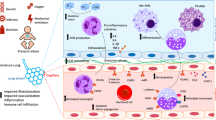
Abstract
Pulmonary neuroendocrine (NE) cells are believed to be the precursor of NE lung carcinomas, including well-differentiated (carcinoids) and moderately/poorly differentiated (atypical carcinoids and small-cell carcinomas, SCLCs) subtypes. In early studies, we determined mechanisms by which NE cell-derived peptides such as bombesin-like peptide (BLP) promote normal fetal lung development. Postnatally, BLP may normally regulate perinatal adaptation of the pulmonary circulation. However, elevated BLP levels in premature infants shortly after birth predict which infants are at high risk for developing bronchopulmonary dysplasia (BPD, chronic lung disease of newborns). An anti-BLP blocking antibody abrogates clinical and pathological evidence of lung injury in two baboon models of BPD. These observations indicate that BLP mediates lung injury in BPD, supporting a role for BLP as pro-inflammatory cytokines. We have directly tested the effects of BLP on eliciting inflammatory cell infiltrates in vivo. Surprisingly, mast cells are the major responding cell population. These data suggest that the diffuse NE system may be a newly recognized component of innate immunity in multiple organ systems. We speculate that overproduction of NE cell-derived peptides such as BLP may be responsible for a variety of chronic inflammatory disorders.
Similar content being viewed by others
References
Anastasi A, Erspamer V, Bucci M. Isolation and structure of bombesin and alytesin, two analogous active peptides from the skin of the European amphibians Bombina and Alytes. Experientia 27:166–169, 1971.
McDonald TJ, Jornvall H, Nilsson G, Vagne M, Ghatei M, Bloom SR, Mutt V. Characterization of a gastrin-releasing peptide from porcine non-antral gastric tissue. Biochem Biophys Res Commun 90:227–233, 1979.
Spindel ER, Chin WW, Price J, Rees LH, Besser GM, Habener JF. Cloning and characterization of cDNAs encoding human gastrin-releasing peptide. Proc Natl Acad Sci USA 81:5699–5703, 1984.
Sausville EA, Lebacq-Verheyden AM, Spindel ER, Cuttitta F, Gazdar AF, Battey JF. Expression of the gastrin-releasing peptide gene in human small cell lung cancer: Evidence for alternative processing resulting in three distinct mRNAs. J Biol Chem 261:2451–2457, 1986.
Lebacq-Verheyden AM, Krystal G, Sartor O, Way J, Battey JF. The prepro gastrin releasing peptide gene is transcribed from two initiation sites in the brain. Mol Endocrinol 2:556–563, 1988.
Sunday ME, Kaplan LM, Motoyama E, Chin WW, Spindel ER. Biology of disease: Gastrin-releasing peptide (mammalian bombesin) gene expression in health and disease. Lab Invest 59:5–24, 1988.
Nagalla SR, Barry BJ, Spindel ER. Cloning of complementary DNAs encoding the amphibian bombesin-like peptides Phe8 and Leu8 phyllolitorin from Phyllomedusa sauvagei: potential role of U to C RNA editing in generating neuropeptide diversity. Mol Endocrinol 8:943–951, 1994.
Erspamer V. Amphibian skin peptides in mammals—looking ahead. Trends Neurosci 6:200–201, 1983.
Minna J. Bombesin receptor gene cloned. Science 249:1377, 1990.
Battey J, Wada E. Two distinct receptor subtypes for mammalian bombesin-like peptides. Trends Neurosci 14:524–528, 1991.
Battey JF, Way JM, Corjay MH, et al. Molecular cloning of the bombesin/gastrin-releasing peptide receptor from Swiss 3T3 cells. Proc Natl Acad Sci USA 88:395–399, 1991.
Spindel ER, Giladi E, Brehm P, Goodman RH, Segerson TP. Cloning and functional characterization of a complementary DNA encoding the murine fibroblast bombesin/gastrin-releasing peptide receptor. Mol Endocrinol 4:1956–1963, 1990.
Lach E, Haddad EB, Gies JP. Contractile effect of bombesin on guinea pig lung in vitro: involvement of gastrin-releasing peptide-preferring receptors. Am J Physiol 264:L80-L86, 1993.
Gorbulev V, Akhundova A, Buchner H, Fahrenholz F. Molecular cloning of a new bombesin receptor subtype expressed in uterus during pregnancy. Eur J Biochem 208:405–410, 1992.
