Abstract
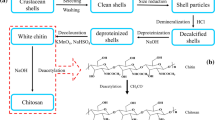
Urease from pigeonpea (Cajanus cajan L.) was covalently linked to crab shell chitosan beads using glutaraldehyde. The optimum immobilization (64% activity) was observed at 4°C, with a protein concentration of 0.24 mg/bead and 3% glutaraldehyde. The immobilized enzyme stored in 0.05 M Trisacetate buffer, pH 7.3, at 4°C had a t 1/2 of 110 d. There was practically no leaching of enzyme (<3%) from the immobilized beads in 30 d. The immobilized urease was used 10 times at an interval of 24 h between each use with 80% residual activity at the end of the period. The chitosan-immobilized urease showed a significantly higher Michaelis constant (8.3 mM) compared to that of the soluble urease (3.0 mM). Its apparent optimum pH also shifted from 7.3 to 8.5. Immobilized urease showed an optimal temperature of 77°C, compared with 47°C for the soluble urease. Time-dependent kinetics of the thermal denaturation of immobilized urease was studied and found to be monophasic in nature compared to biphasic in nature for soluble enzyme. This immobilized urease was used to analyze blood urea of some of the clinical samples from the clinical pathology laboratories. The results compared favorably with those obtained by the various chemical/biochemical methods employed in the clinical pathology laboratories. A column packed with immobilized urease beads was also prepared in a syringe for the regular and continuous monitoring of serum urea concentrations.
Similar content being viewed by others
References
Das, N., Kayastha, A. M., and Malhotra, O. P. (1998), Biotechnol. Appl. Biochem. 27, 25–29.
Alexakis, T., Boadi, D. K., Quong, D., Groboillot, A., O’Neill, I., Poncelet, D., and Neufeld, R. J. (1995), Appl. Biochem. Biotechnol. 50, 93–106.
Tanaka, H., Matsumara, M., and Veliky, I. A. (1984), Biotechnol. Bioeng. 26, 53–58.
Braun, J., Le Chanu, P., and Le Goffic, F. (1989), Biotechnol. Bioeng. 33, 242–246.
Hirano, S. and Miura, O. (1979), Biotechnol. Bioeng. 21, 711–714.
Knoor, D. and Miazaga, M. (1985), Food Technol. 39, 139.
Holst, O. and Mattiasson, B. (1991), Biosens. Bioelectron. 6, 101–108.
Lee, K. B., Boadi, D. K., and Neufeld, R. J. (1995), J. Theor. Biol. 175, 295–303.
Nosé, Y. (1990), Artif. Organs 14, 245.
Thavarungkul, P., Håkanson, H., Holst, O., and Mattiasson, B. (1991), Biosens. Bioelectron. 6, 101–107.
Wang, Y. J., Chen, C. H., Hsiue, G. H., and Yu, B. C. (1992), Biotechnol. Bioeng. 40, 446–449.
Orsonneau, J.-L., Massoubre, C., Cabanes, M., and Lustenberger, P. (1992), Clin. Chem. 38, 357–363.
Das, N., Prabhakar, P., Kayastha, A. M., and Srivastava, R. C. (1997), Biotechnol. Bioeng. 54, 619–623.
Srivastava, P. K., Kayastha, A. M. and Srinivasan (2001), Biotechnol. Appl. Biochem. 34, 55–62.
Das, N. and Kayastha, A. M. (1998), World J. Mirobiol. Biotechnol. 14, 927–929.
Onyezili, F. N. (1988), J. Biochem. Biophys. Methods 16, 255–262.
Abdel, L. M. S. and Guibault, G. G. (1990), J. Biotechnol. 14, 53–62.
Kamath, N., Melo, J. S., and D’Souza, S. F. (1988), Appl. Biochem. Biotechnol. 19, 251–258.
Kamath, N. and D’Souza, S. F. (1991), Enzyme Microbiol. Technol. 13, 935–938.
Kayastha, A. M., Das, N., and Malhotra, O. P. (1995), in Biopolymers and Bioproducts: Structure, Function and Applications, Svasti, J., Rimphanitchayakit, V., Tassanakajorn, A., et al., eds., Dokya, Bangkok, pp. 382–386.
Carrara, C. R. and Rubiolo, A. C. (1994), Biotechnol. Prog. 10, 220–224.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951), J. Biol. Chem. 193, 265–280.
Iyengar, L., Bajpai, P., and Rao, A. V. S. P. (1982), Indian J. Biochem. Biophys. 19, 130–134.
Mosbach, K. (1971), Sci. Am. 224, 26–33.
Moynihan, H. J., Lee, C. K., Clarck, W., and Wang, N.-H. L. (1989), Biotechnol. Bioeng. 34, 951–963.
Weetall, H. H. and Hersh, L. S. (1969), Biochem. Biophys. Acta 185, 464, 465.
Guibault, G. G. and Das, J. (1970), Anal. Biochem. 33, 341–355.
May, S. W. and Li, N. N. (1972), Biochem. Biophys. Res. Commun. 47, 1179–1185.
Stanley, W. L., Watters, G. G., Keiley, G., Chan, B. G., Goribaldi, J. A., and Schade, J. E. (1976), Biotechnol. Bioeng. 18, 439–445.
Sundaram, P. V. and Hornby, W. E. (1970), FEBS Lett. 10, 325–329.
Kayastha, A. M. and Das, N. (1998), J. Plant Biochem. Biotechnol. 7, 121–124.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kayastha, A.M., Srivastava, P.K. Pigeonpea (Cajanus cajan L.) urease immobilized on glutaraldehyde-activated chitosan beads and its analytical applications. Appl Biochem Biotechnol 96, 41–53 (2001). https://doi.org/10.1385/ABAB:96:1-3:041
Issue Date:
DOI: https://doi.org/10.1385/ABAB:96:1-3:041