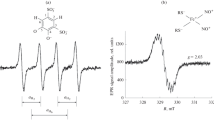
Abstract
The effects of some naturally occurring iron ion chelators and their derivatives on the electron transfer from ferrous ions to oxygen molecules were examined by measuring oxygen consumption rates. Of the compounds examined, quinolinic acid, fusaric acid, and 2-pyridinecarboxylic acid repressed the oxygen consumption, whereas chlorogenic acid, caffeic acid, gallic acid, catechol l-β-(3,4-dihydroxyphenyl) alanine, and xanthurenic acid accelerated it. Theoretical calculations showed that the energies of the highest occupied molecular orbitals (HOMOs) of [Fe(II)(ligand)3]− complexes were relatively high when the ligands were caffeic acid and its derivatives such as catechol, gallic acid, and l-β-(3,4-dihydroxyphenyl) alanine. On the other hand, the energies of the HOMOs of [Fe(II)(ligand)3]− complexes were relatively low when the ligands were quinolinic acid and its derivatives such as 2-pyridinecarboxylic acid and fusaric acid. The energies of the HOMOs appear to be closely related with acceleration or repression of the oxygen consumption; that is to say, when the energy of the HOMO is high, the oxygen consumption is accelerated, and vice versa.
Similar content being viewed by others
References
G. W. Bates, E. F. Workman, Jr., and M. R. Schlabach, Does transferring exhibit feroxidase activity? Biochem. Biophys. Res. Commun. 50, 84–90 (1973).
D. C. Harris and P. Aisen, Facilitation of Fe(II) autoxidation by Fe(III) complexing agents, Biochim. Biophys. Acta 329, 156–158 (1973)
S. J. Klebanoff, A. M. Waltersdorph, B. R. Michel, et al., Oxygen-based free radical generation by ferrous ions and deferoxamine, J. Biol. Chem. 264, 19,765–19,771 (1989).
H. Kosaka, Y. Katsuki, and T. Shiga, Spin trapping study on the kinetics of Fe2+ autoxidation: formation of spin adducts and their destruction by superoxide, Arch. Biochem. Biophys. 293, 401–408 (1992).
G. Matheis and H. D. Belitz, Untersuchungen zur enzymatischen Braunung bei Kartoffeln (Solanum tuberosum), Z. Lebensm. Unters.-Forsch. 163, 186–190 (1977).
J. Van Buren, L. De Vos, and W. Pilnik, Measurement of chlorogenic acid and flavonol glycosides in apple juice by a chromatographic-fluorometric method, J. Food Sci. 38, 656–658 (1973).
J. R. L. Walker, Flavonoid pigments in the skins of New Zealand apples. N. Z. J. Sci. 7, 585–588 (1964).
M. R. Ullah and J. C. Jain, Seasonal variations in the chlorogenic acids content of tea, J. Sci. Food Agric. 31, 355–358 (1980).
B. C. Challis and C. D. Bartlett, Possible cocarcinogenic effects of coffee constituents, Nature 254, 532–533 (1975).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian 98, Revision A.11, Gaussian, Inc., Pittsburgh, PA (2001).
A. K. Rappe, C. J. Casewit, K. S. Colwell, et al., UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations, J. Am. Chem. Soc. 114, 10,024–10,035 (1992).
A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys. 98, 5648–5652 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hirai, T., Fukushima, K., Kumamoto, K. et al. Effects of some naturally occurring iron ion chelators on in vitro superoxide radical formation. Biol Trace Elem Res 108, 77–85 (2005). https://doi.org/10.1385/BTER:108:1-3:077
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:108:1-3:077