Abstract
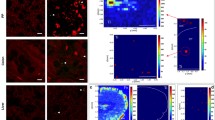
Selenium (Se) is a dietary trace element that is essential for effective immunity and protection from oxidative damage induced by ultraviolet radiation (UVR). Langerhans cells (LC) represent the major antigen-presenting cells resident in the epidermis; a proportion migrate from the skin to the draining lymph nodes in response to UVR. Because it is known that Se deficiency impairs immune function, we determined what effect this has on LC numbers. CH3/HeN mice were weaned at 3 wk and placed on diets containing <0.005 ppm of Se (Se deficient) or 0.1 ppm of Se (Se adequate, control mice). After 5 wk on the diet, the epidermal LC numbers in the Se-adequate group were 966±51 cells/mm2 and LC counts in the epidermis of the Se-deficient mice were 49% lower (p<0.05). Glutathione peroxidase-I (GPx) activity was measured in the epidermis, lymph nodes, and liver. In the epidermis, the activity of GPx in the Se-deficient mice was only 39% (p<0.01) of that seen in epidermis from Se-adequate mice (1.732 U/mg protein). The mice were then irradiated with one dose of 1440 J/m2 of broadband UVB or mock irradiated. After 24 h, the decrease in LC number after UVB was greater in the Se-adequate mice, (40% decrease) compared to the Se-deficient group (10%). Thus, Se deficiency reduces epidermal LC numbers, an effect that might compromise cutaneous immunity.
Similar content being viewed by others
References
G. B. Toews, P. R. Bergstresser, and J. W. Streilein, Epidermal Langerhans cell density determines whether contact hypersensitivity or unresponsiveness follows skin painting with DNFB, J. Immunol. 124, 445–453 (1980).
K. D. Cooper, L. Oberhelman, T. A. Hamilton, et al., UV exposure reduces immunization rates and promotes tolerance to epicutaneous antigens in humans: relationship to dose, CD1a-DR+ epidermal macrophage induction, and Langerhans cell depletion, Proc. Natl. Acad. Sci. USA 89, 8497–8501 (1992).
W. Aberer, G. Schuler, G. Stingl, et al., Ultraviolet light depletes surface markers of Langerhans cells, J. Invest. Dermatol. 76, 202–210 (1981).
A. Tang and M. C. Udey, Inhibition of epidermal Langerhans cell function by low dose ultraviolet B radiation. Ultraviolet B radiation selectively modulates ICAM-1 (CD54) expression by murine Langerhans cells, J. Immunol. 146, 3347–3355 (1991).
M. C. Udey, Cadherins and Langerhans cell immunobiology, Clin. Exp. Immunol. 107(Suppl. 1), 6–8 (1997).
M. S. Duthie, I. Kimber, and M. Norval, The effects of ultraviolet radiation on the human immune system, Br. J. Dermatol 140, 995–1009 (1999).
M. Cumberbatch and I. Kimber, Tumour necrosis factor-alpha is required for accumulation of dendritic cells in draining lymph nodes and for optimal contact sensitization, Immunology 84, 31–35 (1995).
M. Cumberbatch, R. J. Dearman, and I. Kimber, Interleukin 1 beta and the stimulation of Langehans cell migration: comparisons with tumour necrosis factor alpha, Arch. Dermatol. Res. 289, 277–284 (1997).
M. Cumberbatch and I. Kimber, Dermal tumour necrosis factor-alpha induces dendritic cell migration to draining lymph nodes, and possibly provides one stimulus for Langerhans’ cell migration, Immunology 75, 257–263 (1992).
A. M. Moodycliffe, I. Kimber, and M. Norval, Role of tumour necrosis factor-alpha in ultraviolet B light-induced dendritic cell migration and suppression of contact hypersensitivity, Immunology 81, 79–84 (1994).
M. Cumberbatch, I. Fielding, and I. Kimber, Modulation of epidermal Langerhans’ cell frequency by tumour necrosis factor-alpha, Immunology 81, 395–401 (1994).
T. Horio and H. Okamoto, Oxygen intermediates are involved in ultraviolet radiation-induced damage of Langerhans cells, J. Invest. Dermatol. 88, 699–702 (1987).
I. Iwai, M. Hatao, M. Naganuma, et al., UVA-induced immune suppression through an oxidative pathway, J. Invest. Dermatol. 112, 19–24 (1999).
F. Trautinger, Mechanisms of photodamage of the skin and its functional consequences for skin ageing, Clin. Exp. Dermatol. 26, 573–577 (2001).
J. Fleming, A. Ghose, and P. R. Harrison, Molecular mechanisms of cancer prevention by selenium compounds, Nutr Cancer 40, 42–49 (2001).
J. E. Spallholz, L. M. Boylan, and H. S. Larsen, Advances in understanding selenium role in the immune-system, Ann. NY Acad. Sci. 587, 123–139 (1990).
