z, et al. Continuous assessment of cerebral autoregulation: clinical and laboratory experience. Acta Neurochir Suppl 2003;86:581–585.
Steiner LA, Coles JP, Johnston AJ, et al. Assessment of cerebrovascular autoregulation in head-injured patients: a validation study. Stroke 2003;34:2404–2409.
Zuzarte-Luis V, Hurle JM. Programmed cell death in the developing limb. Int J Dev Biol 2002;46:871–876.
Gorczyca W, J Gong, Z Darzynkiewicz. Detection of DNA strand breaks in individual apoptotic cells by the in situ terminal deoxynucleotidyl transferase and nick translation assays. Cancer Res 1993;53:1945–1951.
Kerr JFR, Wyllie AH, Currie AR Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972;26:239–257.
Rideout HJ, Stefanis L. Caspase inhibition: a potential therapeutic strategy in neurological diseases. Histol Histopathol 2001;16:895–908.
Newcomb JK, Zhao X, Pike BR, Hayes RL. Temporal profile of apoptotic-like changes in neurons and astrocytes following controlled cortical impact injury in the rat. Exp Neurol 1999;158:76–88.
Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P. Molecular biology of the cell, 4th ed. New York: Garland Science, 2002:983–1064.
Lodish HA, Berk SL, Matsudaira P, et al. Molecular Cell Biology, 4th ed. New York: W.H. Freeman, 2000:1054–1084.
Dexter TM, Raff MC, Wyllie AH. The role of apoptosis in development, tissue homeostasis and malignancy: death from inside out. London: Chapman & Hall: The Royal Society, 1995:1–45.
Raff MC. Social controls on cell survival and cell death. Nature 1992;356:397–399.
Thompson CB. Apoptosis in the pathogenesis and treatment of disease. Science 1995;267:1456–1462.
Bortner CD, Oldenburg NBE, Cidlowski JA. The role of DNA fragmentation in apoptosis. Trends Cell Biol. 1995;5:21–26.
A novel assay for apoptosis: flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J Immunol Methods 1995;184:39–51.
Gorczyca W, Tuziak T, Kram A, Melamed MR, Darzynkiewicz Z. Detection of apoptosis-associated DNA strand breaks in fine-needle aspiration biopsies by in situ end labeling of fragmented DNA. Cytometry 1994;15:169–175.
Swat W, Ignatowicz L, Kisielow P. Detection of apoptosis of immature Cd4+8+ thymocytes by flow cytometry. J. Immunol Methods 1991;137:79–87.
Sachs LM, Abdallah B, Hassan A, et al. Apoptosis in Xenopus tadpole tail muscles involves Bax-dependent pathways. FASEB J 1997;11:801–808.
Cande C, Cecconi F, Dessen P, Kroemer G. Apoptosis-inducing factor (AIF): key to the conserved caspase-independent pathways of cell death? J Cell Sci 2002;115:4727–4734.
Furth PA, Bar-Peled U, Li M. Apoptosis and mammary gland involution: reviewing the process. Apoptosis 1997;2:19–24.
Yazdanbakhsh K, Choi JW, Li Y, Lau LF, Choi Y. Cyclosporin A blocks apoptosis by inhibiting the DNA binding activity of the transcription factor Nur77. Proc Natl Acad Sci USA 1995;92:437–441.
Grodzicky T, Elkon KB. Apoptosis: a case where too much or too little can lead to autoimmunity. Mt Sinai J Med 2002;69:208–219.
Curtin JF, Cotter TG. Live and let die: regulatory mechanisms in Fas-mediated apoptosis. Cell Signal 2003;15:983–992.
Darzynkiewicz Z, Li X, Gong J. Assay of cell viability: discrimination of cells dying by apoptosis. In: Darzynkiewicz Z, Robinson JP, Crissman HA, eds. Flow cytometry, 2nd ed. Academic Press, New York, 1994:15–38.
Berger RP, Pierce MC, Wisniewski SR, et al. Neuron-specific enolase and S100B in cerebrospinal fluid after severe traumatic brain injury in infants and children. Pediatrics 2002;109:E31.
Raghupathi R, Graham DI, McIntosh TK. Apoptosis after traumatic brain injury. J Neurotrauma 2000;17:927–938.
Liou AK, Clark RS, Henshall DC, Yin XM, Chen J. To die or not to die for neurons in ischemia, traumatic brain injury and epilepsy: a review on the stress-activated signaling pathways and apoptotic pathways. Prog Neurobiol 2003;69:103–142.
Zhang X, Chen J, Graham SH, et al. Intranuclear localization of apoptosis-inducing factor (AIF) and large scale DNA fragmentation after traumatic brain injury in rats and in neuronal cultures exposed to peroxynitrite. J Neurochem 2002;82:181–191.
Lenzlinger PM, Marx A, Trentz O, Kossmann T, Morganti-Kossmann MC. Prolonged intrathecal release of soluble Fas following severe traumatic brain injury in humans. J Neuroimmunol 2002;122:167–174.
Keane RW, Kraydieh S, Lotocki G, Alonso OF, Aldana P, Dietrich WD. Apoptotic and antiapoptotic mechanisms after traumatic brain injury. J Cereb Blood Flow Metab 2001;21:1189–1198.