Fathi Z, Corjay MH, Shapira H, et al. BRS-3: A novel bombesin receptor subtype selectively expressed in testis and lung carcinoma cells. J Biol Chem 268:5979–5984, 1993.
Shan L, Emanuel RL, Dewald D, et al. Bombesin-like peptide (BLP) receptor gene expression, regulation, and function in fetal murine lung. Am J Physiol Lung Cell Mol Physiol 286:L165–173, 2004.
Fischer JB, Schonbrunn A. The bombesin receptor is coupled to a guanine nucleotide-binding protein which is insensitive to pertussis and cholera toxins. J Biol Chem 263:2808–2816, 1988.
Zachary I, Rozengurt E. Focal adhesion kinase (p125FAK): A point of convergence in the action of neuropeptides, integrins, and oncogenes. Cell 71:891–894, 1992.
Rozengurt E. Neuropeptides as cellular growth factors: role of multiple signalling pathways. Eur J Clin Invest 21:123–134, 1991.
Wada E, Battey J, Wray S. Bombesin receptor gene expression in rat embryos: transient GRP-R gene expression in the posterior pituitary. Mol Cell Neurosci 4:13–24, 1993.
King KA, Torday JS, Sunday ME. Bombesin and [leu8]phyllolitorin promote fetal mouse lung branching morphogenesis via a specific receptor-mediated mechanism. Proc Natl Acad Sci USA 92:4357–4361, 1995.
Wang D, Yeger H, Cutz E. Expression of gastrin releasing peptide receptor gene in developing lung. Am J Respir Cell Mol Biol 14:409–416, 1996.
Li K, Nagalla SR, Spindel ER. A rhesus monkey model to characterize the role of gastrin-releasing peptide (GRP) in lung development. J Clin Invest 94:1605–1615, 1994.
Brimhall BB, Sikorski KA, Torday J, Shahsafaei A, Haley KJ, Sunday ME. Syntaxin 1A is transiently expressed in fetal lung mesenchymal cells: potential developmental roles. Am J Physiol Lung Cell Mol Physiol 277:L401-L411, 1999.
Sunday ME, Hua J, Dai HB, Nusrat A, Torday JS. Bombesin increases fetal lung growth and maturation in utero and in organ culture. Am J Respir Cell Mol Biol 3:199–205, 1990.
Wharton J, Polak JM, Bloom SR, et al. Bombesin-like immunoreactivity in the lung. Nature 273:769–770, 1978.
Minna JD, Cuttitta F, Battey JF, et al. Gastrin-releasing peptide and other autocrine growth factors in lung cancer: pathogenetic and treatment implications. In: DeVita VT, Hellman S, Rosenberg SA, eds. Important Advances in Oncology, Philadelphia, PA: Lippincott, 1988; 55–64.
Siegfried JM, Guentert PJ, Gaither AL. Effects of bombesin and gastrin-releasing peptide on human bronchial epithelial cells from a series of donors: Individual variation and modulation by bombesin analogs. Anat Rec 236:241–247, 1993.
Rozengurt E, Sinnett-Smith J. Early signals underlying the induction of the c-fos and c-myc genes in quiescent fibroblasts: studies with bombesin and other growth factors. Prog Nucleic Acid Res Mol Biol 35:261–295, 1988.
Cuttitta F, Carney DN, Mulshine J, et al. Bombesin-like peptides can function as autocrine growth factors in human small cell cancer. Nature 316:823–826, 1985.
Impicciatore M, Bertaccini G. The bronchoconstrictor action of the tetradecapeptide bombesin in the guinea-pig. J Pharm Pharmacol 25:872–875, 1973.
Erspamer GF, Mazzanti G, Farruggia G, Nakajima T, Yanaihara N. Parallel bioassay of litorin and phyllolitorins on smooth muscle preparations. Peptides 5:765–768, 1984.
Sunday ME, Hua J, Reyes B, Masui H, Torday JS. Anti-bombesin antibodies modulate fetal mouse lung growth and maturation in utero and in organ cultures. Anat Rec 236:25–32, 1993.
Kim JS, McKinnis VS, White SR. Migration of guinea pig airway epithelial cells in response to bombesin analogues. Am J Respir Cell Mol Biol 16:259–266, 1997.
Ruff M, Schiffmann E, Terranova V, Pert CB. Neuropeptides are chemoattractants for human tumor cells and monocytes: A possible mechanism for metastasis. Clin Immunol Immunopathol 37:387–396, 1985.
