Abstract
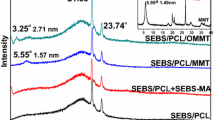
Supercritical carbon dioxide (scCO2) processing has been proven as a method for preparing polymer/montmorillonite (MMT) nanocomposites with improved platelet dispersion. The influence of scCO2 processing on the shape and size of the MMT tactoid/platelet, which is of great importance to the final platelet dispersion in the polymer matrix, is scarcely reported in the literature. In the present study, the pristine MMT was first surface modified with 3-glycidoxypropyltrimethoxysilane (the grafted MMT is labeled as GMMT), and then intercalated using three kinds of intercalating agents, myristyltrimethyl-ammonium bromide (MTAB), tetradecyltrihexylphosphonium chloride (TDTHP), and ethoxyltriphenyl-phosphonium chloride (ETPC), in water or scCO2, to study the effect of intercalating agent type and intercalation method on the morphology and thermal properties of GMMT, as a part of a program devoted to the synthesis of polymer/MMT nanocomposites. The structure of intercalated GMMT was characterized by thermogravimetric analysis, X-ray powder diffraction, and scanning electron microscopy (SEM). The optimum intercalation conditions in scCO2 were established by trying a range of reaction times and pressures. The structures of intercalated GMMT obtained under optimum scCO2 conditions and water were compared. The basal spacing of GMMT intercalated in scCO2 was almost the same as that in water, and both were obviously larger than that of GMMT. The GMMT exhibited a compact spherical morphology (examined using SEM), and the surface structures (including surface morphology, surface roughness, and surface compactness) of samples intercalated in water became ‘less compact’ and the degree of the ‘compactness’ of samples intercalated in scCO2 decreased further. Whether in water or scCO2, samples intercalated with TDTHP exhibited a larger basal spacing and the extent of disorder increased compared to that for samples intercalated with MTAB. The pristine MMT was also intercalated for comparison and silane grafting was proven to contribute to the increased basal spacing and ‘less compact’ surface structure.
Similar content being viewed by others
References
Al-Qadhi, M., Merah, N., and Gasem, Z.M. (2013) Mechanical properties and water uptake of epoxy-clay nanocomposites containing different clay loadings. Journal of Materials Science, 48, 3798-3704.
Bellair, R.J., Manitiu, M., Gulari, E., and Kannan, R.M. (2010) Investigation of clay modifier effects on the structure and rheology of supercritical carbon dioxide-processed polymer nanocomposites. Journal of Polymer Science Part A: Polymer Physics, 48, 823–831.
Bonczek, J.L., Harris, W.G., and Nkedi-Kizza, P. (2002) Monolayer to bilayer transitional arrangements of hexade-cyltrimethylammonium cations on Na-montmorillonite. Clays and Clay Minerals, 5, 11–17.
Chang, J.H., Kim, S.J., Joo, Y.L., and Im, S. (2004) Poly(ethylene terephthalate) nanocomposites by in situ interlayer polymerization: the thermo-mechanical properties and morphology of the hybrid fibers. Polymer, 45, 919–926.
Chou, C.C., Chang, Y.C., Chiang, M.L., and Lin, J.J. (2004) Conformational change of trifunctional poly(oxypropyle-ne)amines intercalated within a layered silicate confinement. Macromolecules, 37, 473–477.
He, W.T., Yao, Y., He, M., Zhang, K., Long, L.J., Zhang, M.M., Qin, S.H., and Yu, J. (2013) Influence of reaction conditions on the grafting pattern of 3-glycidoxypropyltri-methoxysilane on montmorillonite. Bulletin of the Korean Chemical Society, 34, 112–116.
Herrera, N.N., Letoffe, J.M., Reymond, J.P., and Bourgeat-Lami, E. (2005) Silylation of laponite clay particles with monofunctional and trifunctional vinyl alkoxysilanes. Journal of Materials Chemistry, 15, 863–871.
Huang, G.B., Ge, C.H., and He, B.J. (2011) Preparation, characterization and properties of amino-functionalized montmorillonite and composite layer-by-layer assembly with inorganic nanosheets. Applied Surface Science, 257, 7123–7128.
Huang, G.B., Gao, J.R., and Wang, X. (2012) Preparation and characterization of montmorillonite modified by phosphorus-nitrogen containing quaternary ammonium salts. Applied Surface Science, 258, 4054–4062.
