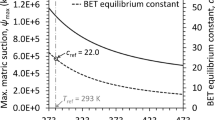
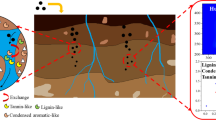
Abstract
Organoclays were prepared by exchanging Ca2+ in a Ca2+-saturated smectite partially or fully with trimethylphenylammonium (TMPA) cations. The mechanistic function of these organoclays as adsorbents for neutral organic compounds in aqueous solution was examined. TMPA cations were found to take a random distribution on the surfaces of mixed Ca/TMPA-smectites. The presence of TMPA, and its random distribution, resulted in water associated with the clay surfaces being held more weakly. Apparently, the interspersing of TMPA and Ca2+ ions prohibits the formation of a stable network of water molecules around Ca2+. Water molecules associated with the siloxane surface in mixed Ca/TMPA-clays are removed during the adsorption of neutral organic compounds from bulk water, leaving only ∼11 strongly held water molecules around each Ca2+, as opposed to ∼58 water molecules in homoionic Ca2+-smectite. These results demonstrate that the amount of water associated with the clay surfaces and interlayers depends on the nature of the exchange cation(s), and not on the amount of available siloxane surface area by itself. We conclude that in TMPA-smectites the TMPA cations function as nonhydrated pillars, and sorption of organic solutes occurs predominantly on the adjacent siloxane surfaces, which are hydrophobic in nature. The water molecules around Ca2+ in mixed Ca/TMPA-smectites obscures some of the siloxane surfaces. This diminishes sorption capacity, in an amount roughly equivalent to the fraction of the CEC occupied by Ca2+, because organic solutes cannot displace the waters of hydration of Ca2+.
Similar content being viewed by others
References
Boyd SA, Jaynes WF, Ross BS. 1991. Immobilization of organic contaminants by organo-clays: Application to soil restoration and hazardous waste containment. In: Baker RS, editor. Organic substances and sediments in waters, vol 1. Boca Raton, FL: CRC Pr. p 181–200.
Boyd SA, Lee J-F, Mortland MM. 1988. Attenuating organic contaminant mobility by soil modification. Nature 333: 345–347.
Boyd SA, Mortland MM, Chiou CT. 1988. Sorption characteristics of organic compounds on hexadecyltrimethylam-monium-smectite. Soil Sci Soc Am J 52:652–657.
Boyd SA, Sun S, Lee J-F, Mortland MM. 1988. Pentachlo-rophenol sorption by organo-clays. Clays Clay Miner 36: 125–130.
Burris DR, Antworth CP. 1992. In-situ modification of aquifer material by a cationic surfactant to enhance retention of organic contaminants. J Contam Hydrol 10:325–327.
Call F. 1957. The mechanism of sorption of ethylene dibro-mide on moist soils. J Sci Food Agric 8:630–639.
Chiou CT, Peters LJ, Freed VH. 1979. A physical concept of soil-water equilibria for nonionic organic compounds. Science 206:831–832.
Chiou CT, Porter PE, Schmedding DW. 1983. Partition equilibria of nonionic organic compounds between soil organic matter and water. Environ Sci Technol 17:227–231.
Gregg SJ, Sing KSW. 1982. Adsorption, surface area, and porosity. New York: Academic Pr 303 p.
Hunt JP. 1965. Metal ions in aqueous solution. New York: W. A. Benjamin, p 16.
Hanson WJ, Nex RW. 1953. Diffusion of ethylene dibromide in soil. Soil Sci 76:209–214.
Jaynes WF, Boyd SA. 1990. Trimethylphenylammonium-smectite as an effective adsorbent of water soluble aromatic hydrocarbons. J Air Waste Manage Assoc 40:1649–1653.
Jaynes WF, Boyd SA. 1991. Hydrophobicity of siloxane surfaces in smectite as revealed by aromatic hydrocarbon adsorption from water. Clays Clay Miner 39:428–436.
Kukkadapu RK, Boyd SA. 1995. Tetramethylphosphonium-and tetramethylammonium-smectites as adsorbents of aromatic and chlorinated hydrocarbons: Effect of water on adsorption efficiency. Clays Clay Miner 43:318–323.
Lee J-F, Mortland MM, Boyd SA, Chiou CT. 1989. Shape-selective adsorption of aromatic molecules from water by tetramethylammonium-smectite. J Chem Soc, Faraday Trans 1 85:2953–2962.
Lee J-F, Mortland MM, Chiou CT, Kile DE, Boyd SA. 1990. Adsorption of benzene, toluene, and xylene by two tetramethylammonium-smectites having different charge densities. Clays Clay Miner 38:113–120.
McBride MB. 1994. Environmental chemistry of soils. New York: Oxford Univ Pr. p 344.
McBride MB, Mortland MM. 1973. Segregation and exchange properties of alkylammonium ions in a smectite and vermiculite. Clays Clay Miner 21:323–329.
Mortland MM, Sun S, Boyd SA. 1986. Clay-organic complexes as adsorbents for phenol and chlorophenols. Clays Clay Miner 34:581–585.
Nye JV, Guerin WF, Boyd SA. 1994. Heterotrophic activity of microorganisms in soils treated with quaternary ammonium compounds. Environ Sci Technol 28:944–951.
Sheng G, Xu S, Boyd SA. 1996a. Mechanisms controlling sorption of neutral organic contaminants by surfactant derived and natural organic matter. Environ Sci Technol 30: 1553–1557.
Sheng G, Xu S, Boyd SA. 1996b. Surface heterogeneity of trimethylphenylammonium-smectite as revealed by adsorption of aromatic hydrocarbons from water. Clays Clay Miner 45:659–669.
Spencer WF, Cliath MM. 1970. Desorption of lindane from soil as related to vapor density. Soil Sci Soc Am Proc 34: 574–578.
Spencer WF, Cliath MM, Farmer WJ. 1969. Vapor density of soil-applied dieldrin as related to soil-water content, temperature, and dieldrin concentration. Soil Sci Soc Am Proc 33:509–511.
Stark FL Jr. 1948. Investigations of chloropicrin as a soil fumigant. New York (Cornell) Agr Exp Sta Mem 178:1–61.
Xu S, Boyd SA. 1994. Cation exchange chemistry of hexade-cyltrimethylammonium in a subsoil containing vermiculite. Soil Sci Soc Am J 58:1382–1391.
Xu S, Boyd SA. 1995. Cationic surfactant sorption to a ver-miculitic subsoil via hydrophobic bonding. Environ Sci Technol 29:312–320.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sheng, G., Boyd, S.A. Relation of Water and Neutral Organic Compounds in the Interlayers of Mixed Ca/Trimethylphenylammonium-Smectites. Clays Clay Miner. 46, 10–17 (1998). https://doi.org/10.1346/CCMN.1998.0460102
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1998.0460102