Abstract
Fludarabine is an active agent in low-grade non-Hodgkin’s lymphoma and chronic lymphocytic leukemia. Paclitaxel is also active in patients with refractory lymphoma, and preclinical data suggest an additive effect with fludarabine in vitro. We performed a phase I trial of fludarabine (25 mg/m2 d 1–3) plus a 3-h infusion of paclitaxel (125, 150, or 175 mg/m2) on d 3 every 28 d in 13 patients with non-Hodgkin’s lymphoma. The paclitaxel dose was escalated in cohorts of 3–4 patients using standard phase I design schema. Dose-limiting toxicity was defined as febrile neutropenia, platelet nadir less than 50,000/µL, or grade 3–4 nonhematologic toxicity.
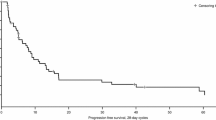
Thirteen patients were accrued to the study, 8 of these 13 patients (62%) had received prior chemotherapy. At the 125-, 150-, and 175-mg/m2 dose levels of paclitaxel, dose-limiting toxicity occurred in 1/4, 0/4, and 0/4 patients, respectively. The single patient with dose-limiting toxicity had febrile neutropenia. Partial response occurred in two of eight patients with low-grade lymphoma and none of five patients with other types of lymphoma. A paclitaxel dose of 175 mg/m2 given as a 3-h infusion on d 3 in conjunction with fludarabine (25 mg/m2 d 1–3 every 4 wk) is a well-tolerated regimen for non-Hodgkin’s lymphoma. Further study will be required in order to determine whether the fludarabine-paclitaxel is more active than fludarabine alone in patients with low-grade lymphoma and chronic lymphocytic leukemia.
Similar content being viewed by others
References
Adkins, J.C., Peters, D.H., Markham, A., et al. (1997). Fludarabine: an update of its pharmacology and use in the treatment of hematological malignancies. Drugs 53:1005–1037.
Solal-Céligny, P., Brice, P., Brousse, N., Caspard, H., Bastion, Y., Haïoun, C., et al. (1996). Phase II trial of fludarabine monophosphate as first line treatment in patients with advanced follicular lymphoma: a multicenter study by the Groupe d’Etude des Lymphomes de l’ adulte. J. Clin. Oncol. 14:514–579.
Foran, J.M., Rohatiner, A.Z., Coiffier, B., Barbui, T., Johnson, S.A., Hiddemann, W., et al. (1999). Multicenter phase II study of fludarabine phosphate for patients with newly diagnosed lymphoplasmacytoid lymphoma, Waldenström’s macroglobulinemia, and mantle cell lymphoma. J. Clin. Oncol. 17:546–553.
Pigaditou, A., Rohatiner, A.Z.S., Whelan, J.S., Johnson, P.W., Ganjoo, R.K., Rossi, A., et al. (1993). Fludarabine in low-grade lymphoma. Semin. Oncol. 20(Suppl. 7):24–27.
Zinzani, P.L., Lauria, F., Rondelli, D., Benfenati, D., Raspadori, D., Bocchia, M., et al. (1993). Fludarabine: an active agent in the treatment of previously treated and untreated low-grade non-Hodgkin’s lymphoma. Ann. Oncol. 4:575–578.
Hochster, H.S., Kim, K., Green, M.D., Mann, R.B., Neiman, R.S., Oken, M.M., et al. (1992). Activity of fludarabine phosphate in previously treated non-Hodgkin’s low-grade lymphoma: results of an Eastern Cooperative Oncology Group Study. J. Clin. Oncol. 10:28–32.
Redman, J.R., Cabanillas, F., Valasques, W.S., McLaughlin, P., Hagemeister, F.B., Forrest, S., et al. (1992). Phase II trial of fludarabine phosphate in lymphoma: an effective new agent in low-grade lymphoma. J. Clin. Oncol. 10:790–794.
O’Brien, M.E., Matutes, E., Cunningham, D., Hill, M., Emmett, E., Ellis, P.A., et al. (1994). Fludarabine in lymphoprolifrative disorders: the Royel Marsden Hospital experience. Leuk. Lymphoma 14(Suppl. 2):17–23.
Hiddemann, W., Unterhalt, M., Pott, C., Wormann, B., Sandford, D., Freuund, M., et al. (1993). Fludarabine single agent therapy for relapsed low-grade non-Hodgkin’s lymphomas: a phase II study of the German Low-Grade Non-Hodgkin’s Lymphoma Study Group. Semin. Oncol. 20(Suppl. 7):28–31.
Pott, C., Unterhalt, M., Sandford, D., et al. (1994). Fludarabine in combination with Mitoxatrone and Dexamethasone in relapsed and refractory low-grade non-Hodgkin’s lymphoma. Onkologie 17(Suppl. 2):114 (abstract).
Whelan, J.S., Davis, C.L., Rule, S., Ranson, M., Smith, O.P., Mehta, A.B., et al. (1991). Fludarabine phosphate for the treatment of low-grade lymphoid malignancy. Br. J. Cancer 64:120–123.
Leiby, J.M., Snider, K.M., Kraut, E.H., Metz, E.N., Malspeis, L., Grever, M.R., et al. (1987). Phase II trial of 9-β-d- arabinofuranosyl-2-fluoroadanine 5′- monophosphate in non-Hodgkin’s lymphoma: prospective comparison of response with deoxycytidine kinase activity. Cancer Res. 47:2719–2722.
Whelan, J.S., Ganjoo, R., Johnson, P.W., Rohatiner, A.Z., and Lister, T.A. (1993). Treatment of low-grade non-Hodgkin’s lymphoma with fludarabine. Leuk. Lymphoma 10(Suppl.):35–37.
