Abstract
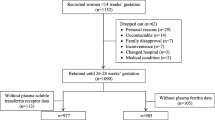
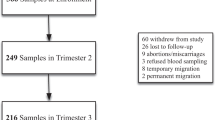
It is well established that Fe and ceruloplasmin interact in animals and in in vitro models. However, Fe-mediated regulation of ceruloplasmin has never been investigated in humans. In an observational study, 53 pregnant women aged 19–39 yr (29.8±0.7 yr, mean ± SEM) were recruited at the Aberdeen Antenatal Clinic, Aberdeen Maternity Hospital, UK. All requirements for local ethical committees were followed. Venous blood samples were taken from each woman at 34 wk gestation for measurement of Fe status and ceruloplasmin. Various parameters were used to test for Fe status. The most sensitive one appeared to be soluble transferrin receptor, which increased with parity. In the population studied, there was no relationship between hemoglobin or ferritin and serum ceruloplasmin. However, using soluble transferrin receptor (sTfR) levels, we were able to demonstrate an inverse linear relationship (r=0.37, p=0.021, n=41) between Fe status and ceruloplasmin. Fe supplementation, number of previous pregnancies, and smoking habits did not affect this relationship. Our data support in vitro results showing regulation of ceruloplasmin by Fe and also suggest that the interactions between Fe and ceruloplasmin should be considered when Fe supplementation is given.
Similar content being viewed by others
References
W. R. Humphries, B. W. Young, M. Phillippo, et al., The effects of iron and molybdenum on copper metabolism in cattle, Proc. Nutr. Soc. 40, 68A (1981).
W. R. Humphries, M. Phillippo, B. W. Young, et al., The influence of dietary iron and molybdenum on copper metabolism in calves, Br. J. Nutr. 49, 77 (1983).
M. A. Johnson and C. L. Murphy, Adverse effects of high dietary iron and ascorbic acid on copper status in copper-deficient and copper-adequate rats, Am. J. Clin. Nutr. 47, 96 (1988).
A. B. Abdel-Mageed, R. Welti, F. W. Oehme, et al., Perinatal hypocuprosis affects synthesis and composition of neonatal lung collagen, elastin, and surfactant, Am. J. Physiol. 267, L679 (1994).
C. K. Mukhopadhyay, Z. K. Attieh, and P. L. Fox, Role of ceruloplasmin in cellular iron uptake, Science 279, 714 (1998).
R. Danzeisen, C. Fosset, Z. Chariana, et al., Placental ceruloplasmin homolog is regulated by iron and copper and is implicated in iron metabolism, Am. J. Physiol. Cell Physiol. 282, 472 (2002).
G. R. Lee, S. Nacht, J. N. Lukens, et al., Iron metabolism in copper-deficient swine, J. Clin. Invest. 47, 2058 (1968).
H. A. Ragan, S. Nacht, G. R. Lee, et al., Effect of ceruloplasmin on plasma iron in copper-deficient swine, Am. J. Physiol. 217, 1320 (1969).
H. P. Roeser, G. R. Lee, S. Nacht, et al., The role of ceruloplasmin in iron metabolism, J. Clin. Invest. 49, 2408 (1970).
L. Gambling, R. Danzeisen, S. Gair, et al., Effect of iron deficiency on placental transfer of iron and expression of iron transport proteins in vivo and in vitro, Biochem. J. 356, 883 (2001).
C. P. Gibbs, Maternal physiology, Clin. Obstet. Gynecol. 24, 525 (1981).
O. Koller, The clinical significance of hemodilution during pregnancy, Obstet. Gynecol. Surveys 37, 649 (1982).
T. H. Bothwell, Iron requirements in pregnancy and strategies to meet them, Am. J. Clin. Nutr. 72, 257S (2000).
R. Gofin, B. Adler, and H. Palti, Effectiveness of iron supplementation compared to iron treatment during pregnancy, Public Health 103, 139 (1989).
J. Liljestrand, S. Bergstrom, and G. Birgegard, Anaemia of pregnancy in Mozambique, Trans. R. Soc. Trop. Med. Hyg. 80, 249 (1986).
A. C. Looker, P. R. Dallman, M. D. Carroll, et al., Prevalence of iron deficiency in the United States, JAMA 277, 973 (1997).
K. Singh, Y. F. Fong, and S. Arulkumaran, Anaemia in pregnancy: a cross-sectional study in Singapore, Eur. J. Clin. Nutr. 52, 65 (1998).
K. Takagi, M. Nakao, Y. Ogura, T. Nabeshima, and A. Kunii, Sensitive colorimetric assay of serum diamine oxidase. Clin. Chim. Acta 226, 67 (1994).
