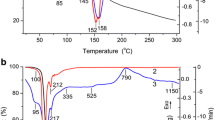
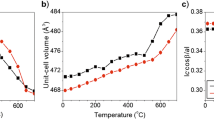
Abstract
Weathering products formed on the surface of K-feldspar in Yakushima Island, Japan were investigated by X-ray powder diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy dispersive X-ray analysis (EDX) and X-ray photoelectron spectroscopy (XPS). XRD confirmed that the weathering products were composed mainly of gibbsite and halloysite. SEM, TEM and EDX clearly showed formation of amorphous aluminum hydroxide exhibiting 2 distinct habits: 1) curled fibrous or circular forms less than 0.02 μm in diameter; and 2) a spherical habit less than 1.0 μm in diameter. The fibrous aluminum hydroxide exhibited curled fibrous forms or circular forms less than 0.02 μm in diameter and gave a diffuse electron diffraction halo. EDX indicated that the material consisted mainly of Al and very small amounts of Si and Fe. The spherical aluminum hydroxide also gave similar EDX and electron diffraction characteristics to the fibrous material. These fibrous and spherical aluminum hydroxides must be formed as a metastable phase in the earliest weathering stages, and transformed into a stable phase of gibbsite and halloysite as the reaction proceeded.
Similar content being viewed by others
References
Allen BL, Hajek BF. 1989. Mineral occurrence in soil environments. In: Dixon JB, Weed SB, editors. Minerals in soil environments. Wisconsin: Soil Science Society of America, p 199–278.
Anand RR, Gilkes RJ, Armitage TM, Hillyer JW. 1985. Feldspar weathering in lateritic saprolite. Clays Clay Miner 33:31–43.
Bamhisel RI, Rich Cl. 1965. Gibbsite, bayerite, and nordstrandite formation as affected by anions, pH, and mineral surfaces. Soil Sci Soc Am Proc 29:531–534.
Brace R, Matijevic E. 1973. Aluminum hydrous oxide sols—I. J Inorg Chem 35:3691–3705.
Casey WH, Westrich HR, Arnold GW. 1988. Surface chemistry of labradorite feldspar reacted with aqueous solutions at pH = 2, 3, and 12. Geochim Cosmochim Acta 52:2795–2807.
Casey WH, Westrich HR, Arnold GW, Banfield JF. 1989. The surface chemistry of dissolving labradorite feldspar. Geochim Cosmochim Acta 53:821–832.
Chesworth W. 1972. The stability of gibbsite and boehmite at the surface of the earth. Clays Clay Miner 20:369–374.
Eggleton RA, Buseck PR. 1980. High resolution electron microscopy of feldspar weathering. Clays Clay Miner 28: 173–178.
Helgeson HC. 1971. Kinetics of mass transfer among silicates and aqueous solutions. Geochim Cosmochim Acta 35: 421–469.
Hellmann R, Eggleston CM, Hochella MF, Jr., Crerar DA. 1990. The formation of leached layers on albite surfaces during dissolution under hydrothermal conditions. Geochim Cosmochim Acta 54:1267–1281.
Hochella MF, Jr., Ponader HB, Turner AM, Harris DW. 1988. The complexity of mineral dissolution as viewed by high resolution scanning Auger microscopy: Labradorite under hydrothermal conditions. Geochim Cosmochim Acta 52: 385–394.
Hsu PH. 1966. Formation of gibbsite from aging hydroxyaluminum solutions. Soil Sci Soc Am Proc 30:173–176.
Hsu PH. 1989. Aluminum hydroxides and oxyhydroxides. In: Dixon JB, Weed SB, editors. Minerals in Soil Environments. Wisconsin: Soil Science Society of America, p 331–378.
Kawano M, and Tomita K. 1994. Growth of smectite from leached layer during experimental alteration of albite. Clays Clay Miner 42:7–17.
Lodding W. 1972. Conditions for the direct formation of gibbsite from K-feldspar-discussion. Am Miner 57:292–294.
Matijevic E. 1985. Production of monodispersed colloidal particles. Ann Rev Mater Sci 15:483–516.
