Abstract
Objective
To compare the long-term efficacy and shortterm escape from effective treatment of 0.005% latanoprost administered once daily vs 0.5% timolol plus 2% dorzolamide twice daily for 24 months to patients with open-angle glaucoma.
Design
Prospective study.
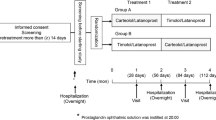
Setting and number of patients
A total of 117 eyes of 61 subjects with open-angle glaucoma were included in this study. The eligible patients were randomized to receive either 0.005% latanoprost once daily or 0.5% timolol and 2% dorzolamide twice daily.
Measurements
The intraocular pressure (IOP) figures were recorded at baseline and at 0.5 1, 3, 6, 9, 12, 15, 18, 21, and 24 months of treatment.
Results
Compared with baseline measurements, both latanoprost and timolol plus dorzolamide treatments caused a significant (p<0.001) reduction of IOP at each visit trroughout the duration of therapy. The IOP was reduced to 17.1±2.4, 16.6±2.2, and 15.9±2.04 with latanoprost and 18.2±2.1, 18.6±2.4, and 18.4±1.9 with timolol plus dorzolamide after 6, 12, and 24 months, respectively. After 24 months, 82% of the patients who initially responsed to treatment were still controlled in the latanoprost group, with a maintained reduction of IOP and no signs of upward drift. However, only 56% of the patients of the timolol plus dorzolamide group were controlled at the end of the study with increase of IOP figures.
Conclusions
Administered once daily, 0.005% latanoprost is more effective than timolol plus dorzolamide twice daily for the long-term treatment of patients with open-angle glaucoma, showing a maintained effect on IOP reduction.
Similar content being viewed by others
References
Giuffré G. The effects of Prostaglandin F2α in human eye. Graefe’s Arch Clin Exp Ophthalmol. 1985;222:139–141.
Gabelt BT, Kaufman PL. Prostaglandin F2α increases uveoscleral outflow in the cynomolgus monkey. Exp Eye Res. 1989;49: 389–402.
Nilsson SPE, Samuelson M, Bill A, et al. Increased uveoscleral out-flow as a possible mechanism of ocular hypotension caused by prostaglandin F2α-1-isopropyl ester in the cynomolgus monkey. Exp Eye Res. 1989;48:707–716.
Toris CB, Camras CB, Yablosnko ME. Effects of PhXA41, a new prostaglandin F2α analog on aqueous humor dynamics in human eyes. Ophthalmology. 1993;100:1297–1304.
Lindsey JD, Kashiwagi K, Kashiwagi F, Weinreb RN. Prostaglandins alter extracellular matrix adjacent to human ciliary muscle cells in vitro. Invest Ophthalmol Vis Sci. 1997;38:2214–2223.
Alm A, Stjernschantz J. Effects on intraocular pressure and side effects of 0.005% Latanoprost applied once daily, evening or morning. Ophthalmology. 1995;12:743–752.
Watson P, Stjernschantz J. A six month, randomized, double-masked study comparing Latanoprost with Timolol in open angle glaucoma and ocular hypertension. Ophthalmology. 1996;103:126–137.
Mishima HK, Masuda K, Kitazawa Y, Azuma I, Araie M. A comparison of Latanoprost and Timolol in primary open-angle glaucoma and ocular hypertension. Arch Ophthalmol. 1996;114: 929–932.
Camras CB, for the United States Latanoprost Study Group. Comparison of Latanoprost and Timolol in patients with ocular hypertension and glaucoma: a six-month, masked muticenter trial in the United States. Ophthalmology. 1996;103:138–147.
Alm A, Widengard I, Kjellgren D, et al. Latanoprost administered once daily causes a maintained reduction of intraocular pressure in glaucoma patients treated concomitantly with Timolol. Br J Ophthalmol. 1994;76:12–16.
Alm A, Widengard I, Mapea O. Combination of latanoprost with dipivefrin in patients with open angle glaucoma or ocular hypertension. Proceedings of the 27th International Congress of Opthalmology, Toronto, Canada. 1994.
Watson PG, Barnett F, Parker V. The Additive Effects of PhXA41 and Propine in Patients with Primary Open Angle Glaucoma. Oxford, England: 1992.
Rulo AH, Greve EL, Hoyng PF. Additive effect of Latanoprost, a prostaglandin F2a analogue, and Timolol in patients with elevated intraocular pressure. Br J Ophthalmol. 1994;78:899–902.
Rulo AH, Greve EL, Hoyng PFJ. Additive ocular hypotensive effect of Latanoprost and Acetazolamide. Ophthalmology. 1997;104: 1503–1507.
Friström B, Nilsson SEG. Interaction of PhXA41, a new prostaglandin analogue, with pilocarpine. A study on patients with elevated intraocular pressure. Arch Ophthalmol. 1993;111:662–665.
McClellan K. Topical eye preparations: optimum use. Curr Ther. 1995;36:61–66.
Capino DG, Leibowitz HM. Glaucoma screening, diagnosis, and therapy. Hosp Pract. 1990;25:73–91.
Coyle D, Drummond M. The economic burden of glaucoma in the UK. The need for a far-sighted policy. PharmacoEconomics. 1995;7: 484–489.
Sherwood MB, Migdal CS, Hitchings RA, et al. Initial treatment of glaucoma; surgery or medications (review). Surv Ophthalmol. 1993; 37:293–305.
Hotehama Y, Mishima HK, Kitazawa Y, Masuda K. Ocular hypotensive effect of PhXA41 in patients with ocular hypertension or primary open-angle glaucoma. Jpn J Ophthalmol. 1993;37:270–274.
Ziai N, Dolan JW, Kacere RD, Brubaker RF. The effects on aqueous dynamics of PhX41, a new prostaglandin F2α analogue, after topical application in normal and ocular hypertensive human eyes. Arch Ophthalmol. 1993;111:1351–1358.
Watson PG, Latanoprost. Two years experience of its use in the United Kingdom. Ophthalmology. 1998;105:83–88.
Hedman K, Alm A. The effect of latanoprost on intraocular pressure up to 24 months’ treatment. Proceedings of the 28th International Congress of Ophthalmology, Amsterdam, Holland, 1998.
Author information
Authors and Affiliations
Additional information
The authors have stated that they do not have a significant financial interest or other relationship with any product manufacturer or provider of services discussed in this article. The authors do, however, discuss the use of off-label products, which includes unlabeled, unapproved, or investigative devices.
Rights and permissions
About this article
Cite this article
Polo, V., Larrosa, J.M., Ferreras, A. et al. Latanoprost vs combined therapy with timolol plus dorzolamide in open-angle glaucoma a 24-month study. Ann Ophthalmol 37, 33–36 (2005). https://doi.org/10.1385/AO:37:1:033
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/AO:37:1:033