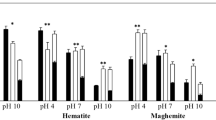
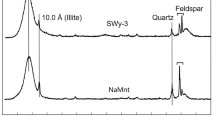
Abstract
The adsorption of arsenate (As(V)) on kaolinite, montmorillonite and illite was investigated at varying pH and competing anion concentration while holding As(V) concentration (6.7 × 10−7M), clay suspension density (2.5 g L−1) and ionic strength (0.1 M NaCl) constant. The effects of 2 concentrations of phosphate (P) or molybdate (Mo) (6.7 × 10−7 and 6.7 × 10−6M) on As(V) adsorption envelopes (adsorption vs. pH) gave evidence for direct competitive adsorption (in the case of As(V) + P) and possibly site-specific non-competitive adsorption (As(V) + Mo). Distinct As(V) adsorption maxima occurred at approximately pH 5.0 for kaolinite, 6.0 for montmorillonite and 6.5 for illite, and ranged from 0.15 to 0.22 mmol As(V) kg−1. When both As(V) and P were present at equimolar concentrations (6.7 × 10−7M), As(V) adsorption decreased slightly, whereas As(V) adsorption substantially decreased in binary As(V)/P systems when the P concentration was 6.7 × 10−6M, which was 10 times greater than As(V). The presence of Mo at equimolar (6.7 × 10−7 M) and 10 times greater (6.7 × 10−6M) concentrations than As(V) caused only slight decreases in As(V) adsorption because the Mo adsorption maximum occurred at pH < 4. The constant capacitance surface complexation model was applied to As(V) and P adsorption data and was used to predict As(V) adsorption at varying P concentrations. The model gave reasonable descriptions of As(V) adsorption on the 3 clay minerals at varying pH and in the presence of a competing oxyanion (P), indicating that surface complexation modeling may be useful in predicting As(V) adsorption in soils.
Similar content being viewed by others
References
Anderson MA, Malotky DT. 1979. The adsorption of protolyzable anions on hydrous oxides at the isoelectric pH. J Coll Interface Sci 72:413–427.
Barrow NJ. 1992. The effect of time on the competition between anions for sorption. J Soil Sci 43:421–428.
Bleam WF, Pfeffer PE, Goldberg S, Taylor RW, Dudley R. 1991. A 31P solid-state nuclear magnetic resonance study of phosphate adsorption at the boehmite/aqueous solution interface. Langmuir 7:1702–1712.
Chen YSR, Butler JN, Stumm W. 1973a. Adsorption of phosphate on alumina and kaolinite from dilute aqueous solutions. J Coll Interface Sci 43:421–436.
Chen YSR, Butler JN, Stumm W. 1973b. Kinetic study of phosphate reaction with aluminum oxide and kaolinite. Environ Sci Technol 7:327–332.
Davis JA, Kent DB. 1990. Surface complexation modeling in aqueous geochemistry. Rev Mineral 23:177–260.
Edzwald JK, Toensing DC, Leung MCY. 1976. Phosphate adsorption reactions with clay minerals. Environ Sci Technol 10:485–490.
Ferguson JF, Gavis J. 1972. A review of the arsenic cycle in natural waters. Water Res 6:1259–1274.
Frost RR, Griffin RA. 1977. Effect of pH on adsorption of arsenic and selenium from landfill leachate by clay minerals. Soil Sci Soc Am J 41:53–57.
Glaubig RA, Goldberg S. 1988. Determination of inorganic arsenic (III) and arsenic (III plus V) using automated hydride-generation atomic-absorption spectrometry. Soil Sci Soc Am J 52:536–537.
Goldberg S. 1986. Chemical modeling of arsenate adsorption on aluminum and iron oxide minerals. Soil Sci Soc Am J 50:1154–1157.
Goldberg S, Glaubig RA. 1988. Anion sorption on a calcareous, montmorillonitic soil-arsenic. Soil Sci Soc Am J 52: 1297–1300.
Herbelin AL, Westall JC. 1994. FITEQL: A computer program for the determination of chemical equilibrium constants from experimental data. Rep. 94-01, Oregon State University, Corvallis.
Hingston FJ, Posner AM, Quirk JP. 1971. Competitive adsorption of negatively charged ligands on oxide surfaces. Discuss Faraday Soc 52:334–342.
Hingston FJ, Posner AM, Quirk JR 1972. Anion adsorption by goethite and gibbsite. I. The role of the proton in determining adsorption envelopes. J Soil Sci 23:177–192.
Hsia TH, Lo SL, Lin CF, Lee DY. 1994. Characterization of arsenate adsorption on hydrous iron oxide using chemical and physical methods. Colloids Surf A: Physicochemical and Engineering Aspects 85:1–7.
