Abstract
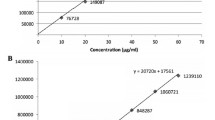
A new method has been developed to determine clonidine hydrochloride in Zhenju Jiangya Tablets using ion-pair HPLC coupled with neutral column chromatography in this paper. Clonidine hydrochloride is an active component and occurs in very low amounts. The sample was purified by neutral Al2O3 column chromatography instead of liquid–liquid extraction and measured by reversed phase ion-pair chromatography with 1-heptanesulfonate at acidic pH with UV-detection. Result showed that clonidine hydrochloride had good linearity in the range of 1.1–11.0 μg mL−1 (r = 0.9993) and the average recoveries were 96.5–99.3% (RSD 2.1–4.8%).



Similar content being viewed by others
References
Bunca G, Kovacic S, Strnad S (2003) Auton Neurosci Basic Clin 105:71–76
Qi XS, Miller RB, Namiki Y, Zhang J, Jacobus R (1997) J Pharm Biomed Anal 16:413–418
Boswell G, Bekersky I, Mekki Q, Eisenach J (1997) Clin Ther 19:1024–1030
Rossi S, Yaksh T (2003) J Pharm Biomed Anal 31:243–250
ChP (Vol II) (2000) Chinese Pharmacopoeia, Chinese Pharmacopoeial Convention, Beijing
Classen AM, Wimbish GH, Kupiec TC (2004) J Pain Symptom Manag 28:603–611
ChP (Vol II) (2005), Chinese Pharmacopoeia, Chinese Pharmacopoeial Convention, Beijing
Liao B, Cong X (2003) J China Pharm Univ 34:38–40
Yu J, Ye XL (2004) Chin Remedies and Clinics 9:715–716
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wei, YQ., Zou, HB., Ma, Y. et al. LC Determination of Clonidine Hydrochloride in Chinese and Western Combine Compound Hypotensor Tablets of Zhenju Jiangya. Chroma 64, 215–218 (2006). https://doi.org/10.1365/s10337-006-0011-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-006-0011-y