Abstract
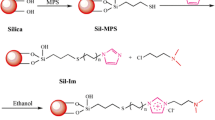
A novel 1,3-alternate 25,27-bis-[p-nitrobenzyloxy]-26,28-bis-[3-propyloxy]-calix[4]arene-bonded silica gel stationary phase has been prepared and used for separating various selected analytes by HPLC. The effect of organic modifier and pH of the mobile phase on retention and selectivity were studied using aromatic positional isomers as an example. Application examples have been provided for separation of alkylbenzenes, PAHs, xanthine derivatives, and purine and pyrimidine bases. A selectivity comparison of the novel phase versus CalixBz and Backerbond PhenylEthyl phases has been performed.









Similar content being viewed by others
References
He L, Toh Ch-S (2006) Anal Chim Acta 556:1–15
Gutsche CD (1989) In: Scoddatr JF (ed) Calixarenes, monographs in supramolecular chemistry. The Royal Society of Chemistry, Cambridge
Gutsche CD (1998) In: Scoddatr JF (ed) Calixarenes revisited, monographs in supramolecular. The Royal Society of Chemistry, Cambridge
Vincens J (1991) In: Böhmer V (ed) Calixarenes. A versatile class of macrocyclic compounds. Topics in inclusion science. Kluwer, Dordrecht
Vincens J, Asfari Z (1994) In: Harrowfield JM (ed) Calixarenes 50th anniversary: commemorative issue. Kluwer, Dordrecht
Asfari Z, Böhmer V, Harrowfield J, Vicens J (2001) In: Vicens J (ed) Calixarenes. Kluwer, Dordrecht
Ludwig R (2000) Fresenius J Anal Chem 367:103–128
Ludwig R (2005) Microchim Acta 152:1–19
Milbrady R, Böhmer V (2001) Calixarenes as stationary phases. In: Vicens J (ed) Calixarenes. Kluwer, Dordrecht, pp 663–674
Meyer R., Jira T (2007) Curr Anal Chem 3:161–170
Śliwka-Kaszyńska M (2007) Crit Rev in Anal Chem 37:211–224
Krawinkler KH, Maier NM, Sajovic E, Lindner W (2004) J Chromatogr A 1053:119–131
Sokoliess T, Opolka A, Menyes U, Roth U, Jira T (2002) Pharmazie 57:589–590
Śliwka-Kaszyńska M, Jaszczołt K, Witt D, Rachoń J (2004) J Chromatogr A 1055:21–28
Śliwka-Kaszyńska M, Jaszczołt K, Hoczyk A, Rachoń J (2006) Anal Chem (Warsaw) 51:123–133
Śliwka-Kaszyńska M, Jaszczołt K, Kołodziejczyk A, Rachoń J (2006) Talanta 68:1560–1566
Gutsche CD, Iqbal M, Stewart D (1986) J Org Chem 51:742–745
Gutsche CD, Lin LG (1986) Tetrahedron 42:1633–1640
Feibush B, Cohen MJ, Karger BL (1983) J Chromatogr 282:3–26
Ihara T, Sugimoto Y, Asada M, Nakagama T, Hobo T (1995) J Chromatogr A 694:49–56
Sykora J, Budka J, Lhotak P, Stibor I, Cisarova I (2005) Org Biomol Chem 3:2572–2578
Karger BL, Gant JR, Hartkopt A, Weiner PH (1976) J Chromatogr 128:65–78
Vigh G, Varga-Puchony Z (1980) J Chromatogr 196:1–9
Gebauer S, Friebe S, Gübitz G, Krauss GJ (1998) J Chromatogr Sci 36:383–387
Menyes U, Haak A, Sokoliess T, Jira T, Roth U, Troeltzsch C (1999) GIT Spez Sep 19:17–19
Sokoliess T, Kessler D, Menyes U, Roth U, Jira T (2000) LaborPraxis 24(7/8):70–72
Li LS, Da SL, Feng YQ, Liu M (2004) J Liq Chrom Rel Technol 27:2167–2188
Xu W, Li JS, Feng YQ, Da SL, Chen YY, Xiao XZ (1998) Chromatographia 48:245–250
Li LS, Da SL, Feng YQ, Liu M (2004) Anal Sci 20:561–564
Li JS, Da SL, Feng YQ, Liu M (2004) J Chromatogr A 1040:53–61
Lee YK, Ryu YK, Ryu JW, Kim BE, Park JH (1997) Chromatographia 46:507–510
Schoenmakers PJ, Billiet HA, Tijssen R, De Galan L (1978) J Chromatogr 149:519–537
Sokoliess T, Menyes U, Roth U, Jira T (2000) J Chromatogr A 898:35–52
Li LS, Liu M, Da SL, Feng YQ (2004) Talanta 62:643–648
Li LS, Da SL, Feng YQ, Liu M (2004) Anal Lett 37:2805–2817
Xiao XZ, Feng YQ, Da SL, Zhang Y (1999) Chromatographia 49:643–648
Huai QY, Zhao B, Zuo YM (2004) Chromatographia 59:637–645
Xiao XZ, Feng YQ, Da SL, Zhang Y (2000) Anal Lett 33:3355–3372
Xiao YX, Xiao XZ, Feng YQ, Wang ZH, Da SL (2001) J Liq Chrom Rel Technol 24:2925–2942
Li LS, Liu M, Da SL, Feng YQ (2004) Talanta 63:433–441
Gezici O, Tabakci M, Kara H, Ylmaz M (2006) J Macro Sci Pure Appl Chem 43:221–231
Pietraszkiewicz O, Pietraszkiewicz M (1998) Polish J Chem 72:2418–2422
Pietraszkiewicz O, Pietraszkiewicz M (1999) J Incl Phenom Macro Chem 35:261–270
Acknowledgments
The authors gratefully acknowledge financial support for this work from the State Committee for Scientific Research (KBN 3 T09 A, Project No. 138 29).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jaszczołt, K., Śliwka-Kaszyńska, M. Preparation and Evaluation of 1,3-alternate 25,27-Bis-[p-nitrobenzyloxy]-26,28-bis-[3-propyloxy]-calix[4]arene-bonded Silica Gel Stationary Phase for LC. Chroma 66, 837–845 (2007). https://doi.org/10.1365/s10337-007-0419-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-007-0419-z