F.W. Sunderman and S. Nomoto, Measurement of human serum ceruloplasmin by its p-phenylenadiamine oxidase activity. Clin. Chem. 16, 903 (1970).
T. H. Bothwell, R. W. Charlton, J. D. Cook, et al., Iron requirements in pregnancy and strategies to meet them, Am. J. Clin. Nutr. 72, 257S (2000).
WHO, Iron deficiency Anaemia (IDA), World Health Organisation, Geneva (2000).
WHO, Micronutrient Deficiencies: Battling Iron Deficiency Anaemia, World Health Organisation, Geneva (2001).
A. F. Fleming, Tropical obstetrics and gynaecology. 1. Anaemia in pregnancy in tropical Africa, Trans. R. Soc. Trop. Med. Hyg. 83, 441 (1989).
A. F. Fleming, The aetiology of severe anaemia in pregnancy in Ndola, Zambia, Ann. Trop. Med. Parasitol. 83, 37 (1989).
S. Khanna, B. Dube, and S. Kumar, Anaemia of pregnancy in northern India. Nature and therapeutic follow-up, Trop. Geogr. Med. 29, 24 (1977).
J. Thomson, Anaemia in pregnant women in eastern Caprivi, Namibia, S. Afr. Med. J. 87, 1544 (1997).
N. G. Carretti, A. G. Eremita, D. Paternoster, et al., Iron balance in pregnancy in relation to anemia, Clin. Exp. Obstet. Gynecol. 19, 218 (1992).
A. J. Dabbagh, C. W. Trenam, C. J. Morris, et al., Iron in joint inflammation, Ann. Rheum. Dis. 52, 67 (1993).
D. A. Lipschitz, J. D. Cook, and C. A. Finch, A clinical evaluation of serum ferritin as an index of iron status, N. Engl. J. Med. 290, 1213 (1974).
M. T. Carriaga, B. S. Skikne, B. Finley, et al., Serum transferrin receptor for the detection of iron deficiency in pregnancy, Am. J. Clin. Nutr. 54, 1077 (1991).
N. Ahluwalia, Diagnostic utility of serum transferrin receptors measurement in assessing iron status, Nutr. Rev. 56, 133 (1998).
H. Khumalo, Z. A. Gomo, V. M. Moyo, et al., Serum transferrin receptors are decreased in the presence of iron overload, Clin. Chem. 44, 40 (1998).
Y. Kohgo, Y. Niitsu, and Y. Nishisato, Quantitation and characterisation of serum transferrin receptor in patients with anemias and polycythemias, Jpn. J. Med. 27, 64 (1988).
U. Rusia, C. Flowers, N. Madan, et al., Serum transferrin receptors in detection of iron deficiency in pregnancy, Ann. Hematol. 78, 358 (1999).
H. C. Lukaski, L. M. Klevay, and D. B. Milne, Effects of dietary copper on human autonomic cardiovascular function, Eur. J. Appl. Physiol. Occup. Physiol. 58, 74 (1988).
D. B. Milne and P. E. Johnson, Assessment of copper status: effect of age and gender on reference ranges in healthy adults, Clin. Chem. 39, 883 (1993).
A. A. Jones, R. A. DiSilvestro, M. Coleman, et al., Copper supplementation of adult men: effects on blood copper enzyme activities and indicators of cardiovascular disease risk, Metabolism 46, 1380 (1997).
C. A. Kehoe, M. S. Faughnan, W. S. Gilmore, et al., Plasma diamine oxidase activity is greater in copper-adequate than copper-marginal or copper-deficient rats, J. Nutr. 130, 30 (2000).
M. Hilton, D. C. Spenser, P. Ross, et al., Characterisation of the copper uptake mechanism and isolation of the ceruloplasmin receptor/copper transporter in human placental vesicles, Biochim. Biophys. Acta 1245, 153 (1995).
S. H. Lee, R. Lancey, A. Montaser, et al., Ceruloplasmin and copper transport during the latter part of gestation in the rat, Proc. Soc. Exp. Biol. Med. 203, 428 (1993).
L. Gambling, S. Gair, and H. J. McArdle, The effect of maternal iron deficiency on fetal growth, iron and copper metabolism in the rat, J. Physiol. 528P, 5S (2000).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fosset, C., McGaw, B.A., Abramovich, D. et al. Interrelations between ceruloplasmin and Fe status during human pregnancy. Biol Trace Elem Res 98, 1–12 (2004). https://doi.org/10.1385/BTER:98:1:01
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:98:1:01