Abstract
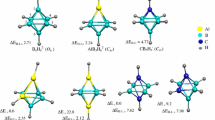
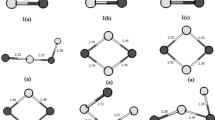
Using density functional theory (DFT) method with 6-31G* basis set, we have carried out the optimizing calculation of geometry, vibrational frequency and thermodynamical stability for (AlN) n + and (AlN) n + (n=1–15) clusters. Moreover, their ionic potential (IP) and electron affinity (EA) were discussed. The results show that the electrical charge condition of the cluster has a relatively great impact on the structure of the cluster and with the increase of n, this kind of impact is reduced gradually. There are no Al-Al and N-N bonds in the stable structure of (AlN) n + or (AlN) n -, and the Al-N bond is the sole bond type. The magic number regularity of (AlN) n + and (AlN) n - is consistent with that for (AlN) n , indicating that the structure with even n such as 2, 4, 6, ... is more stable. In addition, (AlN10 has the maximal ionization power (9.14 eV) and the minimal electron affinity energy (0.19 eV), which manifests that (AlN)10 is more stable than other clusters.
Similar content being viewed by others
References
Wu, H. S., Zhang, C. J., Xu, X. H. et al., The structure and stability for (AlN) n , Science in China, Series B, 2000, 43(6): 634–642.
Lee, Z. Y., Miao, X. S., Liu, X. J. et al., Kerr effect enhancement and corrosion resistance improvement by AlN and AlSiN films, J. Appl. Phys., 1990, 67(9): 5340–5345.
Rille, E., Zarwaseh, R., Pulker, H. K., Properties of reactively d.c. magnetron-sputtered AlN thin films, Thin Solid Films, 1993, 229: 215–217.
Kim, H. J., Egashira, Y., Komiyama, H., Temperature dependence of the sticking probability and molecular size of the film growth species in an atmospheric chemical vapor deposition process to form AlN AlCl3 and NH3, Appl. Phys. Lett., 1991, 59(20): 2521–2523.
Egashira, Y., Kim, H. J., Komiyama, H., Cluster size determination in the chemical vapor deposition of aluminum nitride, J. Am. Ceram. Soc., 1994, 77(8): 2009–2016.
Zheng, L. S., Huang, R. B., The progress of cluster science, National Natural Science Foundation (in Chinese), 1998, 3: 187–189.
Chu, C., Ong, P. P., Chen, H. F. et al., TOF study of pulsed-laser ablation of aluminum nitride for thin film growth, Appl. Surf. Sci., 1999, 137: 9197–9204.
Lynam, M. M., Interrante, L. V., Patterson, C. H. et al., Comparison of isoelectronic aluminum nitrogen and silicon carbon double bonds using valence bond methods, Inorg. Chem., 1991, 30(8): 1918–1922.
Davy, R. D., Jaffrey, K. J., Aluminum-nitrogen multiple bonds in small AlNH molecules: structures and vibrational frequencies of AlNH2, AlNH3 andAlNH4, J. Phys. Chem., 1994, 98(36): 8930–8936.
Muller, J., Aminodimethylalane (Me2AlNH2): matrix isolation and ab initio calculations, J. Am. Chem. Soc., 1996, 118: 6370–6376.
Matsunaga, N., Gordon, M. S., Stabilities and energetics of inorganic benzene isomers: prismanes, J. Am. Chem. Soc., 1994, 116: 11407–11419.
Alexey, Y., Timoshkin, H. F. B., Henry, F. S., The chemical vapor deposition of aluminum nitride: unusual cluster, J. Am. Chem. Soc., 1997, 119: 5668–5679.
Nayak, S. K., Khannas, N., Jena, P., Evolution of bonding in AlnN clusters: A transition from nonmetallic to metallic character, Phys. Rev., 1999, B57(7): 3787–3796.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, H., Xu, X., Zhang, C. et al. Structure and stability for (AlN) n + and (AlN) n + (n=1–15) clusters. Sc. China Ser. B-Chem. 45, 328–336 (2002). https://doi.org/10.1360/02yb9042
Received:
Issue Date:
DOI: https://doi.org/10.1360/02yb9042