Kojima, Y., Usuki, A., Kawasumi, M., Okada, A., Kurauchi, T., and Kamigaito, O. (1993) Synthesis of nylon 6-clay hybrid by montmorillonite intercalated with s-caprolactam. Journal of Polymer Science Part A: Polymer Chemistry, 31, 983–986.
Kong, Q.H., Hu, Y., and Lu, H.D. (2005) Synthesis and properties of polystyrene/Fe-montmorillonite nanocomposites using synthetic Fe-montmorillonite by bulk polymerization. Journal of Materials Science, 40, 4505–4509.
Leu, C.M., Wu, Z.W., and Wei, K.H.(2002) Synthesis and properties of covalently bonded layered silicates/polyimide (BTDA-ODA) nanocomposites. Chemistry of Materials, 14, 3016–3021.
Loring, J.S., Schaef, H.T., Turcu, R.V.F., Thompson, C.J., Miller, Q.R.S., Martin, P.F., Hu, J.Z., Hoyt, D.W., Qafoku, O., Ilton, E.S., Felmy, A.R., and Rosso, K.M. (2012) In situ molecular spectroscopic evidence for CO2 intercalation into montmorillonite in supercritical carbon dioxide. Langmuir, 28, 7125–7128.
Luo, J.J., and Daniel, I.M. (2003) Characterization and modeling of mechanical behavior of polymer/clay nanocomposites. Composites Science and Technology, 63, 1607–1616.
Manitiu, M., Bellair, R.J., Horsch, S., Gulari, E., and Kannan, R.M. (2008) Supercritical carbon dioxide-processed dispersed polystyrene-clay nanocomposites. Macromolecules, 41, 8038–8046.
Pack, S., Kashiwagi, T., Cao, C., Korach, C. S., Lewin, M., and Rafailovich, M.H. (2010) Role of surface interactions in the synergizing polymer/clay flame retardant properties. Macromolecules, 43, 5338–5351.
Park, M., Shim, I.K., Jung, E.Y., and Choy J.H. (2004) Modification of external surface of Laponite by silane grafting. Journal of Physics and Chemistry of Solids, 65, 499–501.
Paul, M.A., Alexandre, M., Degee, P., Henrist, C., Rulmont A., and Dubois, P. (2003) New nanocomposite materials based on plasticized poly(1-lactide) and organo-modified mont-morillonites: thermal and morphological study. Polymer, 44, 443–450.
Schaef, H.T., Ilton, E.S., Qafoku, O., Martin, P.F., Felmy, A.R., and Rosso K.M. (2012) In situ XRD study of Ca2+ saturated montmorillonite (STX-1) exposed to anhydrous and wet supercritical carbon dioxide. International Journal of Greenhouse Gas Control, 6, 220–229.
Semenzato, S., Lorenzetti, A., Modesti, M., Ugel, E., Hrelja, D., Besco, S., Michelin, R.A., Sassi, A., Facchin, G., Zorzi, F., and Bertani, R. (2009) A novel phosphorus polyurethane FOAM/montmorillonite nanocomposite: Preparation, characterization and thermal behaviour. Applied Clay Science, 44, 35–42.
Serhatkulu, G.K., Horsch, S.E., Kannan, R., and Gulari, E. (2005) Structure and rheology of supercritical carbon dioxide exfoliated polymer nanocomposites. Nanotechnology, 2, 123–126.
Song, K. and Sandi, G. (2001) Characterization of montmor-illonite surfaces after modification by organosilane. Clays and Clay Minerals, 49, 119–125.
Vaia, R.A., Ishii, H., and Giannelis, E.P. (1993) Synthesis and properties of two-dimensional nanostructures by direct intercalation of polymer melts in layered silicates. Chemistry of Materials, 51, 694–1696.
Xie, W., Xie, R.C., Pan, W.P., Hunter, D., Tan, L.S., and Vaia, R. (2002) Thermal stability of quaternary phosphonium modified montmorillonites. Chemistry of Materials, 14, 4837–4845.
Zhao, Q. and Samulsk, E.T. (2003) Supercritical CO2-mediated intercalation of PEO in clay. Macromolecules, 36, 6967–6969.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, S., Yao, Y., He, W. et al. Effect of Intercalation Method and Intercalating Agent Type on the Structure of Silane-Grafted Montmorillonite. Clays Clay Miner. 61, 580–589 (2013). https://doi.org/10.1346/CCMN.2013.0610610
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.2013.0610610