De la Fuente M, Del Rio M, Ferrandez MD, Hernanz A. Modulation of phagocytic function in murine peritoneal macrophages by bombesin, gastrin-releasing peptide and neuromedin C. Immunology 73:205–211, 1991.
Meloni F, Ballabio P, Bianchi L, et al. Bombesin enhances monocyte and macrophage activities: possible role in the modulation of local pulmonary defenses in chronic bronchitis. Respiration 63:28–34, 1996.
Meloni F, Bertoletti R, Corsico A, Di Fazio P, Cecchettin M, Gialdroni-Grassi G. Bombesin/gastrin releasing peptide levels of peripheral mononuclear cells, monocytes and alveolar macrophages in chronic bronchitis. Int J Tissue Reactions 14:195–201, 1992.
Yule KA, White SR. Migration of 3T3 and lung fibroblasts in response to calcitonin gene-related peptide and bombesin. Exp Lung Res 25:261–273, 1999.
Kelley MJ, Linnoila RI, Avis IL, et al. Antitumor activity of a monoclonal antibody directed against gastrin-releasing peptide in patients with small cell lung cancer. Chest 112:256–261, 1997.
Wang LH, Coy DH, Taylor JE, et al. Desmethionine alkylamide bombesin analogues: a new class of bombesin receptor antagonists with potent antisecretory activity in pancreatic acini and antimitotic activity in Swiss 3T3 cells. Biochem 29:616–622, 1990.
Wang LH, Coy DH, Taylor JE, et al. Desmethionine alkylamide bombesin analogues: a new class of bombesin. Biochem 29:616–622, 1990.
King KA, Hua J, Torday JS, et al. CD10/Neutral endopeptidase regulates fetal lung growth and maturation in utero by potentiating endogenous bombesin-like peptides. J Clin Invest 91:1969–1973, 1993.
Sunday ME, Hua J, Torday J, Reyes B, Shipp MA. CD10/neutral endopeptidase 24.11 in developing human fetal lung: patterns of expression and modulation of peptide-mediated proliferation. J Clin Invest 90:2517–2525, 1992.
Emanuel RL, Torday JS, Mu Q, Asokananthan N, Sikorski KA, Sunday ME. Bombesin-like peptides and receptors in normal fetal baboon lung: roles in lung growth and maturation. Am J Physiol 277:L1003-L1017, 1999.
Aguayo SM, Kane MA, King TE, Schwarz MI, Grauer L, Miller YE. Increased levels of bombesin-like peptides in the lower respiratory tract of asymptomatic cigarette smokers. J Clin Invest 84:1105–1113, 1989.
Fraslon C, Bourbon JR. Comparison of effects of epidermal and insulin-like growth factors, gastrin releasing peptide and retinoic acid on fetal lung cell growth and maturation in vitro. Biochim Biophysica Acta 1123:65–75, 1992.
Koh S, Yamamoto A, Inoue A, et al. Immunoelectron microscopic localization of the HPC-1 antigen in rat cerebellum. J Neurocytol 22:995–1005, 1993.
Masaki R, Yamamoto A, Akagawa K, Tashiro Y. Important roles of the C-terminal portion of HPC-1/syntaxin 1A in membrane anchoring and intracellular localization. J Biochem 124:311–318, 1998.
Inoue A, Akagawa K. Neuron-specific expression of a membrane protein, HPC-1: tissue distribution, and cellular and subcellular localization of immunoreactivity and mRNA. Molec Brain Res 19:121–128, 1993.
Sesack SR, Snyder CL. Cellular and subcellular localization of syntaxin-like immunore-activity in the rat striatum and cortex. Neuroscience 67:993–1007, 1995.
Akagawa K, Barnstable CJ. Identification and characterization of cell types in monolayer cultures of rat retina using monoclonal antibodies. Brain Res 383:110–120, 1986.
Kong Y, Glickman J, Subramaniam M, et al. Functional diversity of notch family genes in fetal lung development. Am J Physiol Lung Cell Mol Physiol 286:L1075–1083, 2004.
Jan YN, Jan LY. HLH proteins, fly neurogenesis, and vertebrate myogenesis. Cell 75:827–830, 1993.
Wakamatsu Y, Maynard TM, Weston JA. Fate determination of neural crest cells by NOTCH-mediated lateral inhibition and asymmetrical cell division during gangliogenesis. Development 127:2811–2821, 2000.
Campos-Ortega JA. Genetic and molecular bases of neurogenesis in Drosophila melanogaster. Annu Rev Neurosci 14:399–420, 1991.
