Abstract
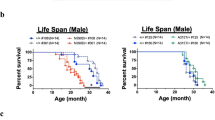
Metallothionein (MT) mRNA expression was investigated in a rodent model (G93A SOD1 transgenic mouse) for a lethal motor neuron disease, amyotrophic lateral sclerosis (ALS). In 8-wk-old mice that did not yet exhibit motor paralysis, MT-I mRNA expression was already significantly upregulated in the region of the spinal cord responsible for motor paralysis. The expression of another isoform, MT-III, was not changed. In the cerebellum, which is not responsible for motor paralysis in ALS, neither the expression profiles of MT-I nor MT-III were altered. In 16-wk-old mice exhibiting motor paralysis, the expression of MT-I mRNA remained upregulated and the MT-III level tended to be elevated. Although no significant differences were found in the levels of both isoforms in the liver or kidney of 8-wk-old mice, the MT-I mRNA expression level was significantly upregulated in the kidney and liver of 16-wk-old mice. These results indicated that the MT-I isoform, but not the MT-III isoform, is associated with motor neuron death in ALS and suggested that the disease might be a systemic disorder to which the spinal cord is particularly susceptible.
Similar content being viewed by others
References
M. Ascher, M. G. Cherian, C. D. Klaassen, R. D. Palmiter, J. C. Erickson, and A. I. Bush, Metallothionein in brain. The role in physiology and pathology, Toxicol. Appl. Pharmacol. 142, 229–242 (1997).
J. Hidalgo, M. Aschner, P. Zatta, and M. Vašák, Roles of the metallothionein family of proteins in the central nervous system, Brain Res. Bull. 55, 133–145 (2001).
M. A. Sens, S. Somji, S. H. Garrett, C. L. Beall, and D. A. Sens, Metallothionein isoform 3 overexpression is associated with breast cancers having a poor prognosis, Am. J. Pathol. 159, 21–26 (2001).
J. G. Hoey, S. H. Garrett, M. A. Sens, J. H. Todd, and D. A. Sens, Expression of MT-3 mRNA in human kidney, proximal tubule cell cultures, and renal cell cartinoma, Toxicol. Lett. 92, 149–160 (1997).
Y. Uchida, K. Takio, K. Titani, Y. Ihara, and M. Tomonaga, The growth inhibitory factor that is deficient in the Alzheimer's disease brain is a 68 amino acid metallothionein-like protein, Neuron 7, 337–347 (1991).
L. P. Rowland and N. A. Shneider, Medical progress. Amyotrophic lateral sclerosis, N. Engl. J. Med. 344, 1688–1700 (2001).
D. R. Rosen, T. Siddique, D. Patterson, et al., Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis, Nature 362, 59–62 (1993).
J-P. Julien, Amyotrophic lateral sclerosis. Undolding the toxicity of the misfolded, Cell 104, 581–591 (2001).
P. C. Wong, C. A. Pardo, D. R. Borchelt, et al. An adverse property of a familial ALS-linked SOD1 mutation causes motor neuron disease characterized by vacuolar degeneration ofmitochondria, Neuron 14, 1105–1116 (1995).
M. E. Gurney, H. Pu, A. Y. Chiu, et al., Motor neuron degeneration in mice that express a human Cu, Zn superoxide dismutase mutation, Science 264, 1772–1775 (1994).
R. H. Brown, Jr., Amyotrophic lateral sclerosis. Recent insights from genetics and transgenic mice, Cell 80, 687–692 (1995).
S. Choudhuri, W. L. Liu, N. E. J. Berman, and C. D. Klaassen, Cadmium accumulation and metallothionein expression in brain of mice at different stages of development, Toxicol. Lett. 84, 127–133 (1996).
S-I. Ono, L. Cai, D. J. Koropatnick, and M. G. Cherian, Radiation exposure does not alter metallothionein III isoform expression in mouse brain, Biol. Trace Element Res. 74, 23–30 (2000).
R. D. Palmiter, S. D. Findley, T. E. Whitmore, and D. M. Durnam, MT-III. a brain specific member of the metallothionein gene family, Proc. Natl. Acad. Sci. USA 89, 6333–6337 (1992).
J. Y. Tso, X. H. Sun, T. H. Kao, K. S. Reece, and R. Wu, Isolation and characterization of rat and human glyceraldehydes-3-phosphalte dehydrogenase cDNAs, Nucleic Acid Res. 13, 2485–2502 (1985).
P. A. E. Sillevis Smitt, H. G. T. Blaauwgeers, D. Troost, and J. M. B. Vianney de Jong, Metallothionein immunoreactivity is increased in the spinal cord of patients with amyotrophic lateral sclerosis, Neurosci. Lett. 144, 107–110 (1992).
P. A. E. Sillevis Smitt, T. P. J. Mulder, H. W. Verspaget, H. G. T. Blaauwgeers, D. Troost and J. M. B. Vianney de Jong, Metallothionein in amyotrophic lateral sclerosis, Biol. Signals 3, 193–197 (1994).
Y. H. Gong and J. H. Elliott, Metallothionein expression is altered in a transgenic murine model of familial amyotrophic lateral sclerosis, Exp. Neurol. 162, 27–36 (2000).
S. Nagano, M. Satoh, H. Sumi, et al., Reduction of metallothioneins promotes the disease expression of familial amyotrophic lateral sclerosis mice in a dose-dependent manner, Eur. J. Neurosci. 13, 1363–1370 (2001).
P. A. E. Sillevis, H. van Beek, A-J. Baars, et al., Increased metallothionein in the liver and kidney of patients with amyotrophic lateral sclerosis, Arch. Neurol. 49, 721–724 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ono, SI., Endo, Y., Tokuda, EI. et al. Upregulation of metallothionein-I mRNA expression in a rodent model for amyotrophic lateral sclerosis. Biol Trace Elem Res 113, 93–103 (2006). https://doi.org/10.1385/BTER:113:1:93
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:113:1:93