Abstract
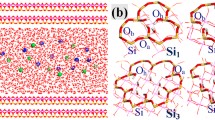
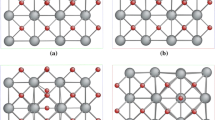
Kaolinite is often a cause of deformation in soft-rock tunnel engineering, leading to safety problems. In order to gain a better predictive understanding of the governing principles associated with this phenomenon, the physical and chemical properties of kaolinite were investigated using an efficient, firstprinciples scheme for studying isomorphic substitution of Al ions in kaolinite by two kinds of other elements (namely, the dual defect). Elements that are relatively common in natural kaolinite were chosen from groups II (Be, Mg, Ca, and Sr) and III (Fe and Sc) of the Periodic Table as dual-defect ions to substitute for Al ions in kaolinite. By systematically calculating the impurity-formation energies (which characterize the difference in the total crystal energy before and after the defect arises) and transitionenergy levels, which characterize the energy cost for the transformation between two different charge states, the (Be + Sc)Al (i.e. the replacement of two specific Al ions in kaolinite by external Be and Sc ions), (Ca + Sc)Al, (Mg + Sc)Al, and (Sr + Sc)Al ion pairs were determined to have low formation energies, suggesting that these combinations of ions can easily substitute for Al ions in kaolinite. The (Be + Fe)Al, (Ca + Fe)Al, (Mg + Fe)Al, and (Sr + Fe)Al ion pairs have relatively high formation energies which make isomorphic substitution (or doping) in kaolinite difficult. Moreover, these combinations of elements from groups II and III were found to have relatively low transition-energy levels compared with other element pairs. Among them, (Sr + Sc)Al have the lowest transition-energy level at 0.06 eV above the valence band maximum. When compared with single external substitutional defects in kaolinite, remarkably, the dual defects have relatively low formation energies and transition-energy levels. The results are helpful in understanding the chemical and physical properties of natural kaolinite.
Similar content being viewed by others
References
Adams, J.M. (1983) Hydrogen atom position in kaolinite by neutron profile refinement. Clays and Clay Minerals, 31, 352–358.
Angel, B.R., Jones, J.P.E., and Hall, P.L. (1974) Electron spin resonance studies of doped synthetic kaolinites. Clays and Clay Minerals, 10, 247–255.
Benco, L., Tunega, D., Hafner, J., and Lischka, H. (2001) Orientation of OH groups in kaolinite and dickite: ab initio molecular dynamics study. American Mineralogist, 86, 1057–1065.
Bish, D.L. (1993) Rietveld refinement of the kaolinite structure at1.5 K. Clays and Clay Minerals, 41, 738–744.
Blöchl, P.E. (1994) Projector augmented-wave method. Physical Review B, 50, 17953–17979.
Chen, J. and Wang, H.N. (2004) Pp. 35–60 in: Geochemistry. (H.Y. Xie and P. Liu, editors). Science Press, Beijing.
Giese, R.F., Jr. (1973) Interlayer bonding in kaolinite dickite and nacrite. Clays and Clay Minerals, 21, 145–149.
Hayashi, S. (1997) NMR study of dynamics and evolution of guest molecules in kaolinite/dimethyl sulfoxide intercalation compound. Clays and Clay Minerals, 45, 724–732.
He, M.C., Jing, H.H., and Yao, A.J. (2000) Research progress of softrock engineering geomechanics in China coal mine. Journal of Engineering Geology, 1, 46–62.
He, M.C., Xie, H.P., and Peng, S.P. (2005) Study on rock mechanics in deep mining engineering. Chinese Journal of Rock Mechanics and Engineering, 24, 2803–2813.
He, M.C., Fang, Z.J., and Zhang, P. (2009) Theoretical studies on the defects of kaolinite in clays. Chinese Physics Letter, 26, 059101–059104.
Hess, A.C. and Saunders, V.R. (1992) Periodic ab initio Hartree-Fock calculation of the low-symmetry mineral kaolinite. The Journal of Physical Chemistry, 11, 4367–4374.
Hobbs, J.D., Cygan, R.T., Nagy, K.L., Schultz, P.A., and Sears, M.P. (1997) All-atom ab initio energy minimization of the kaolinite crystal structure. American Mineralogist, 82, 657–662.
Hu, X.L. and Angelos, M. (2008) Water on the hydroxylated (001) surface of kaolinite: From monomer adsorption to a flat2D wetting layer. Surface Science, 602, 960–974.
Kresse, G. and Furthmüller, J. (1996) Efficientit erative schemes for ab initio total-energy calculations using a plane-wave basis set. Physical Review B, 54, 11169–11186.
Kresse, G. and Joubert, J. (1999) From ultrasoft pseudopotentials to the projector augmented-wave method. Physical Review B, 59, 1758–1762.
Monkhorst, H.J. and Pack, J.D. (1976) Special points for Brillouin-zone integrations. Physical Review B, 13, 5188–5192.
Plançon, A., Giese, R.F. Jr., Snyder, R., Drits, V.A., and Bookin, A.S. (1997) Stacking faults in the kaolinite-group minerals: defect structures of kaolinite. Clays and Clay Minerals, 37, 195–198.
Poole, C.P. Jr. and Farach, H.A. (1986) Electron spin resonance. ASM Handbook, 10, 253–266.
Teppen, B.J., Rasmussen, K., Bertsch, P.M., Miller, D.M., and Schäferll, L. (1997) Molecular dynamic modeling of clay minerals. 1. Gibbsite, kaolinite, pyrophyllite, and beidellite. The Journal of Physical Chemistry B, 101, 1579–1587.
Wei, S.H. and Zhang, S.B. (2002) Chemical trends of defect formation and doping limit in II-VI semiconductors: The case of CdTe. Physical Review B, 66, 155211–155221.
Zhang, S.B., Wei, S.H., and Zunger, A. (1997) Stabilization of ternary compounds via ordered arrays of defect pairs. Physical ReviewL etter, 78, 4059–4062.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, MC., Zhao, J., Fang, ZJ. et al. First-Principles Study of Isomorphic (‘Dual-Defect’) Substitution in Kaolinite. Clays Clay Miner. 59, 501–506 (2011). https://doi.org/10.1346/CCMN.2011.0590507
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.2011.0590507