Abstract
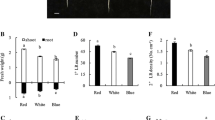
Some characteristics of the rice (Oryza sativa L.) root were found in the experiment of unilaterally irradiating the roots which were planted in water: (i) All the seminal roots, adventitious roots and their branched roots bent away from light, and their curvatures ranged from 25° to 60°. The curvature of adventitious root of the higher node was often larger than that of the lower node, and even larger than that of the seminal root, (ii) The negative phototropic bending of the rice root was mainly due to the larger growth increment of root-tip cells of the irradiated side compared with that of the shaded side, (iii) Root cap was the site of light perception. If root cap was shaded while the root was irradiated the root showed no negative phototropism, and the root lost the characteristic of negative phototropism when root cap was divested. Rice root could resume the characteristic of negative phototropism when the new root cap grew up, if the original cells of root cap were well protected while root cap was divested, (iv) The growth increment and curvature of rice root were both influenced by light intensity. Within the range of 0–100 μmol · m2 -s−1, the increasing of light intensity resulted in the decreasing of the growth increment and the increasing of the curvature of rice root, (v) The growth increment and the curvature reached the maximum at 30°C with the temperature treatment of 10–40°C. (vi) Blue-violet light could prominently induce the negative phototropism of rice root, while red light had no such effect. (vii) The auxin (IAA) in the solution, as a very prominent influencing factor, inhibited the growth, the negative phototropism and the gravitropism of rice root when the concentration of IAA increased. The response of negative phototropism of rice root disappeared when the concentration of IAA was above 10 mg · L−1
Similar content being viewed by others
References
Alan, E. P., Seong, K. M., Stephanie, M. H. et al., shl, a new set of Arabidopsis mutants with exaggerated developmental responses to available red, far-red, and blue light, Plant Physiol., 2001, 127: 295–304.
Winslow, R. B., Margaret, A. O., Phototropins in plant photomorphogenesis to date, five phytochromes, two cryptochromes, one phototropin, and one super-chrome, Plant Physiol., 2001, 125: 85–88.
Tan, Y., Ye, Q. S., A review of progress in study on the plant phototropism, Plant Science of Subtropics (in Chinese), 2001, 30(1): 64–68.
Darwin, F., Über das Wachstum negativ heliotropischer Wurzeln in Licht und im Finstern, Arb Bot Instit Würzburg, 1880, 2: 521–528.
Okada, K., Shimura, Y., Mutational analysis of root gravitropism and phototropism of Arabidopsis thaliana seedlings, Aust. J. Plant Physiol., 1992, 19: 439–448.
Stanislav, V., Liming, Z., Fred, D. S., Interaction of root gravitropism and phototropism in Arabidopsis wild-type and starchless mutants, Plant physiol., 2000, 122: 453–461.
John, Z. K., Kelly, M. M., Lisa, A. O. et al., Phototropism and gravitropism in lateral roots of Arabidopsis wild type and starchless mutants, Plant Cell Physiol., 2002, 43(1): 35–43.
Gu, Y. J., Wang, Z., Wang, W. X. et al., The negative phototropism of rice root, Plant Physiology Communications (in Chinese), 2001, 37(5): 396–398.
Halstead, T. M., Scott, T. K., Plant Gravitational and Space Research, Baltimore: The Waverly Press, 1984.
Takahashi, F., Hishinuma, T., Kataoka, H., Blue-light induced branching in Vaucheria—Requirement of nuclear accumulation in the irradiated region, Plant Physiol., 2001, 42: 274–285.
Mark, E., Transporters on the move, Nature, 2001, 416: 374–375.
Ahamd, M., Cashnore, A. R., HY4 gene of A. thaliana encodes a protein with characteristics of a blue light photoreceptor, Nature, 1993, 366: 162–166.
Lin, C., Ahmad, M., Chan, J. et al., CRY2: a second member of the Arabidopsis cryptochrome gene family, Plant Physiol., 1996, 110: 1047.
Men, F. J., Cell Biology of the Plant Flower Development (in Chinese), Beijing: China Agriculture Press, 2000, 68–70.
Kiss, J. Z., Ruppcl, N. J., Hangartcr, R. P., Phototropism in Arabidopsis roots is mediated by two sensory systems, Adv. Space Res., 2001, 27(5): 877–885.
Behriger, F. J., Davies, P. J., Indole-3-acid level after phytochrome-mediated changes in the stem elongation rate of dark and light-grown Pisum seedlings, Planta, 1992, 188: 85–89.
Ni, W. M., Chen, X. Y., Xu, Z. H. et al., Advance in study of polar auxin transport, Acta Botanica Sinica (in Chinese), 2000, 42(3): 221–222.
Niko, G., Jifi, F., York, D. S. et al., Auxin transport inhibitors block PIN1 cycling and vesicle trafficking, Nature, 2001, 413: 425–428.
Jeane, C. G., Nicole, V., Differential hypocotyls and root development of Arabidopsis auxin-insensitive mutants, J. Plant Res., 2001, 114: 115–123.
Lino, M., Mediation of tropisms by lateral translocation of endogenous indole-3-acetic acid in maize coleoptiles, Plant Cell Environ., 1991, 14: 278–286.
Kaufman, L. S., Transduction of blue-light signals, Plant Physiol., 1993, 102: 333–337.
Yu, R. C., Pan, R. Z., Effects of blue light on the growth and levels of endogenous phytohormones in rice (Oryza sativa L.) seedlings, Acta Phytophysiologica Sinica (in Chinese), 1997, 23(2): 175–180.
Young, L. M., Evans, M. L., Hertel, R., Correlations between gravitropic curvature and auxin movement across gravistimulated roots of Zea mays, Plant Physiol., 1990, 92: 792–796.
Rashotte, A. M., Brady, S. R., Reed, R. C. et al., Basipetal auxin transports is required for gravitropisms in roots of Arabidopsis, Plant Physiol., 2000, 122: 481–490.
Dolan, L., Pointing roots in the right direction: the role of auxin transport in response to gravity, Gene Dev., 1998, 12: 2091–2095.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Z., Mo, Y., Qian, S. et al. Negative phototropism of rice root and its influencing factors. Sci. China Ser. C.-Life Sci. 45, 485–496 (2002). https://doi.org/10.1360/02yc9053
Received:
Issue Date:
DOI: https://doi.org/10.1360/02yc9053