Falkson, C.I. (1996). Fludarabine: a phase II trial in patients with previously treated low-grade lymphoma. Am. J. Clin. Oncol. 19:268–270.
Fenchel, K., Bergmann, L., Wijermans, P., Engert, A., Pralle, H., Mitrou, P.S., et al. (1995). Clinical experience with fludarabine and its immunosuppressive effects in pretreated chronic lymphocytic leukemias and low-grade lymphomas. Leuk. Lymphoma 18:485–492.
Gillis, S., Dan, E.J., Cass, Y., Rochlemer, R.R. and Polliack, A. (1994). Activity of fludarabine in refractory chronic lymphocytic leukemia and low-grade non-Hodgkin’s lymphoma. the Jerusalem experience. Leuk. Lymphoma 15:173–175.
Decuadin, D., Bosq, J., Tertian, G., Nedellec, G., Bennaceur, A., Venuat, A., et al. (1998). Phase II trial fludarabine monophosphate in patients with mantle cell lymphomas. J. Clin. Oncol. 16:579–583.
Klasa, R., Meyer, R., Shustik, C., Sawka, C., Smith, A., Grenier, J.E., et al. (1999). Fludarabine versus CVP in previously treated patients with progressive low-grade non-Hodgkin’s lymphoma. Proc. Am. Soc. Clin. Oncol. 18:9a.
Rai, K.R., Peterson, B.L., Kolitz, J., Applebaum, F.R., Shepherd, L., Hines, J., et al. (1996). A randomized comparison of fludarabine and chlorambucil for patients with previously untreated chronic lymphocytic leukemia: a CALGB, SWOG, CTG/NCI-C, and ECOG Intergroup study. Blood 88:141a (abstract).
Johnson, S., Smith, A.G., Loffler, H., Osby, E., Juliusson, G., Emmerich, B., et al. (1996). Multicentre prospective randomized trial of fludarabine versus cyclophosphamide, doxorubicin, and prednisone (CAP) for treatment of advanced stage chronic lymphocytic leukemia: French Cooperative Group on CLL. Lancet 347:1432–1438.
Leporrier, M., Chevret, S., Cazin, B., et al. (1997). Randomized comparison of fludarabine, CAP, and CHOP in 696 previously untreated stage B and C chronic lymphocytic leukemia (CLL): early stopping of the CAP accrual. Blood 90:529a (abstract).
Rowinsky, E.K., Donehower, R.C., Jones, R.J. and Tucker, R.W. (1988). Microtubule changes and cytotoxicity in leukemic cell lines treated with taxol. Cancer Res. 48:4093–4100.
Haldar, S., Basu, A. and Croce, C.M. (1997). Bcl2 is the guardian of microtubule integrity. Cancer Res. 57:229–233.
Kwock, R., Nandy, P., Solorzano, M. and Avramis, V.I. (1996). Combination regimens of fludarabine and ara-C followed by taxanes (docetaxel and paclitaxel) against human leukemia T-cell lines, CEM/0 and CME/ara-C/7A. Proc. Am. Assoc. Cancer Res. 37:A2753.
Avramis, V.I., Nandy, P., Kwock, R., Solozano, M.M., Mukherjee, S.K., Danenberg, P., et al. (1998). Increased p21/WAF-1 and p53 protein levels following sequential three drug combination regimen of fludarabine, cytarabine, and docetaxel induces apoptosis in human leukemia cells. Anticancer Res. 18:2327–2338.
Younes, A., Sarris, A., Melnyk, A., Romaguera, J., McLaughlin, P., Swan, F., et al. (1995). Three-hour paclitaxel infusion in patients with refractory and relapsed non-Hodgkin’s lymphoma. J. Clin. Oncol. 13:583–587.
Press, O., LeBlanc, M., O’Rourke, T.J., Gagnet, S., Chapman, R.A., Balcerzak, S.P., et al. (1998). Phase II trial of paclitaxel by 24-hour continuous infusion for relapsed non-Hodgkin’s lymphoma. Southwest Oncology Group trial 9246. J. Clin. Oncol. 16:574–578.
Wilson, W.H., Chabner, B.A., Bryant, G., Wittes, R.E., Bates, S., Fojo, A., et al. (1995). Phase II study of paclitaxel in relapsed non-Hodgkin’s lymphomas. J. Clin. Oncol. 13:381–386.
DiRaimondo, F., Palumbo, G.A., Romeo, M.A., Galvagno, F., Stagno, F., Morabito, F., et al. (1997). Evaluation of Taxol cytotoxicity on B-CLL cell in vitro. Leuk. Lymphoma 26:115–119.
Oken, M.M., Creech, R.H., Tormey, D.C., Horton, J., Doris, T.E., McFadden, E.T., et al. (1982). Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 5:649–655.
Younes, A., Rodriguez, M.A., Mclaughlin, P., North, L., Sarris, A.H., Pate, O., et al. (1997). Phase I study of fludarabine and paclitaxel for the treatment of low-grade non-Hodgkin’s lymphoma. Leukemia Lymphoma 26:77–82.
Rohatiner, A.Z. (2000). New Approaches in Follicular Lymphoma, pp 465–72, American Society of Clinical Oncology, Atlanta, GA.
Byrd, J.C. (1999). Therapy of Chronic Lymphocytic Leukemia: It Is Time to Move on to the Next Important Question, pp 167–77, American Society of Clinical Oncology, Atlanta, GA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abbasi, M.R., Sparano, J.A., Sarta, C. et al. Phase I trial of fludarabine and paclitaxel in non-hodgkin’s lymphoma. Med Oncol 20, 53–58 (2003). https://doi.org/10.1385/MO:20:1:53
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/MO:20:1:53