Isaac DD, Andrew DJ. Tubulogenesis in Drosophila: a requirement for the trachealess gene product. Genes & Develop 10:103–117, 1996.
Kent G, Iles R, Bear CE, et al. Lung disease in mice with cystic fibrosis. J Clin Invest 100:3060–3069, 1997.
Artavanis-Tsakonas S, Delidakis C, Fehon RG. The notch locus and the cell biology of neuroblast segregation. Annu Rev Cell Biol 7:427–452, 1991.
Fehon RG, Johansen K, Rebay I, Artavanis-Tsakonas S. Complex cellular and subcellular regulation of Notch expression during embryonic and imaginal development of Drosophila: implications for Notch function. J Cell Biol 113:657–669, 1991.
Ellisen LW, Bird J, West DC, et al. TAN-1, the human homolog of the Drosophila Notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. Cell 66:649–661, 1991.
Schroeder T, Just U. Notch signalling via RBP-J promotes myeloid differentiation. EMBO J 19:2558–2568, 2000.
Anderson AC, Robey EA, Huang YH. Notch signaling in lymphocyte development. Curr Opin Genetics & Develop 11:554–560, 2001.
Karanu FN, Murdoch B, Miyabayashi T, et al. Human homologues of Delta-1 and Delta-4 function as mitogenic regulators of primitive human hematopoietic cells. Blood 97:1960–1967, 2001.
Pui JC, Allman D, Xu L, et al. Notch 1 expression in early lymphopoiesis influences B versus T lineage determination. Immunity 11:299–308, 1999.
Aster J, Pear W, Hasserjian R, et al. Functional analysis of the TAN-1 gene, a human homolog of Drosophila notch. Cold Spring Harbor Symp Quant Biol 59:125–136, 1994.
Rangarajan A, Talora C, Okuyama R, et al. Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J 20:3427–3436, 2001.
Jaleco AC, Neves H, Hooijberg E, et al. Differential effects of Notch ligands Delta-1 and Jagged-1 in human lymphoid differentiation. J Exp Med 194:991–1002, 2001.
Gridley T. Notch signaling during vascular development. Proc Natl Acad Sci USA 98:5377–5378, 2001.
Austin CP, Feldman DE, Ida JA, Jr., Cepko CL. Vertebrate retinal ganglion cells are selected from competent progenitors by the action of Notch. Development 121:3637–3650, 1995.
Borges M, Linnoila RI, van de Velde HJ, et al. An achaete-scute homologue essential for neuroendocrine differentiation in the lung. Nature 386:852–855, 1997.
Han RNN, Mawdsley C, Souza P, Tanswell AK, Post M. Platelet-derived growth factors and growth-related genes in rat lung. III. Immunolocalization during fetal development. Pediatr Res 31:323–329, 1992.
Bostrom H, Willetts K, Pekny M, et al. PDGF-A signaling is a critical event in lung alveolar myofibroblast development and alveogenesis. Cell 85:863–873, 1996.
Souza P, Sedlackova L, Kuliszewski M, et al. Antisense oligodeoxynucleotides targeting PDGF-B mRNA inhibit cell proliferation during embryonic rat lung development. Development 120:2163–2173, 1994.
Souza P, Tanswell AK, Post M. Different roles for PDGF-α and -β receptors in embryonic lung development. Am J Respir Cell Mol Biol 15:551–562, 1996.
Plopper CG, St. George JA, Read LC, et al. Acceleration of alveolar type II cell differentiation in fetal rhesus monkey lung by administration of EGF. Am J Physiol Lung Cell Mol Physiol 262:L313-L321, 1992.
Sen N, Cake MH. Enhancement of disaturated phosphatidylcholine synthesis by epidermal growth factor in cultured fetal lung cells involves a fibroblast-epithelial cell interaction. Am J Respir Cell Mol Biol 5:337–343, 1991.
Raaberg L, Nex E, Buckley S, Luo W, Snead ML, Warburton D. Epidermal growth factor transcription, translation, and signal transduction by rat type II pneumocytes in culture. Am J Resp Cell Mol Biol 6:44–49, 1992.
Sunday ME. Bioactive peptides and lung development. In: Gaultier C, Bourbon JR, Post M, eds. Lung Development, New York, Oxford: Oxford University Press, 1999:304–326.
Johnson DE, Wobken JD, Landrum BG. Changes in bombesin, calcitonin and serotonin immunoreactive pulmonary neuroendocrine cells in cystic fibrosis and following prolonged mechanical ventilation. Am Rev Respir Dis 137:123–131, 1988.
