Abstract
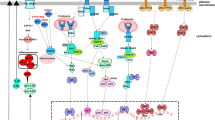
Our laboratory focuses on the signal-transduction basis for mucosal immunity, inflammation, and remodeling, especially in relation to respiratory viral infection. Our approach aims to answer two major questions: (1) What are the mechanisms that control common viral infections? and (2) How can these transient infections cause longterm diseases, such as asthma? Our studies show that antiviral defense depends critically on a specialized network of mucosal epithelial cells and macrophages. When this network is compromised, the host is highly susceptible to infection, but when it is engineered to be broadly hyperresponsive to interferon, the host is markedly resistant to otherwise lethal viral infections. Similar but less effective hyperresponsiveness appears in asthma, suggesting that evolving attempts to improve antiviral defense may instead cause inflammatory disease. Indeed, in susceptible genetic backgrounds, respiratory viruses can also cause a hit-and-run phenomenon that is manifest by the development of a permanent asthmatic phenotype long after the infection has been cleared. This complex phenotype can be segregated into individual traits using pharmacologic, immunologic, and genetic strategies to achieve more precise definition of just how viruses can reprogram host behavior. Evidence of reprogramming is manifest by persistent abnormalities in epithelial cell survival and macrophage activation that when corrected can prevent the development of disease phenotypes. Our results led us to pursue the hypothesis that specific components of the innate immune system may manifest an aberrant antiviral response as a basis for chronic inflammatory diseases and that adjusting this response can improve short-and long-term outcomes after viral infection.
Similar content being viewed by others
References
Murphy KM, Ouyang W, Farrar JD, et al: Signaling and transcription in T helper development. Annu Rev Immunol 2000;18:451–494.
Randolph DA, Stephens R, Carruthers CJL, Chaplin DD: Cooperation between Th1 and Th2 cells in a murine model of eosinophilic airway inflammation. J Clin Invest 1999;104:1021–1029.
Hansen G, Berry G, DeKruyff R, Umetsu D: Allergenspecific Th1 cells fail to counterbalance Th2 cell-induced airay hyperreactivity but cause severe airway inflammation. J Clin Invest 1999;103:175–183.
Holtzman MJ, Look DC, Iademarco MF, Dean DC, Sampath D, Castro M: Asthma; in Jameson, JL (ed): Principles of Molecular Medicine, Totowa, NJ, Humana, 1998, pp 319–327.
Gavett SH, O’Hearn DJ, Li X, Huang S-K, Finkelman FD, Wills-Karp M: Interleukin 12 inhibits antigen-induced airway hyperresponsiveness, inflammation, and Th2 cytokine expression in mice. J Exp Med 1995; 182:1527–1536.
Lack G, Bradley KL, Hamelmann E, et al: Nebulized IFN-γ inhibits the development of secondary allergic responses in mice. J Immunol 1996;157:1432–1439.
Sur S, Wild JS, Choudhury BK, Sur N, Alam R, Klinman DM: Long term prevention of allergic lung inflammation in a mouse model of asthma by CpG oligodeoxynucleotides. J Immunol 1999;162:6284–6293.
Finotto S, Neurath NF, Glickman JN, et al: Development of spontaneous airway changes consistent with human asthma in mice lacking T-bet. Science 2002;295: 336–338.
Kay AB: Allergy and allergic diseases. N Engl J Med 2001;344:30–37.
Ober C, Moffatt MF: Contributing factors to the pathobiology: the genetics of asthma. Clin Chest Med 2000;21:245–261.
Williams CMM, Galli SJ: Mast cells can amplify airway reactivity and features of chronic inflammation in an asthma model in mice. J Exp Med 2000;192:455–462.
Bandeira-Melo C, Herbst A, Weller PF: Eotaxins: contributing to the diversity of eosinophil recruitment and activation. Am J Respir Cell Mol Biol 2001;24:653–657.
Grayson MH, Holtzman MJ: Lessons from allergic rhinitis versus asthma pathogenesis and treatment. Immunol Allergy Clin N Am 2002;22:845–869.
Melhop PD, van de Rijn M, Goldberg AB, et al: Allergen-induced bronchial hyperreactivity and eosinophilic inflammation occur in the absence of IgE in a mouse model of asthma. Proc Natl Acad Sci USA 1997; 94:1344–1349.
Hogan SP, Mould A, Kikutani, H, Ramsay, AJ, Foster PS: Aeroallergen-induced eosinophilic inflammation, lung damage, and airways hyperreactivity in mice can occur independently of IL-4 and allergen-specific immunoglobulins. J Clin Invest 1997;99:1329–1339.
