Abstract
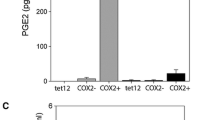
Cyclooxygenase (COX)-2 plays an important role in the development of cancer and has been recognized as a potential therapeutic target. Because nonsteroidal anti-inflammatory drugs (NSAIDs) are able to inhibit the activity of this enzyme, the potential efficacy of such drugs for purposes of cancer prevention or therapy is an area of intense research. Therefore, it is of critical importance to unequivocally determine the expression levels of COX-2 protein in tumor cells. In this regard, there are several conflicting reports in the literature where the same type of tumor cell lines were reported as COX-2 positive and as COX-2 negative. We found that during Western blot analysis of COX-2 positive and COX-2 negative cells, different antibodies to COX-2 protein are able to generate strong signals, which are false-positives and can be confused with COX-2. Thus, we believe that some of the conflicting reports on COX-2 expression in tumor cell lines could be the result of improper interpretation of the Western blot signals. Here, we present some of these pitfalls and suggest the inclusion of appropriate controls to unequivocally identify COX-2 protein levels.
Similar content being viewed by others
References
Dannenberg, A. J. and Subbaramaiah, K. (2003) Targeting cyclooxygenase-2 in human neoplasia: rationale and promise. Cancer Cell 4, 431–436.
Tegeder, I., Pfeilschifter, J., and Geisslinger, G. (2001) Cyclooxygenase-independent actions of cyclooxygenase inhibitors. Faseb. J. 15, 2057–2072.
Wun, T., McKnight, H., and Tuscano, J. M. (2004) Increased cyclooxygenase-2 (COX-2): a potential role in the pathogenesis of lymphoma. Leuk. Res. 28, 179–190.
Kardosh, A., Wang, W., Uddin, J., et al. (2005) Dimethyl-celecoxib (DMC), a derivative of celecoxib that lacks cyclooxygenase-2-inhibitory function, potently mimics the anti-tumor effects of celecoxib on Burkitt's lymphoma in vitro and in vivo. Cancer Biol. Ther. 4, 571–582.
Kobayashi, M., Nakamura, S., Shibata, K., et al. (2005) Etodolac inhibits EBER expression and induces Bcl-2-regulated apoptosis in Burkitt's lymphoma cells. Eur. J. Haematol. 75, 212–220.
Zhang, M., Abe, Y., Matsushima, T., Nishimura, J., Nawata, H., and Muta, K. (2005) Selective cyclooxygenase 2 inhibitor NS-398 induces apoptosis in myeloma cells via a Bcl-2 independent pathway. Leuk. Lymphoma 46, 425–433.
Kardosh, A., Soriano, N., Liu, Y.-T., et al. (2005) Multi-target inhibition of drug-resistant multiple myeloma cell lines by dimethyl-celecoxib (DMC), a non-COX-2-inhibitory analog of celecoxib. Blood 106, 4330–4338.
Harlow, E., and Lane, D. (1988) Antibodies: A Laboratory Manual. Cold Spring Harbor Laboratory. Cold Spring Harbor, NY.
Wu, R.-C. and Schönthal, A. H. (1997) Activation of p53–p21walf pathway in response to disruption of cell-matrix interactions. J. Biol. Chem. 272, 29091–29098.
Krysan, K., Dalwadi, H., Sharma, S., Pold, M. and Dubinett, S. (2004) Cyclooxygenase 2-dependent expression of survivin is critical for apoptosis resistance in non-small cell lung cancer. Cancer Res. 64, 6359–6362.
Ausubel, F.M., Brent, R., Kingston, R. E., et al., editors (1994) Current Protocols in Molecular Biology. New York, John Wiley & Sons, Inc.
Barnett, J., Chow, J., Ives, D., et al. (1994) Purification, characterization and selective inhibition of human prostaglandin G/H synthase 1 and 2 expressed in the baculovirus system. Biochim. Biophys. Acta 1209, 130–139.
Percival, M. D., Ouellet, M., Vincent, C. J., Yergey, J. A., Kennedy, B. P. and O'Neill, G. P. (1994) Purification and characterization of recombinant human cyclooxygenase-2. Arch. Biochem. Biophys. 315, 111–118.
Roos, K. L. and Simmons, D. L. (2005) Cyclooxygenase variants: the role of alternative splicing. Biochem. Biophys. Res. Commun. 338, 62–69.
Eibl, G., Bruemmer, D., Okada, Y., et al. (2003) PGE(2) is generated by specific COX-2 activity and increases VEGF production in COX-2-expressing human pancreatic cancer cells. Biochem. Biophys. Res. Commun. 306, 887–897.
Raut, C. P., Nawrocki, S., Lashinger, L. M., et al. (2004) Celecoxib inhibits angiogenesis by inducing endothelial cell apoptosis in human pancreatic tumor xenografts. Cancer Biol. Ther. 3, 1217–1224.
Uchida, K., Schneider, S., Yochim, J. M., et al. (2005) Intratumoral COX-2 gene expression is a predictive factor for colorectal cancer response to fluoropyrimidine-based chemotherapy. Clin. Cancer Res. 11, 3363–3368.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, YT., Kardosh, A., Cooc, J. et al. Potential misidentification of cyclooxygenase-2 by western blot analysis and prevention through the inclusion of appropriate controls. Mol Biotechnol 34, 329–335 (2006). https://doi.org/10.1385/MB:34:3:329
Issue Date:
DOI: https://doi.org/10.1385/MB:34:3:329