Beer R, Franz G, Schopf M, et al. Expression of Fas and Fas ligand after experimental traumatic brain injury in the rat. J Cereb Blood Flow Metab 2000;20:669–677.
Peli J, Schroter M, Rudaz C, et al. Oncogenic Ras inhibits Fas ligand-mediated apoptosis by downregulating the expression of Fas. EMBO J 1999;18:1824–1831.
Maecker HL, Koumenis C, Giaccia AJ. p53 promotes selection for Fas-mediated apoptotic resistance. Cancer Res 2000;60:4638–4644.
Zhang X, Graham SH, Kochanek PM, et al. Caspase-8 expression and proteolysis in human brain after severe head injury. FASEB J 2003;17:1367–1369.
Knoblach SM, Nikolaeva M, Huang, et al. Multiple caspases are activated after traumatic brain injury: evidence for involvement in functional outcome. J Neurotrauma 2002;19:1155–1170.
Robertson GS, Crocker SJ, Nicholson DW, Schulz JB. Neuroprotection by the inhibition of apoptosis. Brain Pathol 2000;10:283–292.
Clark RS, Chen J, Watkins SC, et al. Apoptosis-suppressor gene bcl-2 expression after traumatic brain injury in rats. J Neurosci 1997;17:9172–9182.
Clark RS, Kochanek PM, Chen M, et al. Increases in Bcl-2 and cleavage of caspase-1 and caspase-3 in human brain after head injury. FASEB J 1999;13:813–821.
Yang J, Liu X, Bhalla K, et al. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 1997;275:1129–1132.
Vink R, Nimmo AJ, Cernak I. An overview of new and novel pharmacotherapies for use in traumatic brain injury. Clin Exp Pharmacol Physiol 2001;28:919–921.
Beer R, Franz G, Srinivasan A, et al. Temporal profile and cell subtype distribution of activated caspase-3 following experimental traumatic brain injury. J Neurochem 2000;75:1264–1273.
Eldadah BA, Faden AI. Caspase pathways, neuronal apoptosis and CNS injury. J Neurotrauma 2000;17:811–829.
Clark RS, Kochanek PM, Watkins SC, et al. Caspase-3 mediated neuronal death after traumatic brain injury in rats. J Neurochem 2000;74:740–753.
Sanchez Mejia RO, Ona VO, Li M, Friedlander RM. Minocycline reduces traumatic brain injury-mediated caspase-1 activation, tissue damage, and neurological dysfunction. Neurosurgery 2001;48:1393–1399.
Szabo I, Zoratti M. The giant channel of the inner mitochondrial membrane is inhibited by cyclosporin A. J Biol Chem 1991;266:3376–3379.
Suvillian PG Rabchevsky AG, Hicks RR, Gibson TR, Fletcher-Turner A, Scheff SW. Dose-response curve and optimal dosing regimen of cyclosporin A after traumatic brain injury in rats. Neuroscience 2000;101:289–295.
Buki A, Okonkwo DO, Povlishock JT. Postinjury cyclosporin A administration limits axonal damage and disconnection in traumatic brain injury. J Neurotrauma 1999;16:511–521.
Lallemend F, Lefebvre PP, Hans G, et al. Substance P protects spiral ganglion neurons from apoptosis via PKC-Ca2+-MAPK/ERK pathways. J Neurochem 2003;87:508–521.
LaPlaca MC, Zhang J, Raghupathi R, et al. Pharmacologic inhibition of poly (ADP-ribose) polymerase is neuroprotective following traumatic brain injury in rats. Neurotrauma 2001;18:369–376.
Movsesyan VA, Yakovlev AG, Fan L, Faden AI. Effect of serine protease inhibitors on posttraumatic brain injury and neuronal apoptosis. Exp Neurol 2001;167:366–375.
Lewen A, Skoglosa Y, Clausen F, et al. Paradoxical increase in neuronal DNA fragmentation after neuroprotective free radical scavenger treatment in experimental traumatic brain injury. J Cereb Blood Flow Metab 2001;21:344–350.
Sullivan PG, Keller JN, Bussen WL, Scheff SW. Cytochrome c release and caspase activation after traumatic brain injury. Brain Res 2002;949:88–96.
Lewen A, Fujimura M, Sugawara T, Matz P, Copin JC, Chan PH. Oxidative stress-dependent release of mitochondrial cytochrome c after traumatic brain injury. J Cereb Blood Flow Metab 2001;21:914–920.
Yakovlev AG, Di X, Movsesyan V, et al. Presence of DNA fragmentation and lack of neuroprotective effect in DFF45 knockout mice subjected to traumatic brain injury. Mol Med 2001;7:205–216.
Nathoo N, Narotam PK, Agrawal DK, Connolly CA, van Dellen JR, Barnett GH, Chetty R. Influence of apoptosis on neurological outcome following traumatic cerebral contusion. J Neurosurg 2004;101:233–240.
Eastman P. Why neuroprotective drugs fail: questions about cell death present new challenges. Neurol Today. August 2003.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wong, J., Hoe, N.W., Zhiwei, F. et al. Apoptosis and traumatic brain injury. Neurocrit Care 3, 177–182 (2005). https://doi.org/10.1385/NCC:3:2:177
Issue Date:
DOI: https://doi.org/10.1385/NCC:3:2:177