Abstract
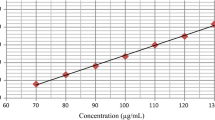
A simple and reliable liquid chromatographic method has been developed and validated for the determination of cefdinir in human urine and capsule samples. A chromatographic separation was achieved on a C18 column using a mobile phase consisting of potassium dihydrogen phosphate (10 mM, pH 4.5)–acetonitrile (90:10, v/v). Quantitation was achieved with UV detection at 285 nm, based on peak area with linear calibration curve at a concentration range of 0.7–39 µg mL−1. This method was successfully applied for the establishment of an urinary excretion pattern after oral dose.



Similar content being viewed by others
References
Sweetman SC, Martindale (2005) The complete drug reference, 35th edn. Pharmaceutical Press, London
Mehta TN, Subbaiah G, Pundarikakshudu K (2005) J AOAC Int 88:1661–1665
Okamoto Y, Itoh K, Namiki Y, Matsushita J, Fujioka M, Yasuda T (1996) J Pharm Biomed Anal 14:739–748. doi:10.1016/0731-7085(95)01687-2
Rajeev J, Ashish D, Ritesh M (2008) J Colloid Interface Sci 318:296–301. doi:10.1016/0731-7085(95)01687-2
Chen ZJ, Zhang J, Yu JC, Cao GY, Wu XJ, Shi YG (2006) J Chromatogr B 834:163–169. doi:10.1016/j.jchromb.2006.02.047
Berzas Nevado JJ, Guiberteau Cabanillas C, Villaseñor Llerena MJ, Rodríguez Robledo V (2005) J Chromatogr A 1072:249–257. doi:10.1016/0731-7085(95)01687-2
Draper NR, Smith H (1981) Applied regression analysis. Wiley, New York, pp 22–40
The European Agency for the Evaluation of Medical Products (1996) ICH topic Q2B note for guidance on validation of analytical procedures: methodology GPMP/ICH/281/95. IFPMA, Geneva, pp 1–8
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hadad, G.M., Emara, S. & Mahmoud, W.M.M. Optimization and Validation of an LC Method for the Determination of Cefdinir in Dosage Form and Human Urine. Chroma 70, 1593–1598 (2009). https://doi.org/10.1365/s10337-009-1365-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-009-1365-8