Abstract
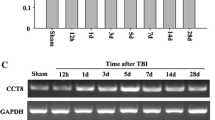
Activity-dependent neuroprotective protein (ADNP), a crucial brain development factor, contains a unique sequence, termed NAPVSIPQ, which protects mice against closed head injury (CHI). The aim of this study was to determine whether CHI affects ADNP mRNA expression in the injured brain hemisphere. Male C57JBL/6J mice were subjected to CHI. Brains were removed 5 h, 24 h, 7 d, and 29 d post-CHI. A comparison was made between ADNP mRNA in the injured versus the noninjured hemisphere using real-time polymerase chain reaction. A nonsignificant change (p>0.05) was found 5 h, 24 h, and 7 d post-CHI. However, a significant increase (p < 0.05) in ADNP mRNA expression was detected in the injured cerebral hemisphere 29 d post-CHI. The data presented may be associated with ADNP’s crucial involvement in brain development and response to injury.
Similar content being viewed by others
References
Bassan M., Zamostiano R., Davidson A., Pinhasov A., Giladi E., Perl O., et al. (1999) Complete sequence of a novel protein containing a femtomolar-activity-dependent neuroprotective peptide. J. Neurochem. 72, 1283–1293.
Beni-Adani L., Gozes I., Cohen Y., Assaf Y., Steingart R. A., Brenneman D. E., et al. (2001) A peptide derived from activity-dependent neuroprotective protein (ADNP) ameliorates injury response in closed head injury in mice. J. Pharmacol. Exp. Ther. 296, 57–63.
Benimetskaya L., Loike J. D., Khaled Z., Loike G., Silverstein S. C., Cao L., et al. (1997) Mac-1 (CD11b/CD18) is an oligodeoxynucleotide-binding protein. Nat. Med. 3, 414–420.
Brenneman D. E., Spong C. Y., and Gozes I. (2000) Protective peptides derived from novel glial proteins. Biochem. Soc. Trans. 28, 452–455.
Chen Y., Constantini S., Trembovler V., Weinstock M., and Shohami E. (1996) An experimental model of closed head injury in mice: pathophysiology, histopathology and cognitive deficits. J. Neurotrauma 13, 557–568.
Delgado M., Abad C., Martinez C., Juarranz M. G., Arranz A., Gomariz R. P., and Leceta J. (2002) Vasoactive intestinal peptide in the immune system: potential therapeutic role in inflammatory and autoimmune diseases. J. Mol. Med. 80, 16–24.
Delgado M. and Ganea D. (2003) Vasoactive intestinal peptide prevents activated microglia-induced neurodegeneration under inflammatory conditions: potential therapeutic role in brain trauma. FASEB J. 17, 1922–1924.
Fuhrmann G., Chung A. C., Jackson K. J., Hummelke G., Baniahmad A., Sutter J., et al. (2001) Mouse germline restriction of Oct4 expression by germ cell nuclear factor. Dev. Cell 1, 377–387.
Glaser T., Jepeal L., Edwards J. G., Young S. R., Favor J., and Maas R. L. (1994) PAX6 gene dosage effect in a family with congenital cataracts, aniridia, anophthalmia and central nervous system defects. Nat. Genet. 7, 463–471.
Gozes I. (2001) Neuroprotective peptide drug delivery and development: potential new therapeutics. Trends Neurosci. 24, 700–705.
Gozes I., Alcalay R., Giladit E., Pinhasov A., Furman S., Brenneman D. E. (2002) NAP accelerates the performance of normal rats in watermaze. J. Mol. Neurosci. 19, 167–170.
Gozes I., Divinsky I., Pilzer I., Fridkin M., Brenneman D. E., and Spier A. D. (2003) From vasoactive intestinal peptide (VIP) through activity-dependent neuroprotective protein (ADNP) to NAP: a view of neuroprotection and cell division. J. Mol. Neurosci. 20, 315–322.
Hailer N. P., Glomsda B., and Blaheta R. A. (2001) Astrocytic factors down-regulate the expression of major histocompatibility complex-class-II and intercellular adhesion molecule-1 on human monocytes. Neurosci. Lett. 298, 33–36.
Hallbergson A. F., Gnatenco C., and Peterson D. A. (2003) Neurogenesis and brain injury: managing a renewable resource for repair. J. Clin. Invest. 112, 1128–1133.
Holmin S. and Mathiesen T. (1999) Long-term intracerebral inflammatory response after experimental focal brain injury in rat. Neuroreport 10, 1889–1891.
Hoshino S., Tamaoka A., Takahashi M., Kobayashi S., Furukawa T., Oaki Y., et al. (1998) Emergence of immunoreactivities for phosphorylated tau and amyloid-beta protein in chronic stage of fluid percussion injury in rat brain. Neuroreport 9, 1879–1883.
