Abstract
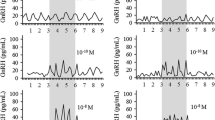
The present study examined the effects of continuous treatment with gonadotropi-releasing hormone (GnRH) on GnRH receptor (GnRH-R) mRNA levels in dispersed cultures of rat pituitary cells. Pituitary GnRH-R mRNA levels were determined by competitive reverse transcriptase polymerase chain reaction. When pituitary cells were continuously exposed to a low dose of GnRH (0.2 nM), GnRH-R mRNA levels were transiently increased. The levels of GnRH-R mRNA were significantly increased up to 6 h and diminished to untreated levels by 24 h. Luteinizing hormone (LH) release was also increased significantly up to 12 h, maintaining similar levels in LH release therafter. When GnRH antagonist([d-pGlu1, d-Phe2, d-Trp3.6]-LH-RH) was added to the cultures together with GnRH (0.2 nM) for 6 h, the stimulatory effect of GnRH on GnRH-R mRNA levels and LH release was significantly diminished in a dose-related manner. In another experiment, pituitary cells were treated with various doses of GnRH (0.02–200 nM) for a relatively short (6 h) or a longer (24 h) period. When pituitary cells were exposed for 6 h, all doses of GnRH 0.02–200 nM) significantly increased GnRH-R mRNA levels in a dose-dependent manner. By contrast, continuous exposure to GnRH for 24 h was ineffective in changing pituitary GnRH-R mRNA levels at any given doses. These results indicate that the duration of GnRH treatment is critical for upregulation of GnRH-R mRNA by continuous GnRH. When pituitary cells were treated for 6 h with either a continuous mode of GnRH (0.2 nM) or an hourly pulsatile mode of GnRH (0.2 nM, 6 mi/h), both treatment significantly augmented GnRH-R mRNA levels. Thus, the modes of GnRH application, if treated for a relatively short period, do not appear to make a significant difference in upregulation of GnRH-R mRNA levels. Collectively, our data provide strong evidence that continuous GnRH application is able to upregulate pituitary GnRH-R mRNA levels, if treated for a relatively short period (6 h).
Similar content being viewed by others
References
Burgus, R., Butcher, M., Amoss, M., Ling, N., Monahan, M., Rivier, J., Fellows, R., Blackwell, R., Vale, W., and Guillemin, R. (1972). Proc. Natl. Acad. Sci. USA 69, 278–282.
Clayton, R. N. (1989). J. Endocrinol. 120, 11–19.
Katt, J. A., Duncan, J. A., Herbon, L., Barkan, A., and Marshall J. C. (1985). Endocrinology 116, 2113–2115.
Loumaye, E. and Catt, K. J. (1983). J. Biol. Chem. 258, 12,002–12,009.
Conn, P. M., Rogers, D. C., and Seay, S. G. (1984). Mol. Pharmacol. 25, 51–55.
McArdle, C. A., Gorospe, W. C., Huckle, W. R., and Conn, P. M. (1987). Mol. Endocrinol. 1, 420–429.
Loumaye, E. and Catt, K. J. (1982), Science 215, 983–985.
Reinhart, J., Mertz, L. M., and Catt, K. J. (1992) J. Biol. Chem. 267, 21281–21284.
Eidne, K. A., Sellar, R. E., Couper, G., Anderson, L., and Taylor, P. L. (1992). Mol. Cell. Endocrinol. 90, R5-R9.
Kaiser, U. B., Jakubowiak, A., Steinberger, A., and Chin, W. W. (1993). Endocrinology 133, 931–934.
Bauer-Dantoin, A. C., Weiss, J., and Jameson, J. L. (1995). Endocrinology 136, 1014–1019.
Kaiser, U. B., Jakubowiak, A., Steinberger, A., and Chin, W. W. (1997). Endocrinology 138, 1224–1231.
