Abstract
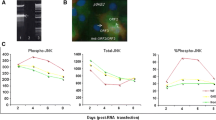
Since cell signal transduction plays an important role in disclosing the nature of human diseases, the pathogenesis of viruses may result from the disturbance of intracellular signal cascades caused by viral proteins. Hepatitis C virus (HCV) is a main causative agent of severe human liver disorders worldwide. So far, the mechanisms of HCV pathogenicity remain unclear. Envelope protein 2 (E2) of HCV is thought to be responsible for initiating virus attachment to host cells, which is a prerequisite of HCV infection. We assume that some early events of HCV pathogenic effects may result from the interaction of HCV E2 protein with its cellular receptor (human CD81), which could regulate cell proliferation and differentiation. To test this hypothesis, the effects of HCV E2 protein on MAPK/ERK pathway in Molt-4 and U937 cells with or without human CD81 expression were investigated. The results showed that HCV E2 protein could specifically activate the MAPK/ERK pathway, and such activation was inhibited by monoclonal antibodies against CD81 or HCV E2, serum antibodies from HCV infected patients, and upstream MEK1 inhibitor PD98059. Moreover, HCV E2-driven MAPK/ERK or downstream transcription factor Elk-1 activation was completely blocked in the presence of PD98059. These findings strongly suggest that the regulation of transmembrane signaling by HCV E2 protein via its receptor(s) on host cells might contribute to the development of HCV-related diseases.
Similar content being viewed by others
References
Favre, D., Berthillon, P., Trepo, C., Removal of cell-bound lipoproteins: A crucial step for the efficient infection of liver cells with hepatitis C virus in vitro, C R Acad. Sci. III, 2001, 324(12): 1141–1148.
Pilen, P., Uematsu, Y., Campagnoli, S. et al., Binding of hepatitis C virus to CD81, Science, 1998, 282(5390): 938–941.
Flint, M., Mckeating, J. A., The role of the hepatitis C virus glycoproteins in infection, Rev. Med. Virol., 2000, 10(2): 101–117.
Flint, M., Maidens, C., Loomis-price, L. D. et al., Characterization of hepatitis C virus E2 glycoprotein interaction with a putative cellular receptor, CD81. J. Virol., 1999, 73(8): 6235–6244.
Jacque, J. M., Mann, A., Enslen, H. et al., Modulation of HIV-1 infectivity by MAPK, a virion-associated kinase, EMBO J., 1998, 17(9): 2607–2618.
Flint, M., Dubuisson, J., Maidens, C. et al., Functional characterization of intracellular and secreted forms of a truncated hepatitis C virus E2 glycoprotein, J. Virol., 2000, 74(2): 702–709.
Huang, H., Fujii, H., Sankila, A. et al., Beta-catenin mutations are frequent in human hepatocellular carcinomas associated with hepatitis C virus infection, Am. J. Pathol., 1999, 155(6): 1795–1801.
Lai, M. M., Hepatitis viruses and signal transduction: True to the core? Hepatology, 2000, 32(2): 427–429.
Tsuchihara, K., Hijikata, M., Fukuda, K. et al., Hepatitis C virus core protein regulates cell growth and signal transduction pathway transmitting growth stimuli, Virology, 1999, 258(1): 100–107.
Takamatsu, M., Fujita, T., Hotta, H., Suppression of serum starvation-induced apoptosis by hepatitis C virus core protein, Kobe J. Med. Sci., 2001, 47(2): 97–112.
Fukuda, K., Tsuchihara, K., Hijikata, M. et al., Hepatitis C virus core protein enhances the activation of the transcription factor, Elkl, in response to mitogenic stimuli, Hepatology, 2001, 33(1): 159–165.
Polyak, S. J., Khabar, K. S., Paschal, D. M. et al., Hepatitis C virus nonstructural 5A protein induces interleukin-8, leading to partial inhibition of the interferon-induced antiviral response, J. Virol., 2001, 75(13): 6095–6106.
Tan, S. L., Katze, M. G., How hepatitis C virus counteracts the interferon response: The jury is still out on NS5A, Virology, 2001, 284(1): 1–12.
Borowski, P., Zurwiesch, J. S., Resch, K. et al., Protein kinase C recognizes the protein kinase A-binding motif of nonstructural protein 3 of hepatitis C virus, J. Biol. Chem., 1999, 274(43): 30722–30728.
Yagnik, A. T., Lahm, A., Meola, A. et al., A model for the hepatitis C virus envelope glycoprotein E2, Protein, 2000, 40(3): 355–366.
Zhao, L. J., Liu, H. Q., Cao, J. et al., Activation of intracellular MAPK/ERK initiated by hepatitis C virus envelope protein E2 in HepG2 cells, Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao, 2001, 33: 691–695.
Petracca, R., Falugi, F., Galli, G. et al., Structure-function analysis of hepatitis C virus envelope-CD81 binding, J. Virol., 2000, 74(10): 4824–4830.
Wellnitz, S., Klumpp, B., Barth, H. et al., Binding of hepatitis C virus-like particles derived from infectious clone H77C to defined human cell lines, J. Virol., 2002, 76(3): 1181–1193.
Allander, T., Drakenberg, K., Beyene, A. et al., Recombinant human monoclonal antibodies against different conformational epitopes of the E2 envelope glycoprotein of hepatitis C virus that inhibit its interaction with CD81, J. Gen. Virol., 2000, 81(pt10): 2451–2459.
Hadlock, K. G., Lanford, R. E., Perkin, S. et al., Human monoclonal antibodies that inhibit binding of hepatitis C virus E2 protein to CD81 and recognize conserved conformational epitopes, J. Virol., 2000, 74(22): 10407–10416.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, L., Liu, H., Zhu, S. et al. A new clue for the pathogenesis of hepatitis C virus infection: Activation of the MAPK/ERK signaling initiated by envelope protein 2. Sci. China Ser. C.-Life Sci. 46, 538–545 (2003). https://doi.org/10.1360/02yc0044
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1360/02yc0044