Wills-Karp M, Luyimbazi J, Xu, X, et al: Interleukin-13: central mediator of allergic asthma Science 1998;282: 2258–2261.
Foster PS, Hogan SP, Ramsay AJ, Matthaei KI, Young, IG: IL-5 deficiency abolishes eosinophilia, airways hyperreactivity, and lung damage in a mouse asthma model. J Exp Med 1995;183:195–201.
Corry DB, Folkesson HG, L. WM, et al: Interleukin 4, but not interleukin 5 or eosinophils, is required in a murine model of acute airway hyperreactivity. J Exp Med 1995;183:109–117.
Holtzman MJ, Sampath D, Castro M, Look DC, Jayaraman S: The one-two of T helper cells: does interferon-γ knockout the Th2 hypothesis for asthma? Am J Respir Cell Mol Biol 1996;14:316–318.
Hakonarson H, Bjornsdottir US, Ostermann E, et al: Allelic frequencies and patterns of single-nucleotide polymorphisms in candidate genes for asthma and atopy in Iceland. Am J Respir Crit Care Med 2001;164: 2036–2044.
Drazen JM, Arm JP, Austen KF: Sorting out the cytokines of asthma. J Exp Med 1996;183:1–5.
Nakajima S, Look DC, Roswit, WT, Bragdon MJ, Holtzman MJ: Selective differences in vascular endothelial-vs. airway epithelial-T cell adhesion mechanisms. Am J Physiol 1994;267:L422-L432.
Nakajima S, Roswit WT, Look DC, Holtzman MJ: A hierarchy for integrin expression and adhesiveness among T cell subsets that is linked to TCR gene usage and emphasizes V δ1+ γσ T cell adherence and tissue retention. J Immunol 1995;155:1117–1131.
Taguchi M, Sampath D, Koga T, et al: Patterns for RANTES secretion and intercellular adhesion molecule-1 expression mediate transepithelial T cell traffic based on analyses in vitro and in vivo. J Exp Med 1998;187:1927–1940.
Holtzman MJ, Castro M, Look DC, O’Sullivan M, Walter MJ: Regulation of epithelial-leukocyte interaction and epithelial immune-response genes; in Busse WW, Holgate ST (eds). Asthma and Rhinitis, Cambridge, MA, Blackwell Scientific, pp 784–800.
Look DC, Keller BT, Rapp SR, Holtzman MJ: Selective induction of intercellular adhesion molecule-1 by interferon-γ in human airway epithelial cells. Am J Physiol 1992;263:L79-L87.
Look DC, Pelletier MR, Holtzman MJ: Selective interaction of a subset of interferon-γ response element binding proteins with the intercellular adhesion molecule-1 (ICAM-1) gene promoter controls the pattern of expression on epithelial cells. J Biol Chem 1994;269:8952–8958.
Look DC, Pelletier MR, Tidwell RM, Roswit WT, Holtzman MJ: Statl depends on transcriptional synergy with Spl. J Biol Chem 1995;270:30,264–30,267.
Walter MJ, Look DC, Tidwell RM, Roswit WT, Holtzman MJ: Targeted inhibition of interferon-γ-dependent ICAM-1 expression using dominant-negative Stat1. J Biol Chem 1997;272:28,582–28,589.
Look DC, Roswit WT, Frick AG, et al: Direct suppression of Statl function during adenoviral infection. Immunity 1998;9:871–880.
Koga T, Sardina E, Tidwell RM, Pelletier MR, Look DC, Holtzman MJ: Virus-inducible expression of a host chemokine gene relies on replication-linked mRNA stabilization. Proc Natl Acad Sci USA 1999;96:5680–5685.
Castro M, Block SR, Jenkersen MV, et al: Asthma exacerbation after glucocorticoid withdrawal reflects T cell recruitment to the airway. Am J Respir Crit Care Med 2004;169:842–849.
Graham BS, Perkins MD, Wright PF, Karzon DT: Primary respiratory syncytial virus infection in mice. J Med Virol 1988;26:153–162.
Walter MJ, Kajiwara N, Karanja P, Castro M, Holtzman MJ: IL-12 p40 production by barrier epithelial cells during airway inflammation. J Exp Med 2001;193:339–352.
Walter MJ, Holtzman MJ: Epithelial production of IL-12 p 80 during viral infection and asthma supports an altered paradigm for airway inflammation; in Eissa NT, Huston DP (eds): Lung Biology in Health and Disease: Therapeutic Targets in Airway Inflammation, New York, Marcel Dekker 2003, pp 535–561.
Tyner JW, Uchida Y, Kajiwara N, et al: Chemokine CCL5 signals required for survival during viral infection. J Allergy Clin Immunol 2004;113:S49.
