Abstract
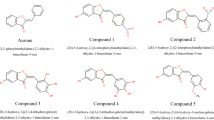
A series of novel 2-aryl benzothiazole derivates substituted with iodine in different positions have been synthesized as amyloid-binding ligands. The affinity of these compounds for synthetic amyloid β (1–40) (Aβ[1–40]) fibrils was determined. Introduction of the iodo group in the position ortho to an amino group increased the binding affinity, whereas the iodination ortho to a hydroxyl group decreased the binding affinity. Selected compounds with high binding affinity and moderate lipophilicity (logP values, 1.65–3.90) were radiolabeled and evaluated in normal mice for brain uptake and clearance. Structure-activity relationship (SAR) studies showed a strong correlation between the lipophilicity of the iodinated compounds and the binding affinity as well as nonspecific binding. As the lipophilicity increased, the affinity for Aβ(1–40) fibrils improved; however, nonspecific binding in mouse brain reflected by low brain clearance also increased with increasing lipophilicity. These results provide important SAR information to guide the development of novel amyloid-binding agents and provide further insights into the molecular interaction between 2-aryl benzothiazole ligands and Aβ fibrils.
Article PDF
Similar content being viewed by others
References
Arnett E. M., Quirk R. P., and Burke J. J. (1970) Weak bases in strong acids. III. Heats of ionization of amines in fluorosulfuric and sulfuric acids. A new general basicity scale. J. Am. Chem. Soc. 92, 1260–1266.
Bard F., Cannon C., Barbour R., Burke R. L., Games D., Grajeda H., et al. (2000) Peripherally administered antibodies against amyloid beta-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nat. Med. 6, 916–919.
Comer J. and Tam K. (2001) Lipophilicity profiles: theory and measurement, in Pharmacokinetic Optimization in Drug Research-Biological, Physicochemical, and Computational Strategies (Testa B., van de Waterbeemd H., Folkers G., and Guy R., eds.), Verlag Helvetica Chimica Acta, Zurich, Switzerland, pp. 275–304.
DeMattos R. B., Bales K. R., Cummins D. J., Dodart J. C., Paul S. M., and Holtzman D. M. (2001) Peripheral anti-A beta antibody alters CNS and plasma A beta clearance and decreases brain A beta burden in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 98, 8850–8855.
Dishino D. D., Welch M. J., Kilbourn M. R., and Raichle M. E. (1983) Relationship between lipophilicity and brain extraction of C-11-labeled radiopharmaceuticals. J. Nucl. Med. 24, 1030–1038.
Engler H., Nordberg A., Blomqvist G., Bergström M., Estrada S., Barletta J., et al. (2002) First human study with a benzothiazole amyloid-imaging agent in Alzheimer’s disease and control sujects. J. Neurobiol. Aging 23, S149.
Hansch C. and Leo A. (1979) Substituent Constants for Correlation Analysis in Chemistry and Biology. John Wiley & Sons, New York.
Klunk W. E., Bacskai B. J., Mathis C. A., Kajdasz S. T., McLellan M. E., Frosch M. P., et al. (2002) Imaging A plaques in living transgenic mice with multiphoton microscopy and methoxy-X04, a systemically administered Congo red derivative. J. Neurophathol. Exp. Neurol. 61, 797–805.
Klunk W. E., Debnath M. L., and Pettegrew J. W. (1994) Development of small molecule probes for the betaamyloid protein of Alzheimer’s disease. Neurobiol. Aging 15, 691–698.
Klunk W. E., Wang Y., Huang G.-F., Debnah M. L., Holt D., Shao L., et al. (2003) The binding of BTA-1 to postmortem brain homogenates is dominated by the amyloid component. J. Neurosci. 23, 2086–2092.
Klunk W. E., Wang Y. M., Hung G. F., Debnah M. L., Holt D. P., and Mathis C. A. (2001) Uncharged thioflavin-T derivative bind to amyloid-beta protein with high affinity and readily enter the brain. Life Sci. 69, 1471–1484.
Massat A., Cosse-Barbi A., and Doucet J. P. (1989) Influence de la structure des cetones et de l’acidite des phenols sur leur mode de complexation. I. Comportement global et échelles d’acidité et de basicité. J. Mol. Structure 212, 13–35.
Mathis C. A., Bacskai B. J., Kajdasz S. T., McLellan M. E., Frosch M. P., Hyman B. T., et al. (2002) A lipophilic thioflavin-T derivative for positron emission tomography (PET) imaging of amyloid in brain. Bioorg. Med. Chem. Lett. 12, 295–298.
Olson R. E., Copeland R. A., and Seiffert D. (2001) Progress towards testing the amyloid hypothesis: inhibitors of APP processing. Curr. Opin. Drug Discov. Devel. 4, 390–401.
Schenk D. B., Seubert P., Lieberburg I., and Wallace J. (2000) Beta-peptide immunization: a possible new treatment for Alzheimer disease. Arch. Neurol. 57, 934–936.
Selkoe D. J. (2000) Imaging Alzheimer’s amyloid. Nat. Biotechnol. 18, 823–824.
Selkoe D. J. (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol. Rev. 81, 741–766.
Skovronsky D. M., Zhang B., Kung M. P., Kung H. F., Trojanowski J. Q., and Lee V. M. (2000) In vivo detection of amyloid plaques in a mouse of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 97, 7609–7614.
Styren S. D., Hamilton R. L., Styren G. C., and Klunk W. E. (2000) X-34, a fluorescent derivative of Congo red: a novel histochemical stain for Alzheimer’s disease pathology. J. Histochem. Cytochem. 48, 1223–1232.
Wang Y., Mathis C. A., Huang G. F., Debnah M. L., Holt D. P., and Klunk W. E. (2002a) Synthesis and 11C-labelling of (E,E)-1-(3′,4′-dihydroxystyryl)-4-(3′-methoxy-4′-hydroxystyryl)benzene for PET imaging of amyloid deposits. J. Labelled Compounds Radiopharmaceuticals 45, 647–664.
Wang Y. M., Klunk W. E., Huang G. F., Debnath M. L., Holt D. P., and Mathis C. A. (2002b) Synthesis and evaluation of 2-(3′-iodo-4′-aminophenyl)-6-hydroxybenzothiazole for in vivo quantitation of amyloid deposits in Alzheimer’s disease. J. Mol. Neurosci. 19, 11–16.
Wengenack T. M., Curran G. L., and Poduslo J. F. (2000) Targeting Alzheimer amyloid plaques in vivo. Nat. Biotechnol. 18, 868–872.
Zhuang Z. P., Kung M. P., Hou C., Skovronsky D. M., Gur T. L., Plossl K., et al. (2001b) Radioiodinated styryl-benzenes and thioflavins as probes for amyloid aggregates. J. Med. Chem. 44, 1905–1914.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Mathis, C.A., Huang, GF. et al. Effects of lipophilicity on the affinity and nonspecific binding of iodinated benzothiazole derivatives. J Mol Neurosci 20, 255–260 (2003). https://doi.org/10.1385/JMN:20:3:255
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:20:3:255