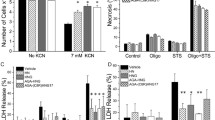
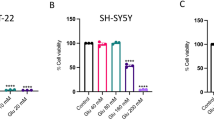
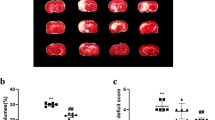
Abstract
An 8-amino-acid peptide, NAPVSIPQ (NAP), was identified as the smallest active element of activity-dependent neuroprotective protein that exhibits potent neuroprotective action. Potential signal transduction pathways include cGMP production and interference with inflammatory mechanisms, tumor necrosis factor-α, and MAC1-related changes. Because of its intrinsic structure, NAP might interact with extracellular proteins and also transverse membranes. NAP-associated protection against oxidative stress, glucose deprivation, and apoptotic mechanisms suggests interference with fundamental processes. This paper identifies p53, a key regulator of cellular apoptosis, as an intracellular target for NAP’s activity.
Similar content being viewed by others
References
Ashur-Fabian O., Giladi E., Furman S., Steingart R. A., Wollman Y., Fridkin M., et al. (2001) Vasoactive intestinal peptide and related molecules induce nitrite accumulation in the extracellular milieu of rat cerebral cortical cultures. Neurosci. Lett. 307, 167–170.
Ashur-Fabian O., Segal-Ruder Y., Skutelsky E., Brenneman D.E., Steingart R. A., Giladi E., and Gozes I. (2003). The effect of the novel peptide NAPVISPQ on the aggregation of the beta-amyloid peptide. Peptides 24, 1413–1423.
Bassan M., Zamostiano R., Davidson A., Pinhasov A., Giladi E., Perl O., et al. (1999) Complete sequence of a novel protein containing a femtomolar-activity-dependent neuroprotective peptide. J. Neurochem. 72, 1283–1293.
Beni-Adani L., Gozes I., Cohen Y., Assaf Y., Steingart R. A., Brenneman D. E., et al. (2001) A peptide derived from activity-dependent neuroprotective protein (ADNP) ameliorates injury response in closed head injured mice. J. Pharmacol. Exp. Ther. 296, 57–63.
Gozes I., and Brenneman D. E. (2000) A new concept in neuroprotection. J. Mol. Neurosci. 14, 61–68.
Gozes I., and Spier A. (2002) Peptides as drug targets in Alzheimer’s disease. Drug Dev. Res. 56, 475–481.
Gozes I., Alcalay R., Giladi E., Pinhasov A., Furman S., and Brenneman D. E. (2002a) NAP accelerates the performance of normal rats in the water maze. J. Mol. Neurosci. 19, 167–170.
Gozes I., Giladi E., Pinhasov A., Bardea A., and Brenneman D. E. (2000) Activity-dependent neurotrophic factor: intranasal administration of femtomolar-acting peptides improve performance in a water maze. J. Pharmacol. Exp. Ther. 293, 1091–1098.
Gozes I., Giladi E., Pinhasov A., Furman S., Romano J., Steingart R. A., Rubinraut S., and Fridkin M. (2002b) Intranasal delivery of bioactive peptides or peptide analogues enhances spatial memory and protects against cholinergic deficits, in The Proceedings of the 44th Oholo Conference: The Blood Brain Barrier Drug Delivery and Brain Pathology, Lustig S., Shapira S., Kobiler O., eds., Plenum Press, New York, NY, pp. 363–370.
Gressens P., Hill J. M., Gozes I., Fridkin M., and Brenneman D. E. (1993) Growth factor function of vasoactive intestinal peptide in whole cultured mouse embryos. Nature 362, 155–158.
Leker R. R., Teichner A., Grigoriadis N., Ovadia H., Brenneman D. E., Fridkin M., et al. (2002) NAP, a femtomolar-acting peptide, protects the brain against ischemic injury by reducing apoptotic death. Stroke 33, 1085–1092.
Newton P., Brenneman D. E., and Gozes I. (2001) 30-Day intranasal toxicity studies of NAP in rats and dogs. J. Mol. Neurosci. Abstr. 16, 61.
Newton P., Spier A. D., and Gozes I (2003) Intravenous and intranasal toxicity and toxicokinetic study of NAPVSIPQ (NAP) in dogs. Neuropeptides (Abstr.) 37, 163.
Offen D., Sherki Y., Melamed E., Fridkin M., Brenneman D. E., and Gozes I. (2000) Vasoactive intestinal peptide (VIP) prevents neurotoxicity in neuronal cultures: relevance to neuroprotection in Parkinson’s disease. Brain Res. 854, 257–262.