Shornick L, Briner D, Lo M, Holtzman MJ: Role of Statl in defense against respiratory viruses. Proc. Keystone Symposium: Molecular Aspects of Viral Immunity 2001;1:56.
Dupuis S, Jouanguy E, Al-Hajjar S, et al: Impaired response to interferon- α/β and lethal viral disease in human STAT1 deficiency. Nat Genet 2003;33:388–391.
Sampath D, Castro M, Look DC, Holtzman MJ: Constitutive activation of an epithelial signal transducer and activator of transcription (Statl) pathway in asthma. J Clin Invest 1999;103:1353–1361.
Holtzman MJ, Morton JD, Shornick LP, et al.: Immunity, inflammation, and remodeling in the airway epithelial barrier: epithelial-viral-allergic paradigm. Physiol Rev 2002;82:19–46.
Domachowske JB, Rosenberg HF: Respiratory syncytial virus infection: immune response, immunopathogenesis, and treatment. Clin Microbiol Rev 1999;12:298–309.
Sigurs N: Epidemiologic and clinical evidence of a respiratory syncytial virus-reactive airway disease link. Am J Respir Crit Care Med 2001;163:S2-S6.
Walter MJ, Morton JD, Kajiwara N, Agapov E, Holtzman MJ: Viral induction of a chronic asthma phenotype and genetic segregation from the acute response. J Clin Invest 2002;110:165–175.
Holtzman MJ, Fabbri LM, O’Byrne PM, Gold BD, Aizawa H, Walters EH, Alpert SE, Nadel JA: Importance of airway inflammation for hyperresponsiveness induced by ozone in dogs. Am Rev Respir Dis 1983;127:686–690.
Shen Y, Zhu H, Shenk T: Human cytomegalovirus IE1 and IE2 proteins are mutagenic and mediate “hit and run” oncogenic transformation in cooperation with the adenovirus E1A proteins. Proc Natl Acad Sci USA 1997;94:3341–3345.
Nevels M, Tauber B, Spruss T, Wolf H, Dobner T: “Hit and run” transformation by adenovirus oncogenes. J Virol 2001;75:3089–3094.
Morton JD, Alevy Y, Peltz G, Holtzman MJ: Functional genomics of asthma: role of CLCA3 in goblet cell metaplasia but not airway hyperreactivity. J Allergy Clin Immunol 2004;113.
Hogan RJ, Usherwood EJ, Zhong W, et al: Activated antigen-specific CD8+ T cells persist in the lungs following recovery from respiratory virus infections. J Immunol 2001;166:1813–1822.
Hogan RJ, Zhong W, Usherwood EJ, Cookenham T, Roberts AD, Woodland DL: Protection from respiratory virus infections can be mediated by antigen-specific CD4+ T cells that persist in the lungs. J Exp Med 2001;193:981–986.
Coyle AJ, Erard F, Bertrand C, Walti S, Pircher H, Le Gros, G: Virus-specific CD8+ cells can switch to interleukin 5 production and induce airway eosinophilia. J Exp Med 1995;181:1229–1233.
Srikiatkhachorn A, Braciale TJ: Virus-specific CD8+ T lymphocytes downregulate T helper cell type 2 cytokine secretion and pulmonary eosinophila during experimental murine respiratory syncytial virus infection. J Exp Med 1997;186:421–432.
Schwarze J, Cieslewicz G, Joetham A, Ikemura T, Hamelmann E, Gelfand EW: CD8 T cells are essential in the development of respiratory syncytial virus-induced lung eosinophilia and airway hyperresponsiveness. J Immunol 1999;162:4207–4211.
Kim EY, Kim J-I, Morton JD, et al: Glucocorticoids suppress goblet cell metaplasia in natural and experimental asthma. J Allergy Clin Immunol 2004;113:S116.
Ashwell JD, Lu FWM, Vacchio MS: Glucocorticoids in T cell development and function. Annu Rev Immunol 2000;18:309–345.
Horwitz MS, La Cava A, Fine C, Rodriguez E, Ilic A, Sarvetnick N: Pancreatic expression of interferon-g protects mice from lethal coxsackievirus B3 infection and subsequent myocarditis. Nat Med 2000;6:693–697.
Yasukawa H, Yajima T, Duplain H, et al: The suppressor of cytokine signaling-1 (SOCS1) is a novel therapeutic target for enterovirus-induced cardiac injury. J Clin Invest 2003;111:469–478.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holtzman, M.J., Kim, E.Y., Lo, M.S. et al. Defining and adjusting divergent host responses to viral infection. Immunol Res 32, 123–141 (2005). https://doi.org/10.1385/IR:32:1-3:123
Issue Date:
DOI: https://doi.org/10.1385/IR:32:1-3:123