Abstract
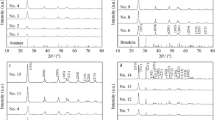
Highly photoactive bi-phase nanocrystalline TiO2 photocatalyst was prepared by a solvent evaporation-induced crystallization (SEIC) method, and calcined at different temperatures. The obtained TiO2 photocatalyst was characterized with X-ray diffraction (XRD), transmission electron microscopy (TEM) and BET surface areas. The photocatalytic activity was evaluated by the photocatalytic oxidation of acetone in air. The results show that solvent evaporation can promote the crystallization and phase transformation of TiO2 at 100°C. When calcination temperatures are below 600°C, the prepared TiO2 powders show bimodal pore size distributions in the mesoporous region. At 700°C, the pore size distributions exhibit monomodal distribution of the inter-aggregated pores due to the collapse of the intra-aggregated pores. At 100°C, the obtained TiO2 photocatalyst by this method shows good photocatalytic activity, and at 400°C, its photocatalytic activity exceeds that of Degussa P25. This may be attributed to the fact that the prepared TiO2 photocatalyst has higher specific surface areas, smaller crystallite size and bimodal pore size distribution.
Similar content being viewed by others
References
Hoffmann, M. R., Martin, S. T., Choi, W. et al., Environmental applications of semiconductor photocatalysis, Chem. Rev., 1995, 95: 69–96.
Fujishima, A., Rao, T. N., Tryk, D. A., Titanium dioxide photocatalysis, J. Photochem. Photobiol. C: Photochem. Rev., 2000, 1: 1–21.
Hagfeldt, A., Gratzel, M., Light-induced redox reactions in nanocrystalline systems, Chem. Rev., 1995, 95: 49–68.
Yu, J. C., Yu, J., Ho, W. et al., Preparation of highly photocatalytic active nano-sized TiO2 particles via ultrasonic irradiation, Chem. Commum., 2001, 14(19): 1942–1943.
Yu, J. C., Yu, J., Ho, W. et al., Effects of F--doping on the photocatalytic activity and microstructures of nanocrystalline TiO2 powders, Chem. Mater., 2002, 14(9): 3808–3816.
Yu, J., Yu, J. C., Ho, W. et al., Effects of calcination temperature on the photocatalytic activity and photo-induced super-hydrophilicity of mesoporous TiO2 thin films, New J. Chem., 2002, 26: 607–613.
Liu, H., Yang, W., Ma, Y. et al., Promoted phase transition of titania nanoparticles prepare by a photo-assisted sol-gel method, New J. Chem., 2002, 26: 975–977.
Zhang, H., Banfield, J. F., Understanding polymorphic phase transformation behavior during growth of nanocrystalline aggregates: insights from TiO2, J. Phys. Chem. B, 2000, 104: 3481–3487.
Sing, K. S. W., Everett, D. H., Haul, R. A. W. et al., Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity, Pure Appl. Chem., 1985, 57: 603–619.
Yu, J., Yu, J. C., Leung, M. K. P. et al., Effects of acidic and basic hydrolysis catalysts on the photocatalytic activity and microstructures of bimodal mesoporous titania, J. Catal., 2003, 217: 69–78.
Kumar, K. N. P., Kumar, J., Keizer, K., Effect of peptization on densification and phase-transformation behavior of sol-gel-derived nanostructured titania, J. Am. Ceram. Soc., 1994, 77: 1396–1400.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, J., Yu, J.C., Cheng, B. et al. Preparation and characterization of highly photoactive nanocrystalline TiO2 powders by solvent evaporationinduced crystallization method. Sc. China Ser. B-Chem. 46, 549–557 (2003). https://doi.org/10.1360/03yb0012
Received:
Issue Date:
DOI: https://doi.org/10.1360/03yb0012