Abstract
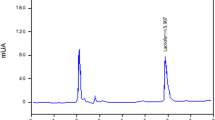
Lactoferrin (Lf), a protein found in human and bovine milk, tears, blood, and other secretory fluids, has been used to prevent infection from potential microbial pathogens by its ability to bind with iron (Fe3+). Currently, bovine lactoferrin can be purified from milk using ion exchange resin, which is a costly procedure making lactoferrin expensive. The purpose of this work was to investigate a low-cost foam fractionation process as the first step in separating lactoferrin from milk.
Similar content being viewed by others
References
Schryvers, A. B., Bonnah, R., Yu, R. H., Wong, H., and Retzer, M., (1998), Adv. Exp. Med. Biol. 443, 123–133.
Yoo, Y. C., Watanabe, S., Watanabe, R., Hata, K., Shimazaki, K., and Azuma, I. (1998), Adv. Exp. Med. Biol. 443, 285–291.
Saito, H. (1994), Adv. Exp. Med. Biol. 357, 219.
Loha, V. (1999), PhD Dissertation, Vanderbilt University, Nashville, TN.
Ko, S., Tanner, R. D., and Prokop, A. (2002) Appl. Biotechnol. Bioprocess. Engin. 6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noel, J., Prokop, A. & Tanner, R.D. Foam fractionation of a dilute solution of bovine lactoferrin. Appl Biochem Biotechnol 98, 395–402 (2002). https://doi.org/10.1385/ABAB:98-100:1-9:395
Issue Date:
DOI: https://doi.org/10.1385/ABAB:98-100:1-9:395