Abstract
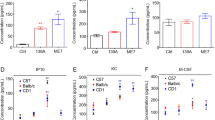
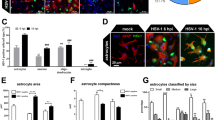
Scrapie is the archetypal unconventional slow infection disease. It has been shown that hamsters injected intracerebrally with scrapie strains 139H or 263K show extensive astrocytosis and that the induced reactive astrocytes produce a variety of factors that can affect brain function. Acidic fibroblast growth factor (aFGF) belongs to a family of growth factors that show a high affinity for heparin sulfate proteoglycans. In the current study, we have used immunohistochemistry to investigate the distribution of aFGF in scrapie-infected brain; we observed a low level of aFGF immunoreactivity (ir-aFGF) in ependymal cells and in a few neurons in the hypothalamus of control hamsters. In contrast, in scrapie-infected hamsters, there was an increase of ir-aFGF in a number of cell types, including neurons, pericytes, astrocytes, and ependymal cells. In 139H-infected hamsters, ir-aFGF staining in astrocytes, neurons and neuropil areas of the cortex, hippocampus, thalamus, and hypothalamus was greater than the staining in control animals. For 263K animals, astrocytic ir-aFGF staining was significantly greater than in either control or 139H-infected hamsters in the following regions: cortex, putamen, corpus callosum, thalamus, hypothalamus, fimbria, hippocampus, subependymal areas, and amygdala. In addition, there was a significant increase in neuronal ir-aFGF in the CA1 hippocampal area and in the amygdala. Our results suggest that neurons and astrocytes can produce and/or absorb aFGF during scrapie infection. These findings indicate that aFGF might play an important role in neuronal protection and in astrocytosis in scrapie-infected hamsters.
Similar content being viewed by others
References
Barrow P. A., Holmgren C. D., Tapper A. J., and Jefferys G. R. (1999) Intrinsic physiological and morphological properties of principal cells of the hippocampus and neocortex in hamsters infected with scrapie. Neurobiol. Dis. 6, 406–423.
Campbell I. L., Eddleston M., Kemper P., Oldstone M. B. A., and Hobbs M. V. (1994) Activation of cerebral cytokine gene expression and its correlation with onset of reactive astrocyte and acute-phase response gene expression in scrapie. J. Virol. 68(4), 2383–2387.
Carp R. I., Kim Y. S., and Callahan S. M. (1990) Pancreatic lesions and hypoglycemia-hyperinsulinemia in scrapie-injected hamsters. J. Infect. Dis. 161, 462–466.
Carp R. I., Ye X., Kascsak R. J., and Rubenstein R. (1994) The nature of the scrapie agent: biological characteristics of scrapie in different scrapie strain-host combinations, in Slow Infections of the Central Nervous System, vol. 724 (Bjornsson J., Carp R. I., Löve A., and Wisniewski H. W., eds.), Annals of New York Academy of Sciences, New York, pp. 221–234.
Caruelle D., Groux-Muscatelli B., Gaudric A., Sestier C., Coscas G., Caruelle J. P., and Barritault D. (1989) Immunological study of acidic fibroblast growth factor (aFGF) distribution in the eye. J. Cell Biochem. 39(2), 117–128.
Choi S. I., Ju W. K., Choi E. K., Kim J., Lea H. Z. Carp R. I., Wisniewski H. M., and Kim Y. S. (1998) Mitochondrial dysfunction induced by oxidative stress in the brains of hamsters infected with the 263K scrapie agent. Acta Neuropathol. 96, 279–286.
Dickinson A. G. (1976) Scrapie in sheep and goats, in Slow Virus Diseases in Animals and Man (Kimberlin R. H., ed.), North-Holland, Amsterdam, pp. 210–243.
Diedrich J. F., Bendheim P. E., Kim Y. S., Carp R. I., and Haase A. T. (1991) Scrapie associated prion protein accumulates in astrocytes during scrapie infection. Proc. Natl. Acad. Sci. USA 88, 375–379.
Forloni G., Angeretti N., Chiesa R., Monzani E., Salmona M., Bugiani O., and Tagliavini F. (1993) Neurotoxicity of a prion protein fragment. Nature 362, 543–546.
Forloni G., Bo R. D., Angeretti N., Chiesa R., Smiroldo S., Doni R., Ghibaudi E., et al. (1994) A neurotoxic prion protein fragment induces rat astroglial proliferation and hypertrophy. Eur. J. Neurosci. 6, 1415–1422.
