Abstract
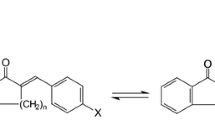
In organic synthesis, the purification of reactional intermediates or final products is generally carried out by normal phase flash chromatography. However, for dihydrodipyridopyrazines, a new family of antitumor agents, the efficiency of this technique is too low to achieve the purification of the isomer mixture. Purification of the DHDPP isomer mixture has therefore been studied using preparative liquid chromatography. With the stationary phase used in flash chromatography, PLC provides greater efficiency and allows to increase the mobile phase flow rate. A complete study of preparative purification was performed, including that of compounds solubility and analytical optimization. This work has allowed to lower the overlap between the two DHDPP isomers, to greatly reduce the total duration of the process, to increase the purified quantity per run and consequently to greatly improve the throughput of the purification. In addition, this technique can be easily and totally automated. Concurrently, another purification method (centrifugal partition chromatography ), based on acidic constants difference of the two components in two immiscible liquids, was developed. CPC has demonstrated its ability to separate the two DHDPP isomers. Finally, the economic aspects of PLC and CPC results are compared.
Similar content being viewed by others
References
Colin H, Hilaireau P (1991) Pharmaceutical Manufacturing International 143–145
Rodriguez I, Kuehm-Caubère C, Vinter-Pasquier K, Renard P, Pfeiffer B, Caubère P (1998) Tetrahedron Lett 39:7283–7286
Blanchard S, Guillaumet G, Kuehm-Caubère C, Atassi G, Renard P, Caubère P, Pfeiffer B, Pierre A, Rodriguez I, Vinter-Pasquier K (2000) U.S. Patent No. 6,127,369
Denny WA, Baguley BC, Cain BF, Waring MJ (1983) In: Antitumor acridines. Molecular aspects of anti-cancer drug action, Neidle S, Warning MJ (eds) Verlag Chemie, England, pp.1–34
Brãna MF, Castellano JM, Morán M, Pérez de Vega MJ, Romerdahl CR, Qian XD, Bousquet P, Emling F, Schlick E, Keilhauer G (1993) Anti-Cancer Drug Des 8:257–268
Garbay-Jaureguiberry C, Barsi MC, Jacquemin-Sablon A, Le Pecq JB, Roques BP (1992) J Med Chem 35:72–81
Leng F, Priebe W, Chaires JB (1998) Biochemistry 37:1742–1753
Hernandez L, Cholody WM, Hudson EA, Resau JH, Pauly G, Michejda CJ (1995) Cancer Res 55:2338–2345
Foucault AP (1995) In: Centrifugal Partition Chromatography, Chromatographic Science Series, Volume 68, Marcel Dekker, New York.
Ito Y, Ma Y (1996) J Chromatogr A 753:1–36
Renold P, Madero E, Maetzke T (2001) J Chromatogr A 908:143–148
Meyer VR (1984) J Chromatogr 316:113–124
Sarker M, Katti AM, Guiochon G (1996) J Chromatogr A 719:275–289
Koh JH, Broyles BS, Guan-Sajonz H, Hu MZC, Guiochon G (1998) J Chromatogr A 813:223–238
Knox JH, Pyper HM (1986) J Chromatogr 363:1–30
Gareil P, Rosset R (1988) J Chromatogr 450:13–25
Guiochon G, Golshan-Shirazi S, Katti AM (1994) In: Fundamentals of Preparative and Nonlinear Chromatography, Academic Press
Viron C, Pennanec R, André P, Lafosse M (2000) J Liq Chrom & Rel Technol 23 (11):1681–1688
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Himbert, F., Pennanec, R., Guillaumet, G. et al. Preparative Liquid Chromatography and Centrifugal Partition Chromatography for Purification of New Anticancer Precursors. Chromatographia 60, 269–274 (2004). https://doi.org/10.1365/s10337-004-0303-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-004-0303-z