Abstract
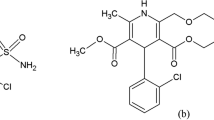
This paper describes a new, simple, precise, and accurate TLC method for simultaneous quantitation of amlodipine (AML) besylate and valsartan (VAL) as the bulk drug and in tablet dosage forms. Chromatographic separation of the drugs was performed on aluminum plates precoated with silica gel 60 F 254 as the stationary phase and the solvent system consisted of toluene:methanol:acetic acid 7:3:0.1 (v/v/v). Densitometric evaluation of the separated zones was performed at 244 nm. The two drugs were satisfactorily resolved with R F values of 0.41 ± 0.02 and 0.54 ± 0.02 for AML and VAL, respectively. The accuracy and reliability of the method was assessed by evaluation of linearity (100–600 ng spot−1 for AML and 1,600–9,600 ng spot−1 for VAL), precision (intra-day RSD 1.51–1.83% and inter-day RSD 1.24–1.95% for AML, and intra-day RSD 0.14–0.39% and inter-day RSD 0.19–0.52% for VAL), accuracy (98.29 ± 1.16% for AML and 98.72 ± 0.50% for VAL), and specificity, in accordance with ICH guidelines.

Similar content being viewed by others
References
European Pharmacopoeia (2005) Directorate for the Quality of Medicines of the Council of Europe, vol II, 5th edn. pp 981–982
O’Neil MJ (2006) The Merck index, 14th edn. Merck Research Laboratories, Whitehouse Station, pp 1705
Streel B, Laine C, Zimmer C, Sibenaler R, Ceccato A (2002) J Biochem Biophys Methods 54:357–368. doi:10.1016/S0165-022X(02)00133-1
Naidu KR, Kale UN, Shingare MS (2005) J Pharm Biomed Anal 39:147–155. doi:10.1016/j.jpba.2005.04.001
Kamat K, Chaturvedi SC (2005) Indian J Pharm Sci 67:236–239
Rajeswari KR, Sankar GG, Rao AL, Seshagirirao JVLN (2006) Indian J Pharm Sci 68:275–277
Mahadik KR, Aggrawal H, Kaul N (2004) Indian Drugs 41:32–35
Meyyanathan SN, Suresh B (2005) J Chromatogr Sci 43:73–75
Gonzalez L, Lopez JA, Alonso RM, Jimenez RM (2002) J Chromatogr A 949:49–60. doi:10.1016/S0021-9673(01)01496-0
Francotte E, Davatz A, Richert P (1996) J Chromatogr B Biomed Appl 686:77–83. doi:10.1016/S0378-4347(96)00242-3
Satana E, Altinay S, Goger NG, Ozkan SA, Senturk Z (2001) J Pharm Biomed Anal 25:1009–1013. doi:10.1016/S0731-7085(01)00394-6
ICH Q2(R1) (2005) Validation of analytical procedures: text and methodology. International conference on harmonization, Geneva
Acknowledgments
The authors would like to thank, Cipla Ltd (Patalganga, Maharashta, India) and Torrent Pharmaceuticals Ltd (Ahmadabad, Gujarat, India) for providing a gift sample of standard amlodepine besylate and valsartan. The authors would like to thank, Dr. K. R. Mahadik, Principal, Poona College of Pharmacy, Pune, India for providing necessary facilities to carry out the work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhaneshwar, S.R., Patre, N.G. & Mahadik, M.V. Validated TLC Method for Simultaneous Quantitation of Amlodipine Besylate and Valsartan in Bulk Drug and Formulation. Chroma 69, 157–161 (2009). https://doi.org/10.1365/s10337-008-0858-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-008-0858-1