Aguayo SM, King TE, Waldron JA, Sherritt KM, Kane MA, Miller YE. Increased pulmonary neuroendocrine cells with bombesin-like immunoreactivity in adult patients with eosinophilic granuloma. J Clin Invest 86:838–844, 1990.
Bousbaa H, Fleury-Feith J. Effects of a longstanding challenge on pulmonary neuroendocrine cells of actively sensitized guinea pigs. Am Rev Respir Dis 144:714–717, 1991.
Sunday ME, Willett CG. Induction and spontaneous regression of intense pulmonary neuroendocrine cell differentiation in a model of preneoplastic lung injury. Cancer Res 52(suppl):2677S-2686S, 1992.
Sunday ME, Willett CG, Patidar K, Graham SA, Kelly D. Modulation of oncogene and tumor suppressor gene expression in a hamster model of chronic lung injury with varying degrees of pulmonary neuroendocrine cell hyperplasia. Lab Invest 70:875–888, 1994.
Haley KJ, Patidar K, Zhang F, Emanuel RL, Sunday ME. Tumor necrosis factor induces neuroendocrine differentiation in small cell lung carcinoma cell lines. Am J Physiol Lung Cell Mol Physiol 275:L311-L321, 1998.
Mabry M, Nakagawa T, Nelkin BD, et al. v-Ha-ras oncogene insertion: A model for tumor progression of human small cell lung cancer. Proc Natl Acad Sci USA 85:6523–6527, 1988.
Sunday ME, Haley KJ, Sikorski K, et al. Calcitonin driven v-Ha-ras induces multilineage pulmonary epithelial hyperplasias and neoplasms. Oncogene 18:4336–4347, 1999.
Johnston D, Hatzis D, Sunday ME. Expression of v-Ha-ras driven by the calcitonin/calcitonin gene-related peptide promoter: a novel transgenic murine model for medullary thyroid carcinoma. Oncogene 16:167–177, 1998.
Mabry M, Nakagawa T, Baylin S, Pettengill O, Sorenson G, Nelkin B. Insertion of the v-Ha-ras oncogene induces differentiation of calcitonin-producing human small cell lung cancer. J Clin Invest 84:194–199, 1989.
Slebos RJC, Kibbelaar RE, Dalesio O, et al. K-ras oncogene activation as a prognostic marker in adenocarcinoma of the lung. N Engl J Med 323:561–565, 1990.
Sunday ME, Willett CG, Graham SA, Oreffo VIC, Linnoila RI, Witschi H. Histochemical characterization of non-neuroendocrine tumors and neuroendocrine cell hyperplasia induced in hamster lung by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone with or without hyperoxia. Am J Pathol 147:740–752, 1995.
Johnson DE, Anderson WR, Burke BA. Pulmonary neuroendocrine cells in pediatric lung disease: alterations in airway structure in infants with bronchopulmonary dysplasia. Anat Rec 236:115–119, 1993.
Sunday ME. Neuropeptides and lung development. In: McDonald JA, ed. Lung Growth and Development, New York: Dekker, 1997;401–494.
Youngson C, Nurse C, Yeger H, Cutz E. Oxygen sensing in airway chemoreceptors. Nature 365:153–155, 1993.
Northway WH, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline membrane disease. N Engl J Med 276:357–368, 1967.
Johnson DE, Lock JE, Elde RP, Thompson TR. Pulmonary neuroendocrine cells in hyaline membrane disease and bronchopulmonary dysplasia. Pediatr Res 16:446–454, 1982.
Parad RB, Berger TM. Chronic lung disease. In: Cloherty JP, Stark AR, eds. Manual of neonatal care. Philadelphia-New York: Lippincott-Raven, 1998;378–388.
Hansen T, Corbet A. Chronic lung disease—bronchopulmonary dysplasia. In: Taeusch HW, Ballard RA, Avery ME, eds. Diseases of the newborn. Toronto: W.B. Saunders Co., 1991;519–526.
Abman SH, Groothius JR. Pathophysiology and treatment of bronchopulmonary dysplasia. Current issues. Pediatr Clin North Am 41:277–315, 1994.
Ireys HT, Anderson GF, Shaffer TJ, Neff JM. Expenditures for care of children with chronic illnesses enrolled in the Washington State Medicaid program, fiscal year 1993. Pediatrics 100:197–204, 1997.
Jobe AH. Pulmonary surfactant therapy. N Engl J Med 328:861–868, 1993.
