Abstract
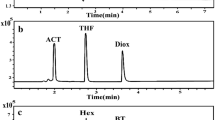
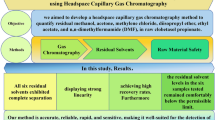
A knowledge base for determining residual solvents in pharmaceuticals has been established using static headspace gas chromatography (HS-GC). Fifty-one solvents which belong to four different classes according to the guideline for residual solvents regulated by the International Conference on Harmonization were studied. Two columns with opposite polarity, SPB-1 and HP-FFAP, were used and the relative retention time was the qualitative parameter in the knowledge base. With the aid of the knowledge base, unknown residual solvents in pharmaceuticals can be identified promptly and suitable methods for determining residual solvents can be developed rapidly.
Similar content being viewed by others
References
United States Pharmacopoeia (2002) In: US Pharmacopoeia 25 and National Formulary 20, USPC Inc., Rockville
British Pharmacopoeia Commission Office (2002) In: British Pharmacopoeia 2002, The Stationary Office, London
European Pharmacopoeia Commission (2002) In: European Pharmacopoeia 4th Edition, Druckerei C.H.Beck, Germany
Society of Japanese Pharmacopoeia (2001) In: Japanese Pharmacopoeia 14th Edition, Shibuya, Tokyo
State Pharmacopoeia Commission of P.R.China (2000) In: Pharmacopoeia of the People’s Republic of China, Chemical Industry Press, Beijing
Li Z, Han Y-H, Martin G.P (2002) J Pharm Biomed Anal 28:673–682
Camarasu CC (2002) Chromatographia 56 slet: s137–s143
Naddaf A, Balla J (2000) Chromatographia, 51 slet: s241–s245
Russo MV (1994) Chromatographia 39:645–648
Clayton B (1988) J Chromatogr 438: 103–107
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, L., Hu, Cq. & Yin, Lh. Establishment of a Knowledge Base for Prescreening Residual Solvents in Pharmaceuticals. Chromatographia 59, 475–480 (2004). https://doi.org/10.1365/s10337-004-0211-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-004-0211-2