Abstract
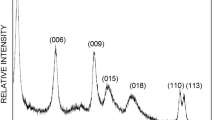
Among the many techniques used to remove toxic dyes from the environment, layered double hydroxides (LDH) are considered to be especially environmentally friendly, but, this quality may be altered by variations in the octahedral Mg/Al molar ratios in the LDH structure. The aim of the present study was to synthesize environmentally sound LDH for use as an economically viable sorbent for the adsorption of reactive brilliant orange X-GN. Layered double hydroxides with Mg/Al molar ratios of 2:1 and 4:1 were prepared by co-precipitation. The materials obtained were characterized by powder X-ray diffraction (XRD), Fourier-transform infrared (FTIR) spectroscopy, X-ray fluorescence spectroscopy (XRF), and surface-area analysis. Batch experiments were carried out to investigate the effects of contact time, pH, adsorbent dosage, and initial dye concentration on the adsorption behavior of the reactive brilliant orange X-GN by Mg-Al LDH. The results showed that the optimum pH value for dye adsorption was 3.0, at which the adsorption capacities of the reactive brilliant orange X-GN by the 2:1 LDH and the 4:1 LDH at 298 K were 79.370 mg/g and 83.343 mg/g, respectively. Further analysis of the dye-adsorption kinetics show that they fit the pseudo second-order model well. The adsorption equilibrium data showed that the Langmuir model provided better correlation of the equilibrium data than the Freundlich model. This result indicates that LDH provide specific homogeneous sites where monolayer dye adsorption occurs. The results of XRD and FTIR analyses of LDH before and after the dye adsorption demonstrated that the adsorption mechanisms were ion exchange and coulombic attraction.
Similar content being viewed by others
References
Aksu, Z. and Dönmez, G. (2003) A comparative study on the biosorption characteristics of some yeasts for Remazol Blue reactive dye. Chemosphere, 50, 1075–1083.
Al-Degs, Y.S., El-Barghouthi, M.I., El-Sheikh, A.H., and Walker, G.M. (2008) Effect of solution pH, ionic strength, and temperature on adsorption behavior of reactive dyes on activated carbon. Dyes and Pigments, 77, 16–23.
Anirudhan, T.S. and Radhakrishnan, P.G. (2009) Kinetics, thermodynamics and surface heterogeneity assessment of uranium (VI) adsorption onto cation exchange resin derived from a lignocellulosic residue. Applied Surface Science, 255, 4983–4991.
Beaudot, P., De Roy, M.E., and Besse, J.P. (2004) Preparation and characterization of intercalation compounds of layered double hydroxides with Metallic Oxalato complexes. Chemistry of Materials, 16, 935–945.
Chen, S.L., Xu, Z.P., Zhang, Q., Max Lu, G.Q., Hao, Z.P., and Liu, S.M. (2009) Studies on adsorption of phenol and 4-nitrophenol on MgAl-mixed oxide derived from MgAl-layered double hydroxide. Separation and Purification Technology, {b67}, 194–200.
Cheng, X., Huang, X.R., Wang, X.Z., Zhao, B.Q., Chen, A.Y., and Sun, D.Z. (2009) Phosphate adsorption from sewage sludge filtrate using zinc-aluminum layered double hydroxides. Journal of Hazardous Materials, 169, 958–964.
Chuang, Y.H., Tzou, Y.M., Wang, M.K., Liu, C.H., and Chiang, P.N. (2008) Removal of 2-Chlorophenol from aqueous solution by Mg/Al layered double hydroxide (LDH) and modified LDH. Industrial & Engineering Chemistry Research, 47, 3813–3819.
Dadwhal, M., Kim, T.W., Sahimi, M., and Tsotsis, T.T. (2008) Study of CO2 diffusion and adsorption on calcined layered double hydroxides: The effect of particle size. Industrial & Engineering Chemistry Research, 47, 6150–6157.