Jobe AH, Mitchell BR, Gunkel H. Beneficial effects of the combined use of prenatal corticosteroids and postnatal surfactant on preterm infants. Am J Obstet Gynecol 168:508–513, 1993.
Parker RA, Lindstrom DP, Cotton RB. Improved survival accounts for most, but not all of the increase in bronchopulmonary dysplasia. Pediatrics 90:663–668, 1992.
Avery ME, Tooley WH, Keller JB, et al. Is chronic lung disease in low birth weight infants preventable? A survey of eight centers. Pediatr 79:26–30, 1987.
Watterberg KL, Derners LM, Scott SM, Murphy S. Chorioamnionitis and early lung inflammation in infants in whom bronchopulmonary dysplasia develops. Pediatr 97:210–215, 1996.
Konishi M, Fujiwara T, Naito T, et al. Surfactant replacement therapy in neonatal respiratory distress syndrome. Eur J Pharmacol 147:20–25, 1988.
Feinberg E, Richardson DK, Als H, Sell E, Parad RB. Late pulmonary outcomes poorly predicted by early risk factors in very low birth weight infants. Pediatr Res 39:263A, 1997.
Escobedo MB, Hilliard JL, Smith F, et al. A baboon model of bronchopulmonary dysplasia: I. clinical features. Exp Mol Pathol 37:323–334, 1982.
Coalson JJ, Kuehl TJ, Escobedo MB, et al. A baboon model of bronchopulmonary dysplasia: II. pathologic features. Exp Mol Pathol 37:335–350, 1982.
Coalson JJ, Winter VT, Siler-Khodr T, Yoder BA. Neonatal chronic lung disease in extremely immature baboons. Am J Respir Crit Care Med 160:1333–1346, 1999.
Coalson JJ, Winter VT, Siler-Khodr T, Yoder BA. Neonatal chronic lung disease in extremely immature baboons. Am J Respir Crit Care Med 160:1333–1346, 1999.
Sunday ME, Yoder BA, Cuttitta F, Haley KJ, Emanuel RL. Bombesin-like peptide mediates lung injury in a baboon model of bronchopulmonary dysplasia. J Clin Invest 102:584–594, 1998.
Sunday ME, Yoder BA, Torday JS, Sikorski KA, Cuttitta F, Emanuel RL. Bombesin-like peptide (BLP) as a mediator of lung injury in two different baboon models of bronchopulmonary dysplasia. FASEB J 13(4):A1154 (abstract #857.4), 1999.
Subramaniam M, Sugiyama K, Coy D, Steiner C, Kong Y, Miller YE, Weller PF, Wada E, Sunday ME. Bombesin-like peptides and mast cell responses: relevance to bronchopulmonary dysplasia? Am J Respir Crit Care Med 168:601–611, 2003.
Sheng H, Enghild JJ, Bowler R, et al. Effects of metalloporphyrin catalytic antioxidants in experimental brain ischemia. Free Radical Biol Med 33:947–961, 2002.
Chang L, Subramaniam M, Yoder BA, et al. A catalytic antioxidant attenuates alveolar structural remodeling in bronchopulmonary dysplasia. Am J Respir Cell Mol Biol 167:57–64, 2003.
Groneck P, Gotze-Speer B, Oppermann M, Eiffert H, Speer CP. Association of pulmonary inflammation and increased microvascular permeability during the development of bronchopulmonary dysplasia: a sequential analysis of inflammatory mediators in respiratory fluids of high-risk preterm neonates. Pediatrics 93:712, 1994.
Lyle RE, Tryka AF, Griffin WS, Taylor BJ. Tryptase immunoreactive mast cell hyperplasia in bronchopulmonary dysplasia. Pediatr Pulmonol 19:336–343, 1995.
Raghavender B, Smith JB. Eosinophil cationic protein in tracheal aspirates of preterm infants with bronchopulmonary dysplasia. J Pediatr 130:944–947, 1997.
Gharaee-Kermani M, Phan SH. The role of eosinophils in pulmonary fibrosis. Int J Molec Med 1:43–53, 1998.
Pesci A, Bertorelli G, Gabrielli M, Olivieri D. Mast cells in fibrotic lung disorders. Chest 103:989–996, 1993.
Reiser KM, Last JA. Early cellular events in pulmonary fibrosis. Exp Lung Res 10:331–355, 1986.
Wasserman SL. The human lung mast cell. Env Health Persp 55:259–269, 1984.