Huang PM. 1975. Retention of arsenic by hydroxy-aluminum on surfaces of micaceous mineral colloids. Soil Sci Soc Am Proc 39:271–274.
Jacobs LW, Syers JK, Kenney DR. 1970. Arsenic sorption by soils. Soil Sci Soc Am Proc 34:750–754.
Keren R, Talpaz H. 1984. Boron adsorption by montmorillonite as affected by particle size. Soil Sci Soc Am J 48: 555–559.
Livesey NT, Huang PM. 1981. Adsorption of arsenate by soils and its relation to selected chemical properties and anions. Soil Sci 131:88–94.
Lumsdon DG, Fraser AR, Russell JD, Livesey NT. 1984. New infrared band assignments for the arsenate ion adsorbed on synthetic goethite. J Soil Sci 35:381–386.
Manning BA, Goldberg S. 1996. Modeling competitive adsorption of arsenate with phosphate and molybdate on oxide minerals. Soil Sci Soc Am J 60:121–131.
Masscheleyn PH, Delaune RD, Patrick WH Jr. 1991a. Effect of redox potential and pH on arsenic speciation and solubility in a contaminated soil. Environ Sci Technol 25:1414–1419.
Masscheleyn PH, Delaune RD, Patrick WH Jr. 1991b. Arsenic and selenium chemistry as affected by sediment redox potential and pH. J Environ Qual 20:522–527.
Michaels AS, Bolger JC. 1964. Particle interactions in aqueous kaolinite suspensions. Ind Eng Chem Fundam 3:14–20.
Motta MM, Miranda CF. 1989. Molybdate adsorption on kaolinite, montmorillonite, and illite: Constant capacitance modeling. Soil Sci Soc Am J 53:380–385.
Muljadi D, Posner AM, Quirk JP. 1966. The mechanism of phosphate adsorption on kaolinite, gibbsite and pseudoboehmite. I. The isotherms and the effect of pH on adsorption. J Soil Sci 17:212–229.
Perrott KW, Langdon AG, Wilson AT. 1974. Sorption of phosphate by aluminum and iron(III)-hydroxy species on mica surfaces. Geoderma 12:223–231.
Rand B, Melton JE. 1975. Isoelectric point of the edge surface of kaolinite. Nature 257:214–216.
Roy WR, Hassett JJ, Griffin RA. 1986. Competitive coefficients for the adsorption of arsenate, molybdate, and phosphate mixtures by soils. Soil Sci Soc Am J 50:1176–1182.
Sadiq M, Zaida TH, Mian AA. 1983. Environmental behavior of arsenic in soils: Theoretical. Water Air Soil Pollut 20:369–377.
Schindler PW, Gamsjager H. 1972. Acid-base reactions of the TiO2 (anatase)-water interface and the point of zero charge of TiO2 suspensions. Kolloid Z Z Polym 250:759–763.
Stumm W, Hohl H, Dalang F. 1976. Interactions of metal ions with hydrous oxide surfaces. Croat Chem Acta 48: 491–504.
Stumm W, Huang CP, Jenkins SR. 1970. Specific chemical interaction affecting the stability of dispersed systems. Croat Chem Acta 42:223–245.
Stumm W, Kummert R, Sigg L. 1980. A ligand exchange model for the adsorption of inorganic and organic ligands at hydrous oxide interfaces. Croat Chem Acta 53:291–312.
Swartzen-AJlen LS, Matijevic E. 1974. Surface and colloid chemistry of clays. Chem Rev 74:385–400.
Waychunas GA, Rea BA, Fuller CC, Davis JA. 1993. Surface chemistry of ferrihydrite: Part 1. EXAFS studies of the geometry of coprecipitated and adsorbed arsenate. Geochim Cosmochim Acta 57:2251–2269.
Westall JC, Hohl H. 1980. A comparison of electrostatic models for the oxide/solution interface. Adv Coll Interface Sci 12:265–294.
Wieland E, Stumm W. 1992. Dissolution kinetics of kaolinite in acidic aqueous solutions at 25°C. Geochim Cosmochim Acta 56:3339–3355.
Xu H, Allard B, Grimvall A. 1988. Influence of pH and organic substance on the adsorption of As(V) on geologic materials. Water Air Soil Pollut 40:293–305.
Zachara JM, Cowan CE, Schmidt RL, Ainsworth CC. 1988. Chromate adsorption by kaolinite. Clays Clay Miner 36: 317–326.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Manning, B.A., Goldberg, S. Modeling Arsenate Competitive Adsorption on Kaolinite, Montmorillonite and Illite. Clays Clay Miner. 44, 609–623 (1996). https://doi.org/10.1346/CCMN.1996.0440504
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1996.0440504