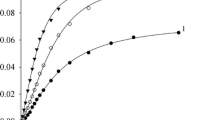
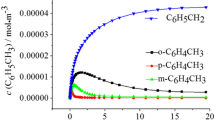
Abstract
Toluene 4-monooxygenase is a four-protein component diiron enzyme complex. The enzyme catalyzes the hydroxylation of toluene to give p-cresol with ∼96% regioselectivity. The performance of the enzyme in two-phase reaction systems consisting of toluene, hexane, or perfluorohexane and an aqueous buffer was tested. In each of the cosolvent systems, containing up to 93% (v/v) of solvent, the enzyme was active and exhibited regioselectivity indistinguishable from the aqueous reaction. Using the perfluorohexane/buffer system, a number of polycyclic aromatic hydrocarbons were oxidized that were not readily oxidized in aqueous buffer. An instability of the hydroxylase component and a substantial uncoupling of NADH utilization and product formation were observed in reactions that were continued for longer than ∼3 min. More stable enzyme complexes will be needed for broad applicability of this hydroxylating system in nonaqueous media.
Similar content being viewed by others
References
March, J. (1992), Advanced Organic Chemistry, 4th ed., John Wiley & Sons, New York.
Wubbolts, M. G., Reuvekamp, P., and Witholt, B. (1994), Enzyme Microb. Technol. 16, 608–615.
Harayama, S. (1997), Curr. Opin. Biotechnol. 8, 268–273.
Guengerich, F. P. (1997), Adv. Pharmacol. 43, 7–35.
Kellner, D. G., Maves, S. A., and Sligar, S. G. (1997), Curr. Opin. Biotechnol. 8, 274–278.
Fox, B. G. (1997), in Comprehensive Biological Catalysis, Sinnott, M., ed., Academic, London, pp. 261–348.
Pikus, J. D., Studts, J. M., Achim, C., Kauffmann, K. E., Münck, E., Steffan, R. J., McClay, K., and Fox, B. G. (1996), Biochemistry 35, 9106–9119.
Yen, K.-M., Karl, M. R., Blatt, L. M., Simon, M. J., Winter, R. B., Fausset, P. R., Lu, H. S., Harcourt, A. A., and Chen, K. K. (1991), J. Bacteriol. 173, 5315–5327.
Yen, K.-M. and Karl, M. R. (1992), J. Bacteriol. 174, 7253–7261.
Pikus, J. D., Studts, J. M., McClay, K., Steffan, R. J., and Fox, B. G. (1997), Biochemistry 36, 9283–9289.
Pikus, J. D., Mitchell, K. H., Studts, J. M., McClay, K., Steffan, R. J., and Fox, B. G. (2000), Biochemistry 39, 791–799.
Vannelli, T. and Hooper, A. B. (1995), Biochemistry 34, 11,743–11,749.
Miller, V. P., Tschirret-Guth, R. A., and Ortiz de Montellano, P. R. (1995), Arch. Biochem. Biophys. 319, 333–340.
Hanzlik, R. P. and Ling, K.-H. J. (1993), J. Am. Chem. Soc. 115, 9363–9370.
Tassaneeyakul, W., Birkett, D. J., Edwards, J. W., Veronese, M. E., Tassaneeyakul, W., Tukey, R. H., and Miners, J. O. (1996), J. Pharmacol. Exp. Ther. 276, 101–108.
Dalton, H. (1980), Adv. Appl. Microbiol. 26, 71–87.
Xia, B., Pikus, J. D., Xia, W., McClay, K., Steffan, R. J., Chae, Y. K., Westler, W. M., Markley, J. L., and Fox, B. G. (1998), Biochemistry 38, 727–739.
Horvath, A. L. (1982), Halogenated Hydrocarbons: Solubility-Miscibility with Water, Marcel Dekker, New York.
Englard, S. and Seifter, S. (1990), Methods Enzymol. 182, 285–300.
Dordick, J. S., Khmelnitsky, Y. L., and Sergeeva, M. V. (1998), Curr. Opin. Microbiol. 19, 103–112.
Chen, K. Q. and Arnold, F. H. (1991), Biotechnology (NY) 9, 1073–1077.
Gupta, M. N. (1992), Eur. J. Biochem. 203, 25–32.
Arnold, F. H. (1990), Trends Biotechnol. 8, 244–249.
Blinkovsky, A. M., Martin, B. D., and Dordick, J. S. (1992), Curr. Opin. Biotechnol. 3, 124–129.
Hamamura, N., Page, C., Long, T., Semprini, L., and Arp, D. J. (1997), Appl. Environ. Microbiol. 63, 3607–3613.
Jiang, Y. and Dalton, H. (1994), Biochim. Biophys. Acta 1201, 76–84.
Clark, T. R. and Roberto, F. F. (1996), Appl. Microbiol. Biotechnol. 45, 658–663.
Loida, P. J. and Sligar, S. G. (1993), Biochemistry 32, 11,530–11,538.
Atkins, W. M. and Sligar, S. G. (1987), J. Am. Chem. Soc. 109, 3754–3760.
Chenault, H. K. and Whitesides, G. M. (1987), Appl. Biochem. Biotechnol. 14, 147–197.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oppenheim, S.F., Studts, J.M., Fox, B.G. et al. Aromatic hydroxylation catalyzed by toluene 4-monooxygenase in organic solvent/aqueous buffer mixtures. Appl Biochem Biotechnol 90, 187–197 (2001). https://doi.org/10.1385/ABAB:90:3:187
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:90:3:187