Mogk DW, Locke WW, III. 1988. Application of Auger electron spectroscopy (AES) to naturally weathered hornblende. Geochim Cosmochim Acta 52:2537–2542.
Muir IJ, Bancroft GM, Nesbitt HW. 1989. Characteristics of altered labradorite surfaces by SIMS and XPS. Geochim Cosmochim Acta 53:1235–1241.
Muir IJ, Bancroft GM, Shotyk W, Nesbitt HW. 1990. A SIMS and XPS study of dissolving plagioclase. Geochim Cosmochim Acta 54:2247–2256.
Nesbitt HW, Muir IJ. 1988. SIMS depth profiles of weathered plagioclase and processes affecting dissolved Al and Si in some acidic soil solutions. Nature 334:336–338.
Nesbitt HW, Macrae ND, Shotyk W. 1991. Congruent and incongment dissolution of labradorite in dilute, acidic, salt solution. J Geol 99:429–442.
Nixon RA. 1979. Differences in incongruent weathering of plagioclase and microcline—cation leaching versus precipitates. Geology 7:221–224.
Petit J-C, Dran J-C, Paccagnella A, Della Mea G. 1989. Structural dependence of crystalline silicate hydration during aqueous dissolution. Earth Planet Sci Lett 93:292–298.
Schoen R, Roberson EC. 1970. Structures of aluminum hydroxide and geochemical implication. Am Mineral 55:43–77.
Shi JL, Gao JH, Lin ZX. 1989. Formation of monosized spherical aluminum hydroxide particles by urea method. Solid State Ionics 32/33:537–543.
Shibata K, Nozawa T. 1968. K-Ar ages of Yakujima granite, Kyushu, Japan. Bull Geol Surv Japan 19:21–24.
Taylor RM. 1987. Non-silicate oxides and hydroxides. In: Newman ACD, editor. Chemistry of clays and clay minerals. London: Mineralogical Society, p 129–201.
Tazaki K. 1976. Scanning electron microscopic study of formation of gibbsite from plagioclase. Inst Thermal Spring Res, Okayama Univ Paper 45:11–24.
Tazaki K. 1979. Micromorphology of halloysite produced by weathering of plagioclase in volcanic ash. In: Mortland MM, Farmer VC, editors. Proc Int Clay Conf, Oxford, 1978. Amsterdam: Elsevier, p 415–422.
Tazaki K, Fyfe WS. 1987a. Formation of primitive clay precursors on K-feldspar under extreme leaching conditions. In: Schultz LG, van Olphen H, Mumpton FA, editors. Proc Inter Clay Conf, Denver, 1985. Bloomington, IN: The Clay Minerals Society, p 53–58.
Tazaki K, Fyfe WS. 1987b. Primitive clay precursors formed on feldspar. Can J Earth Sci 24:506–527.
Violante A, Huang PM. 1985. Influence of inorganic and organic ligands on the formation of aluminum hydroxides and oxyhydrexides. Clays Clay Miner 33:181–192.
Violante A, Huang PM. 1992. Effect of tartaric acid and pH on the nature and physicochemical properties of shortrange ordered aluminum precipitation products. Clays Clay Miner 40:462–469.
Violante A, Huang PM. 1993. Formation mechanism of aluminum hydroxide polymorphs. Clays Clay Miner 41:590–597.
Violante A, Gianffeda L, Violante P 1993. Effect of prolonged aging the transformation of short-range ordered aluminum precipitation products formed in the presence of organic and inorganic ligands. Clays Clay Miner 41:353–359.
Watson JHL, Vallejo-Freire A, Souza Santos P, Parsons J. 1957. The fine structure and properties of fibrous alumina. Kolloid-Z 154:4–15.
Wilke BS, Schwertmann U, Murad E. 1978. An occurrence of polymorphic halloysite in granitic saprolite of the Bayerischer Wald, Germany. Clay Miner 13:67–77.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kawano, M., Tomita, K. Amorphous Aluminum Hydroxide Formed at the Earliest Weathering Stages of K-Feldspar. Clays Clay Miner. 44, 672–676 (1996). https://doi.org/10.1346/CCMN.1996.0440510
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1996.0440510