Fraser J. R., Brown J., Bruce M. E., and Jeffrey M. (1997) Scrapie-induced neuron loss is reduced by treatment with basic fibroblast growth factor. Neuroreport 8(9–10), 2405–2409.
Hafiz F. B. and Brown D. R. (2000) A model for the mechanism of astrogliosis in prion disease. MCN 16, 221–232.
Hanai K., Oomura Y., Kai Y., Nishikawa K., Shimizu N., Morita H., and Plata-Salamán C. R. (1989) Central action of acidic fibroblast growth factor in feeding regulation. Am. J. Physiol. 256, R217-R223.
Iwasahi Y., Shiojima T., Ikeda K., Tagaya N., Kobayashi T., and Kinoshita M. (1995) Acidic and basic fibroblast growth factors enhance neurite outgrowth in cultured rat spinal cord neurons. Neurol. Res. 17, 70–72.
Jeffrey M., Halliday W. G., Bell J., Johnston A. R., MacLeod N. K., Ingham Sayers A. R., et al. (2000) Synapse loss associated with abnormal PrP precedes neuronal degeneration in the scrapie-infected murine hippocampus. Neuropathol. Appl. Neurobiol. 26(1), 41–54.
Kalaria R. N. (1993) The immunopathology of Alzheimer’s disease and some related disorders. Brain Path. 3, 333–347.
Kascsak R. J., Rubenstein R., and Carp R. I. (1991) Evidence for biological and structural diversity among scrapie strains. Curr. Topic Microbiol. Immunol. 172, 139–152.
Kimberlin R. H. and Walker C. A. (1977) Characteristic of a short incubation model of scrapie in the golden hamster. J. Gen. Virol. 34, 295–304.
Kimberlin R. H. and Walker C. A. (1986) Pathogenesis of scrapie (strain 263K) in hamsters infected intracerebrally, intraperitoneally or intraocularly. J. Gen. Virol. 67, 255–263.
Kimberlin R. H., Walker C. A., and Fraser H. (1989) The genomic identity of different strains of mouse scrapie is expressed in hamsters and preserved on reisolation in mice. J. Gen. Virol. 70, 2017–2025.
Kolpakoca E., Frengen E., Stokke T., and Olsnes S. (2000) Organization, chromosomal localization and promoter analysis of the gene encoding human acidic fibroblast growth factor intracellular binding protein. Biochem. J. 352, 629–635.
Li A.-J., Oomura Y., Sasaki K., Suzuki K., Tooyama I., Hanai K., et al. (1998) A single pre-training glucose injection induces memory facilitation in rodents performing various tasks: contribution of acidic fibroblast growth factor. Neuroscience 85(3), 785–794.
McKeehan W. L., Wang F., and Kan M. (1998) The heparan sulfate-fibroblast growth factor family: diversity of structure and function. Prog. Nucleic Acids Res. Mol. Biol. 59, 135–176.
Mehta V. B., Connors L., Wang H. C., and Chui I. M. (1998) Fibroblast variants nonresponsive to fibroblast growth factor 1 are detective in its nuclear translocation. J. Biol. Chem. 273, 4197–4205.
Oomura Y., Sasaki K., Suzuki K., Muto T., Li A.-J., Ogita Z.-I, et al. (1992) A new brain glucosensor and its physiological significance. Am. J. Clin. Nutr. 55, 278S-282S.
Pettmann B., Weibel M., Sensenbrenner M., and Labourdette G. (1985) Purification of two astroglial growth factors from bovine brain. FEBS Lett. 189, 102–108.
Prusiner S. B. (1982) Novel proteinaceous infectious particles cause scrapie. Science 216, 136–144.
Prusiner S. B. (1991) Molecular biology of prion diseases. Science 252, 1515–1522.
Raeber A. J., Race R. E., Brandner S., Priola S. A., Sailer A., Bessen R. A., et al. (1997) Astrocyte-specific expression of hamster prion protein (PrP) renders PrP knockout mice susceptible to hamster scrapie. EMBO J. 16(20), 6057–6065.
Saborlo G. P., Permanne B., and Soto C. (2001) Sensitive detection of pathological prion protein by cyclic amplification of protein misfolding. Nature 411, 810–813.
