Abstract
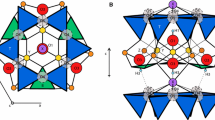
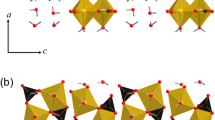
Riet veld X-ray powder diffraction (XRD) analysis has been evaluated as a procedure for characterizing Al-substituted goethite according to the Rietveld scale factor, unit-cell parameters, and atom positional parameters. The study was conducted with three synthetic goethite samples for which the degree of Al substitution for Fe determined by chemical analysis was 8.0 ± 0.4, 12.0 ± 0.4, and 20.1 ± 0.4 mole %. The weight fractions of crystalline material (WFCM) in the specimens, determined from the Rietveld scale factors after correcting for adsorbed water and impurities, were 0.878 (esd = 0.014), 0.919 (0.014), and 0.965 (0.015), respectively. The Al mole % substitutions, inferred from the Rietveld cell parameters according to the method of Schulze (1984), were 10.4 ± 2.5, 16.5 ± 2.6, and 17.1 ± 2.6, respectively. The cause of the significant difference between the second value and the chemical analysis result is not known. The atom positional parameters did not differ significantly within the sample suite and agreed satisfactorily with literature values. The results have demonstrated the value of using Rietveld XRD analysis to determine simultaneously the WFCM and Al mole % substitutions, as well as to confirm the non-hydrogen atom positions.
Similar content being viewed by others
References
Bish, D. L. and Howard S. A. (1988) Quantitative phase analysis using the Rietveld method: J. Appl. Crystallogr. 21, 86–91.
Busing, W. R. and Levy, H.A. (1958) A single crystal neuron diffraction study of diaspore, AlO(OH): Acta Crystallogr. 11, 798–803.
Cambier, P. (1986) Infrared study of goethites of varying crystallinity and particle size: II. Crystallographic and morphological changes in series of synthetic goethites: Clay Miner. 21, 201–210.
Cornell, R. M., Mann, S., and Skarnulis, A. J. (1983) A high resolution electron microscopy examination of domain boundaries in crystals of synthetic goethite: J. Chem. Soc. Faraday Trans. 1 79, 2679–2684.
Dollase, W. A. (1986) Correction of intensities for preferred orientation in powder diffractometry: Application of the March model: J. Appl. Crystallogr. 19, 267–272.
Forsyth, J. B., Hedley, I. G., and Johnson, C. E. (1968) The magnetic structure and hyperfine field of goethite (α-Fe-OOH): Proc. Phys. Soc. London, sect. C 1, 179–188.
Golden, D. C. (1978) Physical and chemical properties of aluminum-substituted goethite: Ph.D. thesis, North Carolina State University, Raleigh, North Carolina, 74 pp. (University Microfilms Inc., 78/20029).
Hill, R. J. (1979) Crystal structure refinement and electron density distribution in diaspore: Phys. Chem. Minerals 5, 179–200.
Hill, R. J. and Howard, C. J. (1986) A computer program for Rietveld analysis of fixed wavelength X-ray and neutron powder diffraction patterns: Version LHPM1: Australian Atomic Energy Commission Research Establishment, Lucas Heights, New South Wales, Australia.
Hill, R. J. and Howard, C. J. (1987) Quantitative phase analysis from neutron powder diffraction data using the Rietveld method: J. Appl. Crystallogr. 20, 467–474.
Hubbell, J. H., McMaster, W. H., Del Grande, N. K., and Mallett, J. H. (1974) International Tables for X-ray Crystallography (1974), Vol. IV: Kynoch Press, Birmingham, United Kingdom, 47–70.
Jordan, B., O’Connor, B. H., and Li Dey. (1990) Use of Rietveld pattern-fitting to determine the weight fraction of crystalline material in natural low-quartz specimens: Powder Diffraction 5, 64–69.
Klug, A. and Farkas, L. (1981) Structural investigations of polycrystalline diaspore samples by X-ray powder diffraction: Phys. Chem. Minerals 7, 138–140.
March, A. (1932) Mathematische Theorie der Regelung nach der Korngestalt bei affiner Deformation: Zeit. Kristallogr. 81, 285–297.
Megaw, H. D. (1973) Crystal Structures—A Working Approach: W. B. Sanders Co., Philadelphia, 356–359.
O’Connor, B. H. and Raven M. D. (1988) Application of the Rietveld refinement procedure in assaying powdered mixtures: Powder Diffraction 3, 2–6.
Reynolds, R. C. (1968) Effect of particle size on apparent lattice spacings: Acta Crystallogr. A24, 319–320.
Reitveld, H. M. (1969) A profile refinement method for nuclear and magnetic structures: J. Appl. Crystallogr. 2, 65–71.
Schulze, D. G. (1984) The influence of aluminum on iron oxides, VIII. Unit-cell dimensions of Al-substituted goethites and estimation of Al from them: Clays & Clay Minerals 32, 36–44.
Schulze, D. G. and Schwertmann, U. (1987) The influence of aluminum on iron oxides: XIII. Properties of goethites synthesised in 0.3 M KOH at 25°: Clay Miner. 22, 83–92.
Shannon, R. D. and Prewitt, C. T. (1969) Effective ionic radii in oxides and fluorides: Acta Crystallogr. B25, 925–946.
Trunz, V. (1976) Influence of crystallite size on the apparent basal spacings of kaolinite: Clays & Clay Minerals 24, 84–87.
Wiles, D. B. and Young, R. A. (1981) A new computer program for Rietveld analysis of X-ray powder diffraction patterns: J. Appl. Crystallogr. 14, 149–151.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fazey, P.G., O’Connor, B.H. & Hammond, L.C. X-Ray Powder Diffraction Rietveld Characterization of Synthetic Aluminum-Substituted Goethite. Clays Clay Miner. 39, 248–253 (1991). https://doi.org/10.1346/CCMN.1991.0390304
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1991.0390304