R. C. McKenzie, T. S. Rafferty, and G. J. Beckett, Selenium: an essential element for immune function, Immunol. Today 19, 342–345 (1998).
R. C. McKenzie, T. S. Rafferty, J. R. Arthur, et al., Effects of selenium on immunity and ageing, in Selenium Its Molecular Biology and Role in Human Health, D. L. Hatfield, ed., Kluwer Academic, Boston, pp. 258–272 (2001).
C. B. Allan, G. M. Lacourciere, and T. C. Stadtman, Responsiveness of selenoproteins to dietary selenium, Annu. Rev. Nutr. 19, 1–16 (1999).
R. C. McKenzie, J. R. Arthur, and G. J. Beckett, Selenium and the regulation of cell signalling, growth and survival: molecular and mechanistic aspects, Antiox. Redox Signal. 4, 339–355 (2002).
K. Overvad, E. B. Thorling, P. Bjerring, et al., Selenium inhibits UV-light-induced skin carcinogenesis in hairless mice, Cancer Lett. 27, 163–170 (1985).
K. E. Burke, G. F. Combs, Jr., E. G. Gross, et al., The effects of topical and oral l-selenomethionine on pigmentation and skin cancer induced by ultraviolet irradiation, Nutr. Cancer 17, 23–37 (1992).
B. C. Pence, E. Delver, and D. M. Dunn, Effects of dietary selenium on UVB-induced skin carcinogenesis and epidermal antioxidant status, J. Invest. Dermatol. 102, 759–761 (1994).
L. C. Clark, G. F. Graham, R. G. Crounse, R. Grimson, B Hulka, and C. M. Shy, Plasma selenium and skin neoplasms: a case-control study, Nutr. Cancer, 6, 13–21 (1984).
M. S. Stewart, G. S. Cameron, and B. C. Pence, Antioxidant nutrients protect against UVB-induced oxidative damage to DNA of mouse keratinocytes in culture, J. Invest. Dermatol. 106, 1086–1089 (1996).
T. S. Rafferty, C. Walker, J.A.A. Hunter, et al., Inhibition of ultraviolet radiation B (UVB)-induced IL-10 expression in murine keratinocytes by selenium compounds, Br. J. Dermatol. 146, 485–489 (2002).
R. C. McKenzie, Selenium, ultraviolet radiation and the skin, Clin. Exp. Dermatol. 25, 631–636 (2000).
J. R. Arthur, P. C. Morrice, F. Nicol, et al., The effects of selenium and copper deficiencies on glutathione S-transferase and glutathione peroxidase in rat liver, Biochem. J. 248, 248–544 (1987).
A. G. Abdel-Rahim, J. R. Arthur, and C. F. Mills, Effects of dietary copper, cadmium, iron, molybdenum and manganese on selenium utilization by the rat, J. Nutr. 116, 403–411 (1986).
J. T. Rotruck, A. L. Pope, H. E. Ganther, et al., Selenium: biochemical role as a component of glutathione peroxidase, Science 179, 588–590 (1973).
J. R. Arthur and R. Boyne, Superoxide dismutase and glutathione peroxidase activities in neutrophils from selenium deficient and copper deficient cattle, Life Sci. 36, 1569–1575 (1985).
M. M. Bradford, A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principal of protein-dye binding, Anal. Biochem. 72, 248–254 (1976).
L. Kiremidjian-Schumacher, M. Roy, H. I. Wishe, et al., Supplementation with selenium augments the functions of natural killer and lymphokine-activated killer cells, Biol. Trace Element Res. 52, 227–239 (1996).
H. T. Petrie, L. W. Klassen, P. S. Klassen, et al., Selenium and the immune response: 2. Enhancement of murine cytotoxic T-lymphocyte and natural killer cell cytotoxicity in vivo, J. Leuk. Biol. 45, 215–220 (1989).
G. J. Beckett, J. R. Arthur, S. Miller, et al., Selenium, in Dietary Enhancement of Human Immune Function, D. A. Hughes, A. Bendich, and G. Darlington, eds., Humana, Totowa, NJ, in press (2003).
J. R. Arthur, F. Nicol, and G. J. Beckett, Selenium deficiency, thyroid hormone metabolism, and thyroid hormone deiodinases, Am. J. Clin. Nutr. 57, 236S-239S (1993).
K. M. Thompson, H. Haibach, and R. A. Sunde, Glutathione peroxidase and phospholipid hydroperoxide glutathione peroxidase are differently regulated in rats by dietary selenium, J. Nutr. 125, 1438–1446 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rafferty, T.S., Norval, M., El-Ghorr, A. et al. Dietary selenium levels determine epidermal langerhans cell numbers in mice. Biol Trace Elem Res 92, 161–171 (2003). https://doi.org/10.1385/BTER:92:2:161
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:92:2:161