Sasaki K., Oomura Y., Suzuki K., Hanai K., and Yagi H. (1992) Acidic fibroblast growth factor prevents death of hippocampal CA1 pyramidal cells following ischemia. Neurochem. Int. 21, 397–402.
Sasaki K., Oomura Y., Figorov A., and Yagi H. (1994) Acidic fibroblast growth factor facilitates generation of long-term potentiation in rat hippocampal slices. Brain Res. Bull. 33, 505–511.
Sasaki K., Tooyama I., Li A.-J., Oomura Y., and Kimura H. (1999) Effects of an acidic fibroblast growth factor fragment analog on learning and memory and on medial septum cholonergic neurons in senescence-accelerated mice. Neuroscience 92(4), 1287–1294.
Scott J. R. and Fraser H. (1984) Degenerative hippocampal pathology in mice infected with scrapie. Acta Neuropathol. 65, 62–68.
Smallwood P. M., Munoz-Sanjuan I., Tong P., Macke J. P., Hendry S. H., Gilbert D. J., et al. (1996) Fibroblast growth factor (FGF) homologous factors: new members of the FGF family implicated in nervous system development. Proc. Natl. Acad. Sci. USA 93, 9850–9857.
Thomas K. A. (1993) Biochemistry and molecular biology of fibroblast growth factors, in Neurotrophic Factors (Fallon J. and Loughlin S., eds.), Academic Press, Orlando, FL., pp. 285–312.
Tooyama I., Hara Y., Tasuhara O., Oomura Y., Sasaki K., Muto T., et al. (1991) Production of antisera to acidic fibroblast growth factor and their application to immunohistochemical studies in the rat brain. Neuroscience 40, 769–779.
Walter M. A., Kurouglu R., Caufield J. B., Vasconez L. O., and Thompson J.A. (1993) Enhanced peripheral nerve regeneration by acidic fibroblast growth factor. Lymphokine Cytokine Res. 12, 135–141.
Williams A., Van Dam A. M., Ritchie D., Eikelenboom P., and Fraser H. (1997) Immunocytochemical appearance of cytokines, prostaglandin E2 and lipocortin-1 in the CNS during the incubation period of murine scrapie correlates with progressive PrP accumulations. Brain Res. 754, 171–180.
Williame A. E., Ryder S., and Blakemore W. F. (1995) Monocyte recruitment into the scrapie-affected brain. Acta Neuropathol. 90, 164–169.
Wu D. K., Maciag T., and DeVellis J. (1988) Regulation of neuroblast proliferation by hormones and growth factors in chemically defined medium. J. Cell Physiol. 136, 367–372.
Ye X., Carp R. I., Kozielski R., and Kozlowski P. (1992) Scrapie-induced increase in basic fibroblast growth factor in the hamster brain. Brain Pathol. 2, 262.
Ye X., Carp R. I., Yu Y., Kozielski R., and Kozlowski P. (1994) Hyperplasia and hypertrophy of B-Cells in the islets of Langerhans in hamsters infected with the 139H strain of scrapie. J. Comp. Pathol. 110, 169–183.
Ye X. and Carp R. I. (1995) The pathological changes in the peripheral organs in scrapie-infected animals. Histol. Histopathol. 10(4), 995–1021.
Ye X., Scallet A. C., Kascsak R. J., and Carp R. I. (1998) Astrocytosis and amyloid deposition in scrapie-infected hamsters. Brain Res. 809, 277–287.
Ye X., Scallet A. C., Kascsak R. J., and Carp R. I. (1999) Astrocytosis and proliferating cell nuclear antigen expression in brains of scrapie-infected hamsters. J. Mol. Neurosci. 11, 253–263.
Yong V. W., Kim M. W., and Kim S. U. (1989) Human glial cells and growth factors, in Myelination and Demyelination: Implications for Multiple Sclerosis (Kim S. U., ed.), Plenum Press, New York, pp. 29–48.
Yoshida K. and Gage F. H. (1991) Fibroblast growth factors stimulate nerve growth factor synthesis and secretion by astrocytes. Brain Res. 538, 118–126.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ye, X., Carp, R.I. Increase of acidic fibroblast growth factor in the brains of hamsters infected with either 263K or 139H strains of scrapie. J Mol Neurosci 18, 179–188 (2002). https://doi.org/10.1385/JMN:18:3:179
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:18:3:179