Forgacs, E., Cserháti, T., and Oros, G. (2004) Removal of synthetic dyes from wastewaters: a Review. Environment International, 30, 953–971.
Goh, K.-H., Lim, T.-T., and Dong, Z. (2008) Application of layered double hydroxides for removal of oxyanions: A review. Water Research, 42, 1343–1368.
Goh, K.-H., Lim, T.-T., and Dong, Z. (2009) Enhanced arsenic removal by hydrothermally treated nanocrystalline Mg/Al layered double hydroxide with nitrate intercalation. Environmental Science & Technology, 43, 2537–2543.
Justi, K.C., Fávere, V.T., Laranjeira, M.C.M., Neves, A., and Peralta, R.A. (2005) Kinetics and equilibrium adsorption of Cu(II), Cd(II), and Ni(II) ions by chitosan functionalized with 2[-bis-(pyridylmethyl)aminomethyl]-4-methyl-6-formylphenol. Journal of Colloid and Interface Science, 291, 369–374.
Nakayama, H., Hirami, S., and Tsuhako, M. (2007) Selective adsorption of mercury ions by mercaptocarboxylic acid intercalated Mg-Al layered double hydroxide. Journal of Colloid and Interface Science, 315, 177–183.
Netpradit, S., Thiravetyan, P., and Towprayoon, S. (2004) Adsorption of three azo reactive dyes by metal hydroxide sludge: effect of temperature, pH, and electrolytes. Journal of Colloid and Interface Science, 270, 255–261.
Noeline, B.F., Manohar, D.M., and Anirudhan, T.S. (2005) Kinetic and equilibrium modeling of lead (II) sorption from water and wastewater by polymerized banana stem in a batch reactor. Separation and Purification Technology, 45, 131–140.
Özacar, M. and Sengil, I.A. (2005) A kinetic study of metal complex dye sorption onto pine sawdust. Process Biochemistry, 40, 565–572.
Pearce, C.I., Lloyd, J.R., and Guthrie, J.T. (2003) The removal of colour from textile wastewater using whole bacterial cells: a review. Dyes and Pigments, 58, 179–196.
Reinholdt, M.X., Babu, P.K., and Kirkpatrick, R.J. (2009) Preferential adsorption of lower-charge glutamate ions on layered double hydroxides: an NMR investigation. The Journal of Physical Chemistry C, 113, 3378–3381.
Robinson, T., McMullan, G., Marchant, R., and Nigam, P. (2001) Remediation of dyes in textile effluent: a critical review on current treatment technologies with a proposed alternative. Bioresource Technology, 77, 247–255.
Wang, S.-L., Liu, C.H., Wang, M.K., Chuang, Y.H., and Chiang, P.N. (2009) Applied Clay Science, 43, 79–85.
Weisburger, J.H. (2002) Comments on the history and importance of aromatic and heterocyclic amines in public health. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis, 506–507, 9–20.
Yang, L., Shahrivari, Z., Liu, P.K.T., Sahimi, M., and Tsotsis, T.T. (2005) Removal of trace levels of arsenic and selenium from aqueous solutions by calcined and uncalcined layered double hydroxides (LDH). Industrial & Engineering Chemistry Research, 44, 6804–6815.
Yong, Z., Mata, V., and Rodrigues, A.E. (2000) Adsorption of carbon dioxide onto hydrotalcite-like compounds (HTlcs) at high temperatures. Industrial & Engineering Chemistry Research, 40, 204–209.
You, Y., Vance, G.F., and Zhao, H. (2001) Selenium adsorption on Mg-Al and Zn-Al layered double hydroxides. Applied Clay Science, 20, 13–25.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, P., Zhang, Q., Dai, Y. et al. Removal of Reactive Brilliant Orange X-GN from Aqueous Solutions By Mg-Al Layered Double Hydroxides. Clays Clay Miner. 59, 438–445 (2011). https://doi.org/10.1346/CCMN.2011.0590501
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.2011.0590501