Ashour K, Sunday ME. Decreased alveolar development in fetal and newborn mice given bombesin. Pediatr Res 51(4)(Part 2 of 2):61A, Abstract 353, 2002.
Toti P, Buonocore G, Tanganelli P, et al. Bronchopulmonary dysplasia of the premature baby: an immunohistochemical study. Pediatr Pulmonol 24:22–28, 1997.
Sun G, Stacey MA, Bellini A, Marini M, Mattoli S. Endothelin-1 induces bronchial myofibroblast differentiation. Peptides 18:1449–1451, 1997.
Northway WH, Jr. Commentary. Bronchopulmonary dysplasia: twenty-five years later. Pediatr 89:969–973, 1992.
Zetter BR. Angiogenesis and tumor metastasis. Ann Rev Med 49:407–424, 1998.
Beck L, D’Amore PA. Vascular development: cellular and molecular regulation. FASEB J 11:365–373, 1997.
Munshi UK, Niu JO, Siddiq MM, Parton LA. Elevation of interleukin-8 and interleukin-6 precedes the influx of neutrophils in tracheal aspirates from preterm infants who develop bronchopulmonary dysplasia. Pediatr Pulmonol 24:331–336, 1997.
Brusselle GG, Kips JC, Tavernier JH, et al. Attenuation of allergic airway inflammation in IL-4 deficient mice. Clin Exp Allergy 24:73–80, 1994.
Schwarze J, Cieslewicz G, Hamelmann E, et al. IL-5 and eosinophils are essential for the development of airway hyperresponsiveness following acute respiratory syncytial virus infection. J Immunol 162:2997–3004, 1999.
Viola JP, Kiani A, Bozza PT, Rao A. Regulation of allergic inflammation and eosinophil recruitment in mice lacking the transcription factor NFAT1: role of interleukin-4 (IL-4) and IL-5. Blood 91:2223–2230, 1998.
Matthaei KI, Foster P, Young IG. The role of interleukin-5 (IL-5) in vivo: studies with IL-5 deficient mice [Review]. Memorias do Instituto Oswaldo Cruz 92:63–68, 1997.
Temann UA, Geba GP, Rankin JA, Flavell RA. Expression of interleukin 9 in the lungs of transgenic mice causes airway inflammation, mast cell hyperplasia, and bronchial hyperresponsiveness. J Exp Med 188:1307–1320, 1998.
Mould AW, Ramsay AJ, Matthaei KI, Young IG, Rothenberg ME, Foster PS. The effect of IL-5 and eotaxin expression in the lung on eosinophil trafficking and degranulation and the induction of bronchial hyperreactivity. J Immunol 164:2142–2150, 2000.
Zhu Z, Homer RJ, Wang Z, et al. Pulmonary expression of interleukin-13 causes inflammation, mucus hypersecretion, subepithelial fibrosis, physiologic abnormalities, and eotaxin production. J Clin Invest 103:779–788, 1999.
Lee JJ, McGarry MP, Farmer SC, et al. Interleukin-5 expression in the lung epithelium of transgenic mice leads to pulmonary changes pathognomonic of asthma. J Exp Med 185:2143–2156, 1997.
Bozic CR, Lu B, Hopken UE, Gerard C, Gerard NP. Neurogenic amplification of immune complex inflammation. Science 273:1722–1725, 1996.
Hopken UE, Lu B, Gerard NP, Gerard C. Impaired inflammation responses in the reverse Arthus reaction through genetic deletion of the C5a receptor. J Exp Med 186:749–756, 1997.
Broide DH, Campbell K, Gifford T, Sriramarao P. Inhibition of eosinophilic inflammation in allergen-challenged, IL-1 receptor type 1-deficient mice is associated with reduced eosinophil rolling and adhesion on vascular endothelium. Blood 95:263–269, 2000.
Pryhuber GS, O’Brien DP, Baggs R, et al. Ablation of tumor necrosis factor receptor type 1 (p55) alters oxygen-induced lung injury. Am J Physiol Lung Cell Mol Physiol 278:L1082-L1090, 2000.
Ward NS, Waxman AB, Homer RJ, et al. Interleukin-6 induced protection in hyperoxic acute lung injury. Am J Respir Cell Mol Biol 22:535–542, 2000.
Yang X, Wang S, Fan Y, Han X. IL-10 deficiency prevents IL-5 overproduction and eosinophilic inflammation in a murine model of asthma-like reaction. Eur J Immunol 30:382–391, 2000.
