Abstract
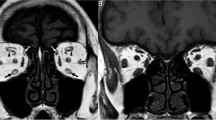
Twenty-two patients with Graves’ ophthalmopathy underwent biopsy, and two patients had both eyes biopsied. The samples for the control group (n = 4) were obtained during routine non-thyroid-related corrective strabismus surgery. Ophthalmological evaluation with clinical activity score (CAS), endocrinological evaluation, and ultrasound were used in our study. Correlation between clinical and histological analyses in connective tissues and extraocular muscles from patients with Graves’ ophthalmopathy was done. The echography results disclosed an enlargement in all extraocular recti muscles with the exception of one patient. Periodic acid-Schiff and Giemsa stains revealed a moderate number of mast cells in the endomysial connective tissue, none of which displayed significant degranulation. There were no signs of muscle cell damage. Fifteen of the biopsies showed weak cellular reactions with only scattered inflammatory cells. Furthermore, the inflammatory process may be localized and not equally distributed throughout the muscle. Thus, the biopsies might not be representative for the whole muscle. Statistical significance analysis was found when sex and CAS were compared (p = 0.001683) using the Fisher test.
In conclusion, our investigation indicates a pleomorphic pattern of histologic findings in connective tissue and extraocular muscles in Graves’ ophthalmopathy.
Similar content being viewed by others
References
Heufelder AE. Pathogenesis of Graves’ ophthalmopathy: recent controversies and progress. Eur J Endocrinol 132:532–541, 1995.
Bahn, RS, Heufelder, AE. Mechanisms of disease; pathogenesis of Graves’ ophthalmopathy. N Engl J Med 329:1468–1475, 1993.
Burch HB, Wartofsky L. Graves’ ophthalmopathy: current concepts regarding pathogenesis and management. Endocr Rev 14:747–793, 1993.
Weetman AP. Thyroid associated eye disease: pathophysiology. Lancet. 338:25–32, 1991.
Gorman CA, Heufelder AE, Bartley GB. Ophthalmology. In: DeGroot LJ (ed). Endocrinology. Saunders, Philadelphia, PA, 1994, pp. 712–725.
Hufnagel TJ, Hickey, WF, Cobbs, WH, Jacobiec, FA, Eagle, RC. Immunohistochemical and ultrastructural studies on the exentered orbital tissues of a patient with Graves’ Ophthalmopathy. Ophthalmology 91:1411–1419, 1984.
Tallstedt L, Norberg R. Immunohistochemical staining of normal and Graves’ extraocular muscle. Invest Ophtalmol Vis Sci 29:175–184, 1988.
Heufelder AE, Bahn RS. Elevated expression in situ of selection and immunoglobulin superfamily adhesion molecules in retroocular connective tissues from patients with Graves’ Ophthalmopathy. Clin Exp Immunol 91:381–389, 1993.
Weetmann AP, Cohen S, Gatter KC, Fells D, Shine B. Immunohistochemical analysis of the retrobulbar tissues in Graves’ ophthalmopathy. Clin Exp Immuno. 75:222–227, 1989.
Trokel SL, Jacobiec FA, Correlation of CT scanning and pathologic Graves’ disease. Ophthalmology 88:553–564, 1981.
Campbell RJ. Pathology of Graves’ ophthalmopathy. In: Gormann, CA, Waller RA, Dyer JA, (eds.). The eye and the orbit in thyroid disease. Raven Press, New York, 1984; pp. 25–31.
Kroll AJ, Kuwabara T. Dysthyroid ocular myopathy; anatomy, histology, and electron microscopy. Arch Ophthalmol 76:244–257, 1966.
Scott WE, Thalacker JA. Diagnosis and treatment of thyroid myopathy. Ophthalmology 88:493–498, 1981.
Kiljanski JI, Nebes V, Wall JR. The ocular muscle cell is a target of the immune system in endocrine ophthalmology. Int Arch Allergy Immunol 106:204–212, 1995.
Riley FC. Orbital pathology in Graves’ disease. Mayo Clin Proc 47:975, 1972.
Wegelius O, Asboe-Hansen G, Lamberg B-A. Retrobulbar connective tissue changes in malignant exophthalmos. Acta Endocrinol (Copenh) 25:452, 1957.