Pike C. J., Burdick D., Walencewicz A. J., Glabe C. G., and Cotman C. W. (1993) Neurodegeneration induced by β-amyloid peptides in vitro: The role of peptide assembly state. J. Neurosci. 13, 1676–1687.
Pinhasor A., Mandel S., Torchinsky A., et al., (2003) Activity-dependent neuroprotective protein: a novel gene essential for brain formation. Brain Res. Dev. Brain Res. 144, 83–90.
Poduslo J. F., Curran G. L., Kumar A., Frangione B., and Soto C. (1999) β-Sheet breaker peptide inhibitor of Alzheimer’s amyloidogenesis with increased blood-brain-barrier permeability and resistance to proteolytic degradation in plasma. J. Neurobiol. 39, 371–382.
Romano J., Beni-Adani L., Levy Nissenbaum O., Brenneman D. E., Shohami E., and Gozes I. (2002) A single administration of the peptide NAP induces long-term protective changes against the consequences of head injury: gene atlas array analysis. J. Mol. Neurosci. 18, 137–450.
Seubert P., Vigo-Pelfrey C., Esch F., Lee M., Dovey H., Davis D., et al. (1992) Isolation and quantification of soluble Alzheimer’s beta-peptide from biological fluids. Nature 359, 325–327.
Shoji M., Golde T. E., Ghiso J., Cheung T. T., Estus S., Shaffer L. M., et al. (1992) Production of the Alzheimer amyloid beta protein by normal proteolytic processing. Science 258, 126–129.
Sigalov E., Fridkin M., Brenneman D. E., and Gozes I. (2000) VIP-related protection against iodoacetate toxicity in pheochromocytoma (PC12) cells: a model for ischemic/hypoxic injury. J. Mol. Neurosci. 15, 147–154.
Soto C. (1999) Plaque busters: strategies to inhibit amyloid formation in Alzheimer’s disease. Mol. Med. Today. 5, 343–350.
Soto C., Kascsak R. J., Saborio G. P., Aucouturier P., Wisniewski T., Prelli F., et al. (2000) Reversion of prion protein conformational changes by synthetic β-sheet breaker peptides. Lancet. 355, 192–197.
Spong C. Y., Abebe D. T., Gozes I., Brenneman D. E., and Hill J. M. (2001) Prevention of fetal demise and growth restriction in a mouse model of fetal alcohol syndrome. J. Pharmacol. Exp. Ther. 297, 774–779.
Steingart R. A., Solomon B., Brenneman D. E., Fridkin M., and Gozes I. (2000) VIP and peptides related to activity-dependent neurotrophic factor protect PC12 cells against oxidative stress. J. Mol. Neurosci. 15, 137–145.
Tamagno E., Parola M., Guglielmotto M., Santoro G., Bardini P., Marra L., et al. (2003) Multiple signaling events in amyloid beta-induced, oxidative stressdependent neuronal apoptosis. Free Radic. Biol. Med., 35(1), 45–58.
Velez-Pardo C., Ospina G. G., and Jimenez del Rio M. (2002) Aβ[25–35] peptide and iron promote apoptosis in lymphocytes by an oxidative stress mechanism: involvement of H2O2, caspase-3, NF-κB, p53 and c-Jun. Neurotoxicology 23(3), 351–365.
Wilkemeyer M. F., Chen S. Y., Menkari C. E., Brenneman D. E., Sulik K. K., and Charness M. E. (2003) Differential effects of ethanol antagonism and neuroprotection in peptide fragment NAPVSIPQ prevention of ethanol-induced developmental toxicity. Proc. Natl. Acad. Sci. U. S. A. 100(14), 8543–8548.
Zaltzman R., Beni S. M., Giladi E., Pinhasov A., Steingart R. A., Romano J., et al. (2003) Injections of the neuroprotective peptide NAP to newborn mice attenuate head-injury related dysfunction in adults. Neuroreport 14, 481–484.
Zamostiano R., Pinhasov A., Gelber E., Steingart R. A., Seroussi E., Giladi E., et al. (2001) Cloning and characterization of the human activity-dependent neuroprotective protein (ADNP). J. Biol. Chem. 276, 708–714.
Zemlyak I., Furman S., Brenneman D. E., and Gozes I. (2000) A novel peptide (NAP) prevents death in enriched neuronal cultures. Regul. Pept. 96, 39–43.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gozes, I., Steingart, R.A. & Spier, A.D. NAP mechanisms of neuroprotection. J Mol Neurosci 24, 67–72 (2004). https://doi.org/10.1385/JMN:24:1:067
Issue Date:
DOI: https://doi.org/10.1385/JMN:24:1:067