Waxman AB, Einarsson O, Seres T, et al. Targeted lung expression of interleukin-11 enhances murine tolerance of 100% oxygen and diminishes hyperoxia-induced DNA fragmentation. J Clin Invest 101:1970–1982, 1998.
Minamino T, Christou H, Hsieh C-M, et al. Targeted expression of heme oxygenase-1 prevents the pulmonary inflammatory and vascular responses to hypoxia, 2001. Proc. Natl Acad Sci USA 98:8798–8803.
Choi AMK, Alam J. Heme oxygenase-1: function, regulation, and implication of a novel stress-inducible protein in oxidant-induced lung injury. Am J Respir Cell Mol Biol 15:9–19, 1996.
Christou H, Morita T, Hsieh C-M, et al. Prevention of hypoxia-induced pulmonary hypertension by enhancement of endogenous heme oxygenase-1 in the rat. Circ Res 86:1224–1229, 2000.
Contreras M, Hariharan N, Lewandoski JR, Ciesielski W, Koscik R, Zimmerman JJ. Bronchoalveolar oxyradical inflammatory elements herald bronchopulmonary dysplasia. Crit Care Med 24:29–37, 1996.
Parker RA, Lindstrom DP, Cotton RB. Evidence from twin study implies possible genetic susceptibility to bronchopulmonary dysplasia. Semin Perinatol 20:206–209, 1996.
Evans M, Palta M, Sadek M, Weinstein MR, Peters ME. Associations between family history of asthma, bronchopulmonary dysplasia, and childhood asthma in very low birth weight children. Am J Epidemiol 148:460–466, 1998.
Nickerson BG, Taussig LM. Family history of asthma in infants with bronchopulmonary dysplasia. Pediatrics 65:1140–1144, 1980.
Weitzman M, Gortmaker S, Walker DK, Sobol A. Maternal smoking and childhood asthma. Pediatrics 85:505–511, 1990.
Martinez FD, Cline M, Burrows B. Increased incidence of asthma in children of smoking mothers. Pediatrics 89:21–26, 1992.
Hanrahan JP, Tager IB, Segal MR, et al. The effect of maternal smoking during pregnancy on early infant lung function. Am Rev Respir Dis 145:1129–1135, 1992.
Hoff C, Wertelecki W, BlackBurn WR, Mendenhall H, Wiseman H, Stumpe A. Trend associations of smoking with maternal, fetal and neonatal morbidity. Obstet Gynecol 68:317–321, 1986.
Chen MF, Kimizuka G, Wang NS. Human fetal lung changes associated with maternal smoking during pregnancy. Pediatr Pulmonol 3:51–58, 1987.
Chen MF, Lewis SJ, Jagoe R, et al. Gastrin-releasing peptide gene products in mid-trimester human fetal lung with and without maternal smoking history during pregnancy. Pediatr Pulmonol 10:30–35, 1991.
Huang M-H, Friend DS, Sunday ME, et al. An intrinsic adrenergic system in mammalian heart. J Clin Invest 98:1298–1303, 1996.
Wuenschell CW, Sunday ME, Singh G, Minoo P, Slavkin HC, Warburton D. Embryonic mouse lung epithelial progenitor cells co-express immunohistochemical markers of diverse mature cell lineages. J Histochem Cytochem 44:113–123, 1996.
Lemaire I. Bombesin-related peptides modulate interleukin-1 production by alveolar macrophages. Neuropeptides 20:217–223, 1991.
Lemaire I, Jones S, Khan MF. Bombesin-like peptides in alveolar macrophage: increased release in pulmonary inflammation and fibrosis. Neuropeptides 20:63–72, 1991.
Author information
Authors and Affiliations
Corresponding author
Additional information
This review is dedicated to the memory of Dr. Hugh Wolfe, one of the pioneers in the field of Endocrine Pathology. Hugh was much admired around the world as a leading scientist, an enthusiastic mentor, an ever-loyal friend, a cheerful neighbor, a beloved husband, and a proud father and grandfather. He died suddenly on September 6, 2001, and is greatly missed by all who knew him.
Rights and permissions
About this article
Cite this article
Sunday, M.E., Shan, L. & Subramaniam, M. Immunomodulatory functions of the diffuse neuroendocrine system: Implications for bronchopulmonary dysplasia. Endocr Pathol 15, 91–106 (2004). https://doi.org/10.1385/EP:15:2:091
Issue Date:
DOI: https://doi.org/10.1385/EP:15:2:091