Koshinaga M., Katayama Y., Fukushima M., Oshima H., Suma T., and Takahata T. (2000) Rapid and widespread microglial activation induced by traumatic brain injury in rat brain slices. J. Neurotrauma 17, 185–192.
Leker R. R. and Shohami E. (2002) Cerebral ischemia and trauma-different etiologies yet similar mechanisms: neuroprotective opportunities. Brain Res. Brain Res. Rev. 39, 55–73.
Lu D., Mahmood A., Zhang R., and Copp M. (2003) Upregulation of neurogenesis and reduction in functional deficits following administration of DEtA/NONOate, a nitric oxide donor, after traumatic brain injury in rats. J. Neurosurg. 99, 351–361.
Morganti-Kossmann M. C., Rancan M., Stahel P. F., and Kossmann T. (2002) Inflammatory response in acute traumatic brain injury: a double-edged sword. Curr. Opin. Crit. Care 8, 101–105.
Pinhasov A., Mandel S., Torchinsky A., Giladi E., Pittel Z., Goldsweig A. M., et al. (2003) Activity-dependent neuroprotective protein: a novel gene essential for brain formation. Brain Res. Dev. Brain Res. 144, 83–90.
Poggi S. H., Goodwin K., Hill J. M., Brenneman D. E., Tendi E., Schinelli S., et al. (2003) The role of activity-dependent neuroprotective protein in a mouse model of fetal alcohol syndrome. Am. J. Obstet. Gynecol. 189, 790–793.
Poggi S. H., Vink J., Goodwin K., Hill J. M., Brenneman D. E., Pinhasov A., et al. (2002) Differential expression of embryonic and maternal activity-dependent neuroprotective protein during mouse development. Am. J. Obstet. Gynecol. 187, 973–976.
Rall J. M., Matzilevich D. A., and Dash P. K. (2003) Comparative analysis of mRNA levels in the frontal cortex and the hippocampus in the basal state and in response to experimental brain injury. Neuropathol. Appl. Neurobiol. 29, 18–31.
Ralph P. and Nakoinz I. (1977) Antibody-dependent killing of erythrocyte and tumor targets by macrophage-related cell lines: enhancement by PPD and LPS. J. Immunol. 119, 950–954.
Romano J., Beni-Adani L., Nissenbaum O. L., Brenneman D. E., Shohami E., and Gozes I. (2002) A single administration of the peptide NAP induces long-term protective changes against the consequences of head injury: gene Atlas array analysis. J. Mol. Neurosci. 18, 37–45.
Roy N. S., Wang S., Jiang L., Kang J., Benraiss A., Harrison-Restelli C., et al. (2000) In vitro neurogenesis by progenitor cells isolated from the adult human hippocampus. Nat. Med. 6, 271–277.
Smith-Swintosky V. L., Gozes I., Brenemman D., and Plata-Salaman C. R. (2000) Activity dependent neurotrophic factor-9 and NAP promote neurite outgrowth in rat hippocampal and cortical cultures. Society for Neuroscience’s 30th Annual Meeting. New Orleans, LA, November 4–9, 2000. Abstract 317-6. Available at http://sfn.scholarone.com/itin2000/
Tan S. M., Hyland R. H., Al-Shamkhani A., Douglass W. A., Shaw J. M., and Law S. K. (2000) Effect of integrin beta 2 subunit truncations on LFA-1 (CD11a/CD18) and Mac-1 (CD11b/CD18) assembly, surface expression, and function. J. Immunol. 165, 2574–2581.
Yoshimura S., Teramoto T., Whalen M. J., Irizarry M. C., Takagi Y., Qiu J., et al. (2003) FGF-2 regulates neurogenesis and degeneration in the dentate gyrus after traumatic brain injury in mice. J. Clin. Invest. 112, 1202–1210.
Zaltzman R., Beni S. M., Giladi E., Pinhasov A., Steingart R. A., Romano J., et al. (2003) Injections of the neuroprotective peptide NAP to newborn mice attenuate head-injury-related dysfunction in adults. Neuroreport 14, 481–484.
Zamostiano R., Pinhasov A., Gelber E., Steingart R. A., Seroussi E., Giladi E., et al. (2001) Cloning and characterization of the human activity-dependent neuroprotective protein. J. Biol. Chem. 276, 708–714.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaltzman, R., Alexandrovich, A., Beni, S.M. et al. Brain injury-dependent expression of activity-dependent neuroprotective protein. J Mol Neurosci 24, 181–187 (2004). https://doi.org/10.1385/JMN:24:2:181
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:24:2:181