Belchetz, P. E., Plant, T. M., Nakai, Y., Keogh, E. J., and Knobil, E. (1978). Science 202, 631–633.
Hubert, J. F., Simard, J., Gagne, B., Barden, N., and Labrie, F. (1988). Mol. Endocrinol. 2, 521–527.
Burrin, J. M. and Jameson, J. L. (1989). Mol. Endocrinol. 3, 1643–1651.
Weiss, J., Jameson, J. L., Burrin, J. M., and Crowley, W. F. Jr. (1990). Mol. Endocrinol. 4, 557–564.
Shupnik, M. A. (1990). Mol. Edocrinol. 4, 1444–1450.
Chedrese, P. J., Kay, T. W., and Jameson, J. L. (1994). Endocrinology 134, 2475–2481.
Lin, X., and Conn, P. M. (1998). Endocrinology 139, 3896–3902
Cheon, M., Park, Y., Kam, K., and Ryu, K. (1997). Program of the 79th annual meeting of endocrinesociety, Minneaplis, MN, (abstract P2-145).
Haisenleder, D. J., Cox, M. E., Parsons, S. J., and Marshall, J. C. (1998). Endocrinology 139, 3104–3111.
Li, X., Lei, Z. M., and Rao, C. V. (1996). Endocrinology 137, 899–904.
Park, D., Cheon, M., Kim, C., Kim, K., and Ryu, K. (1996) Eur. J. Endocrinol. 134, 236–242.
Bouamoud, N., Lerrant, Y., Ribot, G., and Counis, R. (1992). Mol. Cell. Endocrinol. 88, 143–151.
Tsutsumi, M., Laws, S. C., and Sealfon, S. C. (1993). Mol. Endocrinol. 7, 1625–1633.
Tsutsumi, M., Laws, S. C., Rodic, V., and Sealfon, S. C. (1995). Endocrinology 136, 1128–1136.
Yasin, M., Dalkin, A. C., Haisenleder, D. J., Kerrigan, J. R., and Marshall, J. C. (1995), Endocrinology 136, 1559–1564.
Turzillo, A. M., Juengel, J. L., and Nett, T. M. (1995). Biol. Reprod. 53, 418–423.
Brooks, J., and McNeilly, A. S. (1994). J. Endocrinol. 143, 175–182.
Sakurai, H., Adams, B. M., and Adams, T. E. (1997). J. Anim. Sci. 75, 189–194.
Adams, B. M., Sakurai, H., and Adams, T. E. (1997). J. Reprod. Fertil. 111, 207–212.
Moenter, S. M., Brand, R. C., and Karsch, F. J. (1992). Endocrinology 130, 2978–2984.
Nishihara, M., Sano, A., and Kimura, F. (1994). Neuroendocrinology 59, 513–519.
Dauer-Dantoin, A. C., Holleberg, A. N., and Jameson, J. L. (1993). Itendocrinology 133, 1911–1914.
Wilfinger, W. W., Larsen, W. J., Downs, T. R., and Wilbur, D. L. (1984). Tissue Cell 16, 483–497.
Chomczynski, P. and Sacchi, N. (1987). Anal. Biochem. 162, 156–159.
Seong, J. Y., Yang, S. S., Kam, K., Han, Y. G., Kwon, H. B., Ryu, K., and Kim, K. (1998). Mol. Brain Res. 53, 226–235.
Kaiser, U. B., Zhao, D., Cardona, G. R., and Chin, W. W. (1992) Biochem. Biophys. Res. Commun. 189, 1645–1652.
Seong, J. Y. and Kim, K. (1996). Neurosci. Protocols 20, 1–17.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheon, M., Park, D., Kim, K. et al. Homologous upregulation of GnRH receptor mRNA by continuous GnRH in cultured rat pituitary cells. Endocr 11, 49–55 (1999). https://doi.org/10.1385/ENDO:11:1:49
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1385/ENDO:11:1:49