Abstract
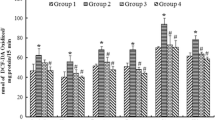
Lead (Pb) is known to disrupt the pro-oxidant/antioxidant balance of tissues, which leads to biochemical and physiological dysfunction. Oxidative stress is considered a possible molecular mechanism involved in Pb neurotoxicity. Considering the vulnerability of the brain to oxidative stress under Pb neurotoxicity, this study investigated the effects of exposure of the thiol antioxidant N-acetylcysteine (NAC) on lead-induced oxidative damage and lipid peroxidation in brain regions of the rat. Wister strain rats were exposed to lead in the form of lead acetate (20 mg/kg body wt/d) for a period of 2 wk and the effects of NAC on lead-induced neurotoxicity in rat brain regions were assessed by postadministration of NAC (160 mg/kg body wt/d) for a period of 3 wk. The lipid peroxidation byproduct, malondialdehyde (MDA) increased following lead exposure in both of the regions, and the antioxidant capacities of the cell in terms of the activity of antioxidant enzymes superoxide dismutase (SOD) and catalase (CAT) was diminished. Following NAC treatment, lead-induced lipid peroxidation decreased and antioxidant enzyme activities improved, with CAT showing enhancement in the cerebral region only and SOD showing enhancements in the cerebellar region. Our result suggests that thiol-antioxidant supplementation following Pb exposure might enhance the reductive status of brain regions by arresting the lipid peroxidative damage in brain regions.
Similar content being viewed by others
References
J. P. Bressler and G. W. Goldstein, Mechanism of lead neurotoxicity, Biochem. Pharmacol. 41, 479–484 (1991).
D. Behne, H. Hilmert, S. Scheid, et al., Evidence for specific selenium target tissues and new biologically important selenoproteins, Biochim. Biophys. Acta 966, 12–21 (1988).
T. D. Buckman, M. S. Sutphin, and C. D. Eckhert, A comparison of the effects of dietary selenium on selenoprotein expression in rat brain and liver, Biochim. Biophys. Acta 1163, 176–184 (1993).
K. Haung, E. Lauridsen, and J. Clausen, The uptake of Na-selenite in rat brain. Localization of new glutathione peroxidases in the rat brain, Biol. Trace Element Res. 46, 91–102 (1994).
N. Ercal, P. Treeratphan, P. Lutz, et al., N-Acetylcysteine protects Chinese hamster ovary (CHO) cells from lead induced oxidative stress, Toxicology 108, 57–64 (1996).
O. I. Aruoma, B. Halliwell, B. M. Hoey, et al., The antioxidant action of N-acetylcysteine: its reaction with hydrogen peroxide, hydroxyl radical, superoxide, and hypochlorous acid, Free Radical Biol. Med. 6, 593–597 (1989).
E. D. Wills, Mechanism of lipid peroxide formation in animal tissues, Biochem. J. 99, 667–676 (1966).
Y. Kono, Generation of superoxide radical during auto oxidation of hydroxylamine and an assay for superoxide dismutase, Arch. Biochem. Biophys. 186, 189–195 (1978).
H. Luck, Methods in Enzymatic Analysis, Academic, New York, pp. 885–893 (1971).
O. H. Lowry, N. J. Rosebrough, A. L. Farr, et al., Protein measurement with the Folin-phenol reagent, J. Biol. Chem. 193, 265–275 (1951).
V. N. Adonalyo and P. L. Oteiza, Lead inoxication: antioxidant defense and oxidative damage in rat brain, Toxocology 135, 77–85 (1999).
L. J. Machlin and A. Bendich, Free radical tissue damage: protective role of antioxidants nutrients, FASEB J. 4, 441–445 (1987).
S. U. Rehman, Lead induced regional lipid peroxidation in brain, Toxicol. Lett. 21, 333–337 (1984).
R. Neal, K. Cooper, H. Gurer, et al., Effects of N-acetylcysteine and 2,3-dimercaptosuccinic acid on lead induced oxidative stress in rat lenses, Toxicology 130, 167–174 (1998).
R. Sandhir, D. Julka, and K. D. Gill, Lipoperoxidative damage on lead exposure in rat brain and its implications on membrane bound enzymes, Pharmacol. Toxicol. 74, 66–71 (1994).
B. L. Vallee and D. D. Ulmer, Biochemical effects of mercury, cadmium and lead, Annu. Rev. Biochem. 41, 91–128 (1972).
I. Fridovich, Superoxide radical as an endogenous toxicant, Annu. Rev. Pharmacol. Toxicol. 23, 239–257 (1983).
A. A. Mylorie, H. Collins, C. Umbles, et al., Erythrocyte superoxide dismutase activity and other parameters of coper status in rats ingesting lead acetate, Toxicol. Appl. Pharmacol. 82, 512–520 (1986).
E. I. Dressel and J. E. Falk, Studies on the biosynthesis of blood pigment: haem synthesis in hemolysed erythrocytes of chicken blood, Biochem. J. 56, 156–163 (1954).
V. Etus, N. Gazioglu, and A. Belce, N-Acetylcysteine reduces cerebral lipid peroxidation in a rat model of infantile hydrocephalus, J. Neurolog. Sci. (Turkish), 18 (2001).
A. Pastor, P. S. Collado, M. Almar, et al., Antioxidant enzyme status in biliary obstructed rats: effects of N-acetylcysteine, J. Hepatol. 27, 363–370 (1997).
P. C. Mangal and N. Gulati, Trace elements in animal feed and animal tissues; a correlational study by neutron activation method, Indian J. Exp. Biol. 19, 441–444 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nehru, B., Kanwar, S.S. N-acetylcysteine exposure on lead-induced lipid peroxidative damage and oxidative defense system in brain regions of rats. Biol Trace Elem Res 101, 257–264 (2004). https://doi.org/10.1385/BTER:101:3:257
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/BTER:101:3:257