Mourits MP, Prummel MF, Wiersinga WM, Koornneef L. Clinical activity score as a guide in the management of patients with Graves’ ophthalmopathy. Clinical Endocrinology 47:9–14, 1997.
Werner SC. Modification of the classification of the eye changes of Graves’ disease: Recommendations of the Ad Hoc Committee of the American Thyroid Association. Clin Endocrinol Metab 44:203–204, 1977.
Asboe-Hansen G, Iversen K. Influence of thyrotrophic hormone on connective tissue: pathogenetic significance of mucoplysaccharides in experimental exoftalmos. Acta Endocrinol (Kbh) 8:90–96, 1951.
Wegelius O, Asboe-Hansen G, Lamberg B-A. Retrobulbar connective tissue changes in malignant exophathalmos. Acta Endocrinol (Kbh) 25:452–456, 1957.
Tengroth B. Histological studies of orbital tissues in a case of endocrine exophthalmos before and after remission. Acta Ophthalmo (Kbh) 42:588–591, 1964.
Ludwig AW, Boas NF, Soffer LJ. Role of mucopolysaccharides in pathogenesis of experimental exophthalmos. Proc Soc Exp Biol Med 73:137–140, 1950.
Smelser GK, Ozanics V. Hydrophilia of the orbital connective tissue in experimental exophthalmos. Am J Ophthalmol 47:380–387, 1959.
Smelser GK. A comparative study of experimental and clinical exophthalmos. Am J Ophthalmol 20:1189–1203, 1937.
Ossoinig KC. Standardized echography: basic principles clinical applications and results. Int Ophthalmol Clin 19(4):127–210, 1979.
Prummel MF, Suttorp-Schulten MSA, Wiersinga WM, Verbeek AM, Mourits MP, Koornneef L. A new ultrasonographic method to detect disease activity and predict response to immunosuppressive treatment in Graves’ ophthalmopathy. Ophthalmology 100:556–561, 1993.
Marcocci C, Vitti P, Cetani F, et al. Thyroid ultrasonography helps to identify patients with diffuse lymphocytic thyroiditis who are prone to develop hypothyroidism. J Clin Endocrinol Metab 72:209–213, 1991.
Skalka HW. The use of ultrasonography in the diagnosis of endocrine orbitopathy. Neuro Ophthalmology 1:109–116, 1980.
Harrie RP. A—scan in Graves’ ophthalmopathy- letters to editor, Opthalmology, 100:10, 1993.
Byrne SF, Gendron EK, Glaser JS, Fever W, Atta HR. Diameter of normal extraocular recti muscles with echography. Am J Ophthalmol 112:706–713, 1991.
Bahn RS. The fibroblast is the target cell in the connective tissue manifestations of Graves’ disease. Int Arch Allergy Immunol 106:213–218, 1995.
Heufelder AE, Bahn RS. Detection and localisation of cytokine immunoreactivity in retroocular connective tissue in Graves’ ophthalmopathy. Eur J Clin Invest 23:10–17, 1993.
Pappa A, Calder V, Ajjan R, Fells P, Ludgates M, Weetman AP, Lightman S. Analysis of extraocular muscle-infiltrating T cells in thyroid-associated ophthalmopathy (TAO). Clin Exp Immunol 109:362–369, 1997.
Perros P, Kendall-Taylor P. The pathogenesis of thyroid-associated ophthalmopathy. J Endocrinol 122:619–624, 1989.
Bartley GB, Gorman CA. Diagnostic criteria for Graves’ ophthalmopathy. Am J Ophthalmol 119, pp. 6, 792–795, 1995.
Author information
Authors and Affiliations
Additional information
Address correspondence to Escola Paulista de Medicina, Sao Paulo, Brazil
Rights and permissions
About this article
Cite this article
Matos, K., Nosé, V., Manso, P.G. et al. Correlation between clinical and histological analyses in retroocular connective tissues and extraocular muscles from patients with Graves’ ophthalmopathy. Endocr Pathol 11, 185–194 (2000). https://doi.org/10.1385/EP:11:2:185
Issue Date:
DOI: https://doi.org/10.1385/EP:11:2:185